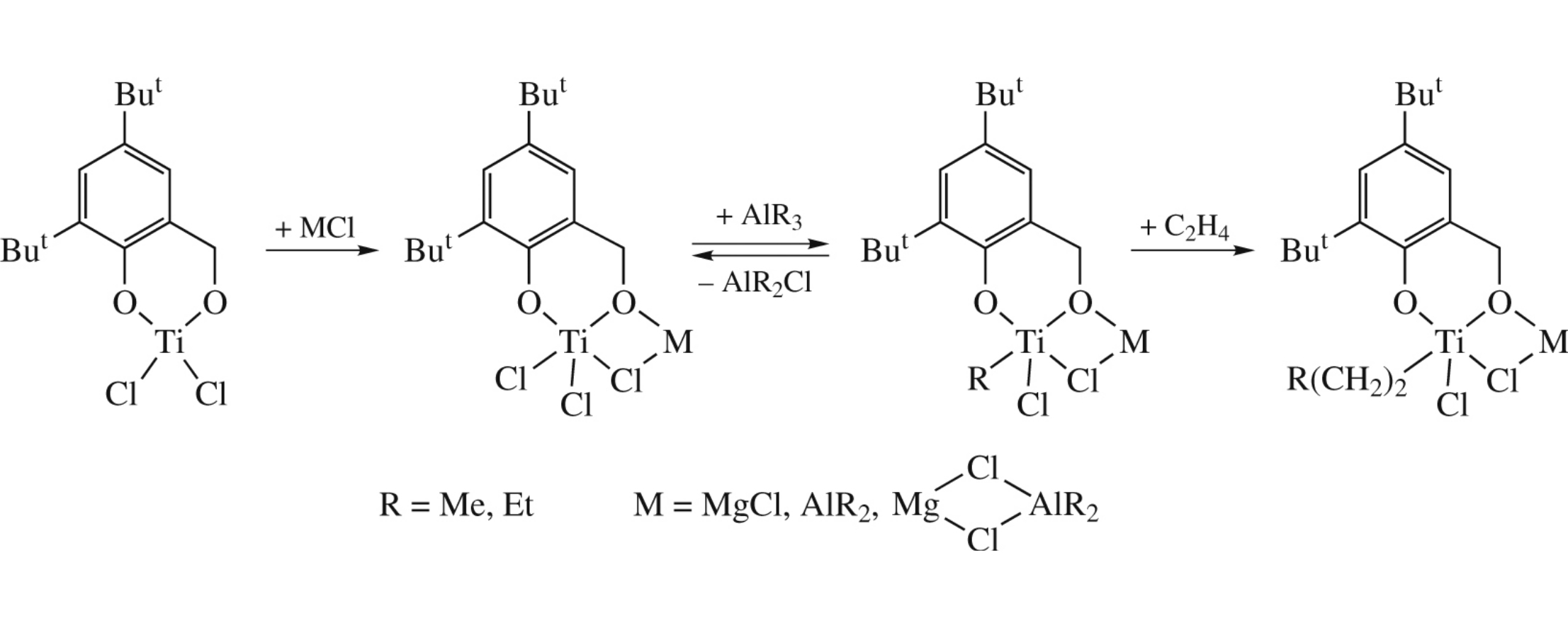
Abstract
The quantum-chemical modeling of post-titanocene catalytic systems for alkene polymerization suggests that the activation effect of Mg-containing compounds in alkene polymerization can be caused by a greater exothermicity of Cl ligand exchange in bi- and trinuclear heterocomplexes of Ti, Mg and Al for the alkyl group of AlR3 with respect to a Ti complex and their higher stability and reactivity toward alkenes, as compared to Ti and Al compounds formed in the Mg-free system.
References
1.

Solov’ev M.V., Gagieva S.C., Tuskaev V.A., Bravaya N.M., Gadalova O.E., Khrustalev V.N., Borissova A.O., Bulychev B.M.
Russian Chemical Bulletin,
2011
2.

Tuskaev V.A., Gagieva S.C., Solov'ev M.V., Kurmaev D.A., Kolosov N.A., Fedyanin I.V., Bulychev B.M.
Journal of Organometallic Chemistry,
2015
3.

Tuskaev V.A., Gagieva S.C., Maleev V.I., Borissova A.O., Solov'ev M.V., Starikova Z.A., Bulychev B.M.
Polymer,
2013
4.
Ustynyuk L.Y., Bulychev B.M.
Mendeleev Communications,
2015
5.

Ustynyuk L.Y., Bulychev B.M.
Journal of Organometallic Chemistry,
2015
6.
Ustynyuk L.Y., Bulychev B.M.
Mendeleev Communications,
2016
7.

Laikov D.N.
Chemical Physics Letters,
1997
8.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
9.

Kissin Y.V., Nowlin T.E., Mink R.I., Brandolini A.J.
Macromolecules,
2000
10.

Kissin Y.V., Mink R.I., Brandolini A.J., Nowlin T.E.
Journal of Polymer Science, Part A: Polymer Chemistry,
2009