Abstract
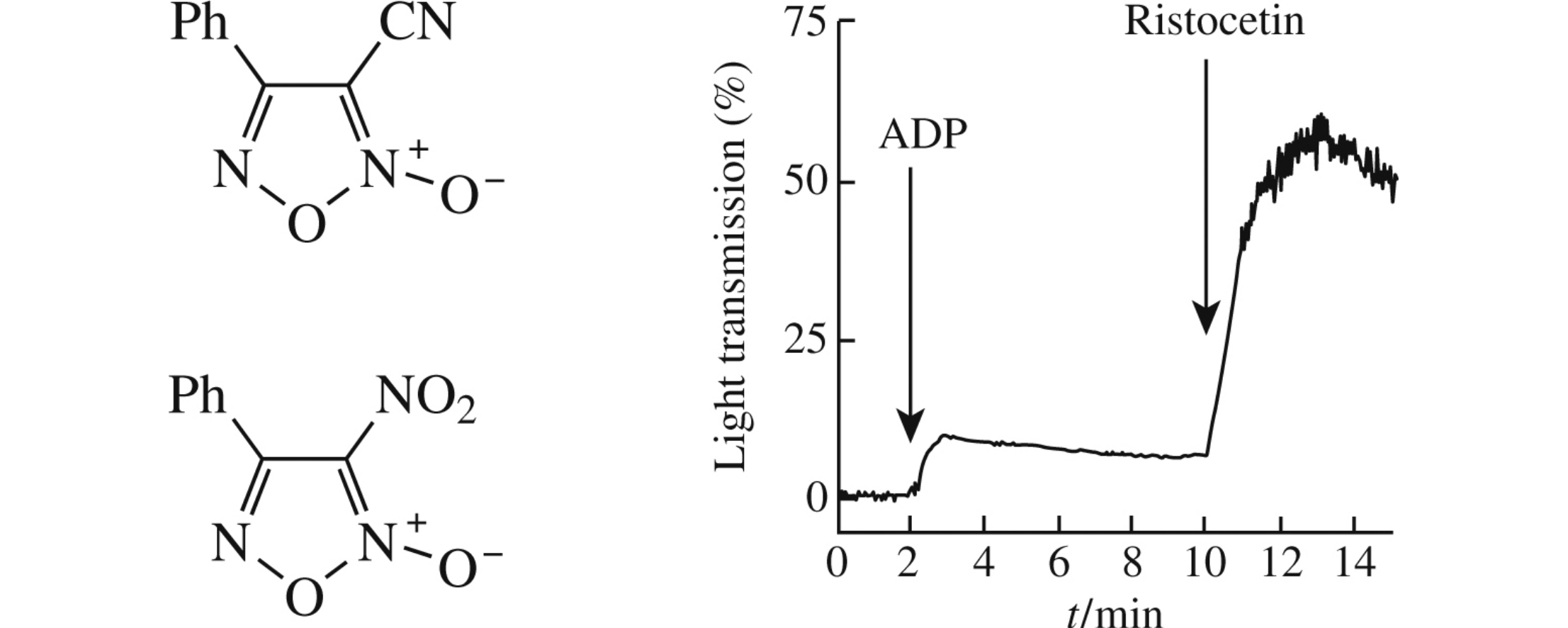
The measurement of the antiaggregant properties of 3-cyano-4-phenylfuroxan and 3-nitro-4-phenylfuroxan has demonstrated that these compounds effectively inhibit platelet agglutination induced by adenosine diphosphate and adrenaline but not ristocetin. When collagen was used as an inducer, only a slight delay of aggregate formation was observed. Therefore, these furoxan derivatives could be considered as a basis for the development of next generation agents with improved antiaggregant activity.
References
1.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
2.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
3.

Jovene C., Chugunova E.A., Goumont R.
Mini-Reviews in Medicinal Chemistry,
2013
4.

Cerecetto H., Porcal W.
Mini-Reviews in Medicinal Chemistry,
2005
5.

Gasco A., Fruttero R., Sorba G., Di Stilo A., Calvino R.
Pure and Applied Chemistry,
2004
6.

Schiefer I.T., VandeVrede L., Fa’ M., Arancio O., Thatcher G.R.
Journal of Medicinal Chemistry,
2012
7.

Lai Y., Shen L., Zhang Z., Liu W., Zhang Y., Ji H., Tian J.
Bioorganic and Medicinal Chemistry Letters,
2010
8.
10.1016/j.mencom.2016.11.018_sbref0020b
Aguirre
Pharmazie,
2006
9.

Zhao J., Gou S., Sun Y., Fang L., Wang Z.
Inorganic Chemistry,
2012
10.

Treger R.S., Cook A., Rai G., Maloney D.J., Simeonov A., Jadhav A., Thomas C.J., Williams D.L., Cappello M., Vermeire J.J.
International Journal for Parasitology: Drugs and Drug Resistance,
2012
11.

Dos Santos J.L., Lanaro C., Chelucci R.C., Gambero S., Bosquesi P.L., Reis J.S., Lima L.M., Cerecetto H., González M., Costa F.F., Chung M.C.
Journal of Medicinal Chemistry,
2012
12.

Rai G., Thomas C.J., Leister W., Maloney D.J.
Tetrahedron Letters,
2009
13.

Lazzarato L., Cena C., Rolando B., Marini E., Lolli M.L., Guglielmo S., Guaita E., Morini G., Coruzzi G., Fruttero R., Gasco A.
Bioorganic and Medicinal Chemistry,
2011
14.

Zou X., Peng S., Hu C., Tan L., Deng H., Li Y.
Bioorganic and Medicinal Chemistry Letters,
2011
15.

Chegaev K., Rolando B., Guglielmo S., Fruttero R., Gasco A.
Journal of Heterocyclic Chemistry,
2009
16.

Balbo S., Lazzarato L., Di Stilo A., Fruttero R., Lombaert N., Kirsch-Volders M.
Toxicology Letters,
2008
17.

Buonsanti M.F., Bertinaria M., Di Stilo A., Cena C., Fruttero R., Gasco A.
Journal of Medicinal Chemistry,
2007
18.

Turnbull C.M., Cena C., Fruttero R., Gasco A., Rossi A.G., Megson I.L.
British Journal of Pharmacology,
2006
19.

Krause P., Wätzig E., Acil H., König S., Unthan-Fechner K., Tsikas D., Probst I.
Nitric Oxide - Biology and Chemistry,
2010
20.
Cena C., Bertinaria M., Boschi D., Giorgis M., Gasco A.
Arkivoc,
2006
21.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
22.

Ferioli R., Fazzini A., Folco G.C., Fruttero R., Calvino R., Gasco A., Bongrani S., Civelli M.
Pharmacological Research,
1993
23.

Ferioli R., Folco G.C., Ferretti C., Gasco A.M., Medana C., Fruttero R., Civelli M., Gasco A.
British Journal of Pharmacology,
1995
24.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
25.

Ovchinnikov I.V., Finogenov A.O., Epishina M.A., Kulikov A.S., Strelenko Y.A., Makhova N.N.
Russian Chemical Bulletin,
2009
26.

Puri R.N., Colman R.W., Liberman M.A.
Critical Reviews in Biochemistry and Molecular Biology,
1997
27.

Ueno M., Kodali M., Tello-Montoliu A., Angiolillo D.J.
Journal of Atherosclerosis and Thrombosis,
2011