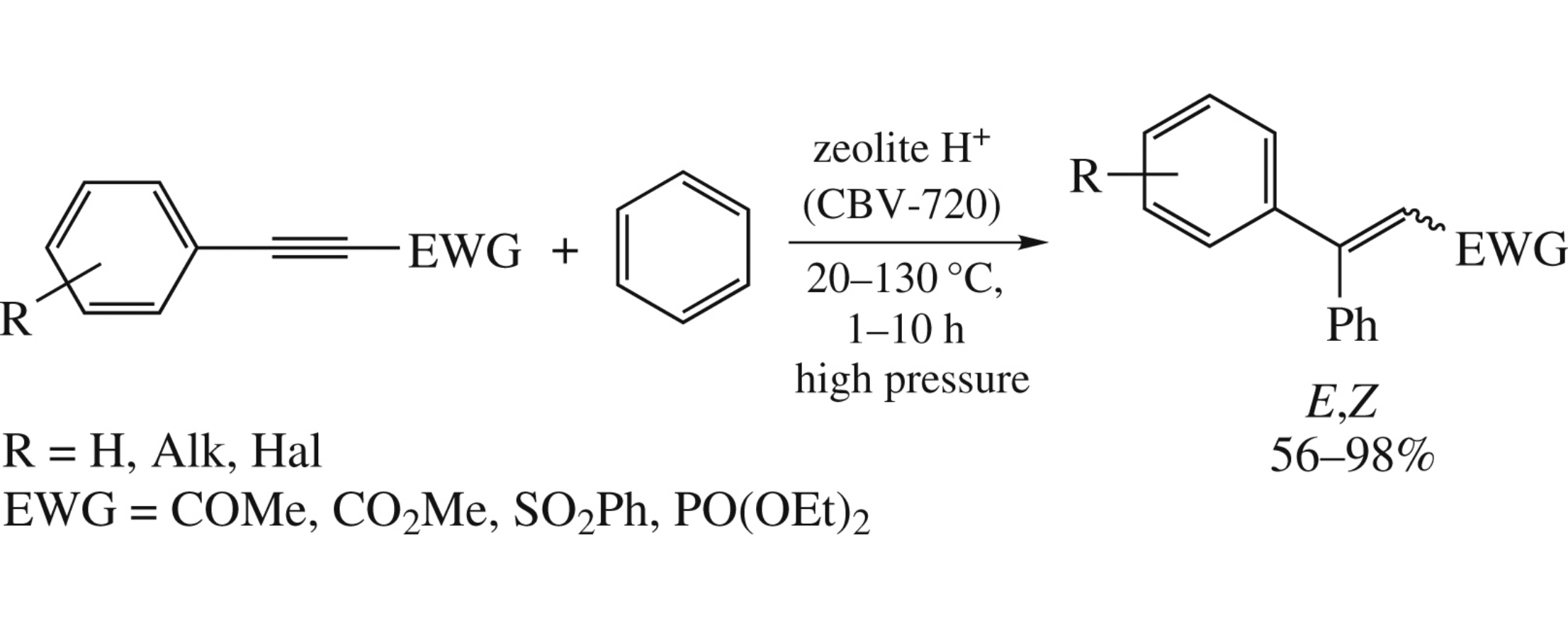
Abstract
Arylacetylenes, conjugated with electron-withdrawing groups, ArC≡C(EWG) [EWG = SO2Ph, PO(OEt)2, COMe, CO2Me] in reaction with benzene at room temperature or 130°C (glass high pressure tube) for 1–10h under the action of acidic zeolite HUSY (Si /Al ratio is 15) afford products of hydrophenylation of acetylene bond Ar(Ph)C=CH(EWG) in yields of 56–98%.
References
1.

Smith K., El-Hiti G.A.
Green Chemistry,
2011
2.

Wang W., Hunger M.
Accounts of Chemical Research,
2008
3.

Turro N.J.
Accounts of Chemical Research,
2000
4.

Sartori G., Maggi R.
Chemical Reviews,
2011
5.

Zhou C., Xia X., Lin C., Tong D., Beltramini J.
Chemical Society Reviews,
2011
6.

Sartori G., Maggi R.
Chemical Reviews,
2006
7.

Sartori G., bigi F., Pastorío A., Porta C., Arienti A., Maggi R., Moretti N., Gnappi G.
Tetrahedron Letters,
1995
8.

Koltunov K.Y., Walspurger S., Sommer J.
Chemical Communications,
2004
9.

Mohan D.C., Patil R.D., Adimurthy S.
European Journal of Organic Chemistry,
2012
10.

Chassaing S., Kumarraja M., Pale P., Sommer J.
Organic Letters,
2007
11.

Koltunov K.Y., Walspurger S., Sommer J.
Journal of Molecular Catalysis A Chemical,
2006
12.

Koltunov K.Y., Walspurger S., Sommer J.
Tetrahedron Letters,
2005
13.

Sani Souna Sido A., Chassaing S., Kumarraja M., Pale P., Sommer J.
Tetrahedron Letters,
2007
14.

Sani-Souna-Sido A., Chassaing S., Pale P., Sommer J.
Applied Catalysis A: General,
2008
15.
![Click chemistry in CuI-zeolites: the Huisgen [3 + 2]-cycloaddition.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chassaing S., Kumarraja M., Sani Souna Sido A., Pale P., Sommer J.
Organic Letters,
2007
16.

Bénéteau V., Olmos A., Boningari T., Sommer J., Pale P.
Tetrahedron Letters,
2010
17.

Boningari T., Olmos A., Reddy B.M., Sommer J., Pale P.
European Journal of Organic Chemistry,
2010
18.

Kuhn P., Pale P., Sommer J., Louis B.
Journal of Physical Chemistry C,
2009
19.

Kuhn P., Alix A., Kumarraja M., Louis B., Pale P., Sommer J.
European Journal of Organic Chemistry,
2009
20.

Patil M.K., Keller M., Reddy B.M., Pale P., Sommer J.
European Journal of Organic Chemistry,
2008
21.

Alix A., Chassaing S., Pale P., Sommer J.
Tetrahedron,
2008
22.

Vasilyev A.V.
Russian Chemical Reviews,
2013
23.

Ryabukhin D.S., Vasilyev A.V.
Tetrahedron Letters,
2015
24.
Kalugina A.V., Ryabukhin D.S., Artamonova T.O., Khodorkovsky M.A., Zarubin M.Y., Vasilyev A.V.
Mendeleev Communications,
2014
25.

Shchukin A.O., Vasil’ev A.V., Grinenko E.V.
Russian Journal of Organic Chemistry,
2010