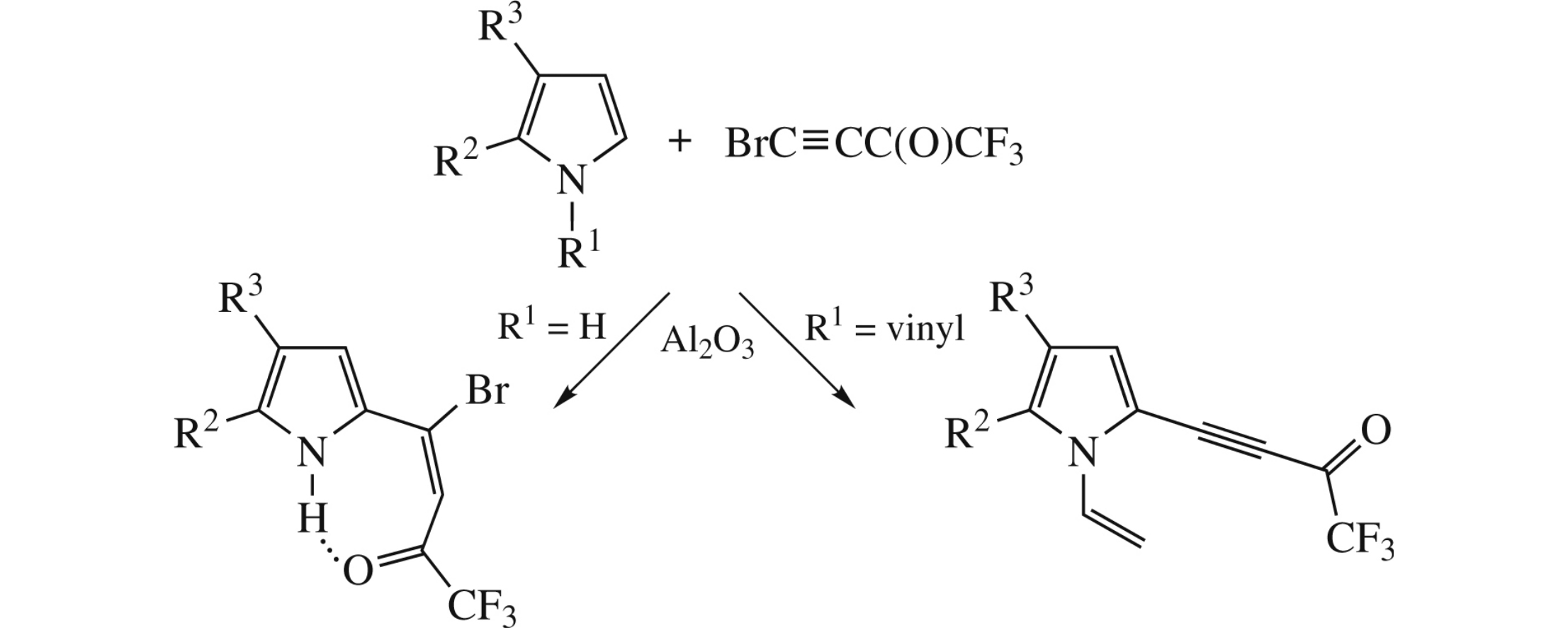
Abstract
Cross-coupling of NH-pyrroles with 1-bromo-2-(trifluoroacetyl) acetylene in the Al2O3 medium affords (E)-4-bromo-1,1,1-trifluoro-4-(pyrrol-2-yl)but-3-en-2-ones instead of the expected 2-ethynylated products. Quantum-chemical analysis shows strong intramolecular hydrogen bonding between NH and trifluoroacetyl groups, higher NH acidity and deeper charge transfer to the ethenyl moiety, which determines the reaction pathway.
References
1.

Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Ushakov I.A.
Tetrahedron Letters,
2004
2.
Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Sinegovskaya L.M., Potekhin K.A., Fedyanin I.V.
Mendeleev Communications,
2005
3.

Sobenina L.N., Demenev A.P., Mikhaleva A.I., Ushakov I.A., Vasil’tsov A.M., Ivanov A.V., Trofimov B.A.
Tetrahedron Letters,
2006
4.

Petrova O.V., Sobenina L.N., Ushakov I.A., Mikhaleva A.I.
Russian Journal of Organic Chemistry,
2008
5.
10.1016/j.mencom.2016.11.006_bib0025
Trofimov
Chemistry of Pyrroles,
2014
6.
10.1016/j.mencom.2016.11.006_bib0030
Trofimov
2009
7.

Sobenina L.N., Tomilin D.N., Trofimov B.A.
Russian Chemical Reviews,
2014
8.

Gotsko M.D., Sobenina L.N., Tomilin D.N., Ushakov I.A., Dogadina A.V., Trofimov B.A.
Tetrahedron Letters,
2015
9.

Tomilin D.N., Pigulski B., Gulia N., Arendt A., Sobenina L.N., Mikhaleva A.I., Szafert S., Trofimov B.A.
RSC Advances,
2015
10.

Passarella D., Lesma G., Deleo M., Martinelli M., Silvani A.
Journal of the Chemical Society Perkin Transactions 1,
1999
11.

Cordell G.A.
Journal of Organic Chemistry,
1975
12.
10.1016/j.mencom.2016.11.006_bib0060
Bird
1984
13.

Espinosa E., Molins E., Lecomte C.
Chemical Physics Letters,
1998