Abstract
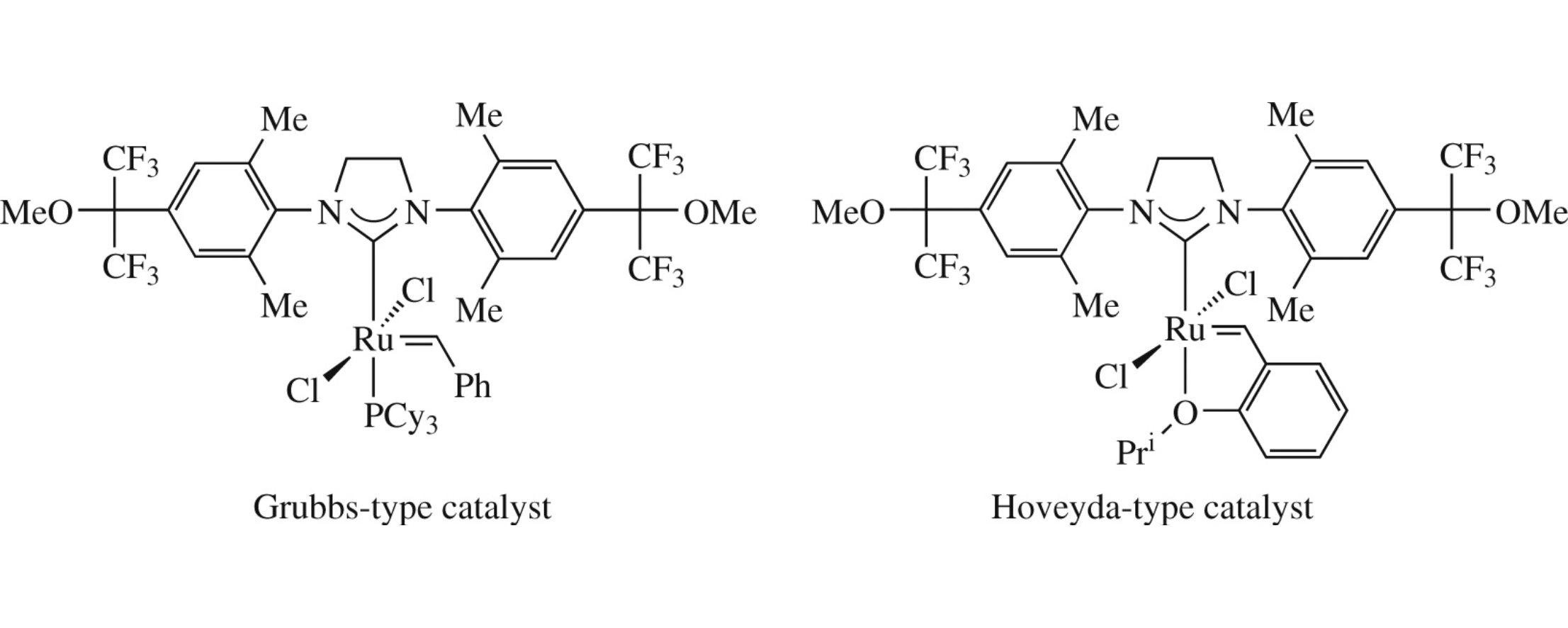
New olefin metathesis catalysts, analogues of Grubbs II ones, bearing hexafluoroisopropylmethoxy groups in NHC ligand, provide high conversions in model ring closing metathesis of diallylmalonate and cross metathesis of allylbenzene with 1,3-diacetoxybut-2-ene.
References
1.

Schrock R.R.
Angewandte Chemie - International Edition,
2006
2.

Grubbs R.H.
Angewandte Chemie - International Edition,
2006
3.
10.1016/j.mencom.2016.11.004_sbref0005c
Connon
2004
4.
10.1016/j.mencom.2016.11.004_sbref0005d
Olefin Metathesis Theory and Practice,
2014
5.

Vougioukalakis G.C., Grubbs R.H.
Chemical Reviews,
2009
6.
10.1016/j.mencom.2016.11.004_sbref0010b
Fischmeister
Metathesis Chemistry: From Nanostructure Design to Synthesis of Advanced Materials,
2007
7.

