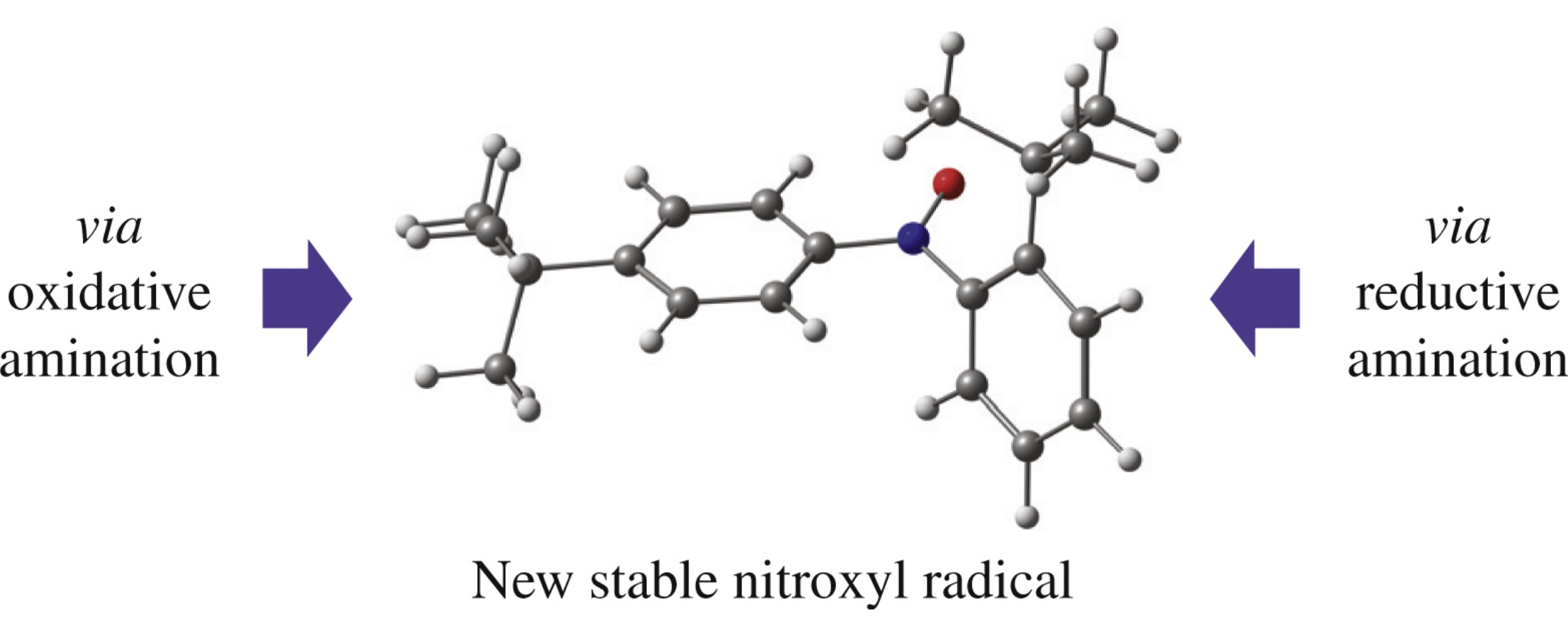
Abstract
The title compound, first example of a stable diarylnitroxyl with vacant para-position, was best synthesized by CuCl-assisted coupling of o-tert-butylnitrosobenzene and p-tert-butylphenylboronic acid followed by N-xidation of the thus obtained unsymmetrical diarylamine. ESR investigation showed that ortho-substituted aromatic ring is removed from the conjugation plane providing unusual stability of this radical.
References
1.

Denisov E.T.
Russian Chemical Reviews,
1996
2.

Surry D.S., Buchwald S.L.
Chemical Science,
2011
3.

Lalic G., Rucker R.
Synlett,
2012
4.

Correa A., Carril M., Bolm C.
Chemistry - A European Journal,
2008
5.
10.1016/j.mencom.2016.11.026_bib0025
Shekhar
The Chemistry of Anilines,
2007
6.

Ballester M., Riera J., Onrubia C.
Tetrahedron Letters,
1976
7.
![Bis[3-tert-butyl-5-(N-oxy-tert-butylamino)phenyl] nitroxide in a quartet ground state: a prototype for persistent high-spin poly[(oxyimino)-1,3-phenylenes]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ishida T., Iwamura H.
Journal of the American Chemical Society,
1991
8.

Tanaka M., Matsuda K., Itoh T., Iwamura H.
Journal of the American Chemical Society,
1998
9.

Delen Z., Lahti P.M.
Journal of Organic Chemistry,
2006
10.

Oka H., Kouno H., Tanaka H.
Journal of Materials Chemistry A,
2007
11.

Rajca A., Shiraishi K., Rajca S.
Chemical Communications,
2009
12.

Golubev V.A., Sen’ V.D.
Russian Journal of Organic Chemistry,
2013
13.

Ley S.V., Thomas A.W.
Angewandte Chemie - International Edition,
2003
14.

Vantourout J.C., Law R.P., Isidro-Llobet A., Atkinson S.J., Watson A.J.
Journal of Organic Chemistry,
2016
15.

Qiao J., Lam P.
Synthesis,
2010
16.
Tsedilin A.M., Fakhrutdinov A.N., Eremin D.B., Zalesskiy S.S., Chizhov A.O., Kolotyrkina N.G., Ananikov V.P.
Mendeleev Communications,
2015