8.

Leitgeb A., Wappel J., Slugovc C.
Polymer,
2010
9.

Samojłowicz C., Bieniek M., Grela K.
Chemical Reviews,
2009
10.

Deraedt C., d'Halluin M., Astruc D.
European Journal of Inorganic Chemistry,
2013
11.
10.1016/j.mencom.2016.11.004_sbref0020a
Kirsch
Modern Fluoroorganic Chemistry,
2004
12.

Kirk K.L.
Journal of Fluorine Chemistry,
2006
13.

O'Hagan D.
Chemical Society Reviews,
2008
14.

Nie J., Guo H., Cahard D., Ma J.
Chemical Reviews,
2010
15.
10.1016/j.mencom.2016.11.004_sbref0025a
Uneyama
Organofluorine Chemistry,
2006
16.

Jagodzinska M., Huguenot F., Candiani G., Zanda M.
ChemMedChem,
2009
17.

Tuba R., Corrêa da Costa R., Bazzi H.S., Gladysz J.A.
ACS Catalysis,
2011
18.

Tuba R., Brothers E.N., Reibenspies J.H., Bazzi H.S., Gladysz J.A.
Inorganic Chemistry,
2012
19.

Matsugi M., Kobayashi Y., Suzumura N., Tsuchiya Y., Shioiri T.
Journal of Organic Chemistry,
2010
20.

Kvíčala J., Schindler M., Kelbichová V., Babuněk M., Rybáčková M., Kvíčalová M., Cvačka J., Březinová A.
Journal of Fluorine Chemistry,
2013
21.

Lim J., Lee S.S., Ying J.Y.
Chemical Communications,
2008
22.

Yao Q., Zhang Y.
Journal of the American Chemical Society,
2003
23.

Yang L., Mayr M., Wurst K., Buchmeiser M.R.
Chemistry - A European Journal,
2004
24.

Halbach T.S., Mix S., Fischer D., Maechling S., Krause J.O., Sievers C., Blechert S., Nuyken O., Buchmeiser M.R.
Journal of Organic Chemistry,
2005
25.

Monfette S., Camm K.D., Gorelsky S.I., Fogg D.E.
Organometallics,
2009
26.

Fustero S., Simón-Fuentes A., Barrio P., Haufe G.
Chemical Reviews,
2014
27.
10.1016/j.mencom.2016.11.004_sbref0045b
Siano
Cent. Eur. J. Chem.,
2011
28.

Fürstner A., Ackermann L., Gabor B., Goddard R., Lehmann C.W., Mynott R., Stelzer F., Thiel O.R.
Chemistry - A European Journal,
2001
29.

Ritter T., Day M.W., Grubbs R.H.
Journal of the American Chemical Society,
2006
30.

Anderson D., O'Leary D., Grubbs R.
Chemistry - A European Journal,
2008
31.

Masoud S.M., Mailyan A.K., Dorcet V., Roisnel T., Dixneuf P.H., Bruneau C., Osipov S.N.
Organometallics,
2015
32.

Gilbert E.E., Jones E.S., Sibilia J.P.
Journal of Organic Chemistry,
1965
33.

Chkanikov N.D., Sviridov V.D., Zelenin A.E., Galakhov M.V., Kolomiets A.F., Fokin A.V.
Russian Chemical Bulletin,
1990
34.
![A Series of Well‐Defined Metathesis Catalysts–Synthesis of [RuCl2(CHR′)(PR3)2] and Its Reactions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Schwab P., France M.B., Ziller J.W., Grubbs R.H.
Angewandte Chemie International Edition in English,
1995
35.

Kingsbury J.S., Harrity J.P., Bonitatebus P.J., Hoveyda A.H.
Journal of the American Chemical Society,
1999
36.

Ritter T., Hejl A., Wenzel A.G., Funk T.W., Grubbs R.H.
Organometallics,
2006
37.

Scholl M., Ding S., Lee C.W., Grubbs R.H.
Organic Letters,
1999
38.

Garber S.B., Kingsbury J.S., Gray B.L., Hoveyda A.H.
Journal of the American Chemical Society,
2000