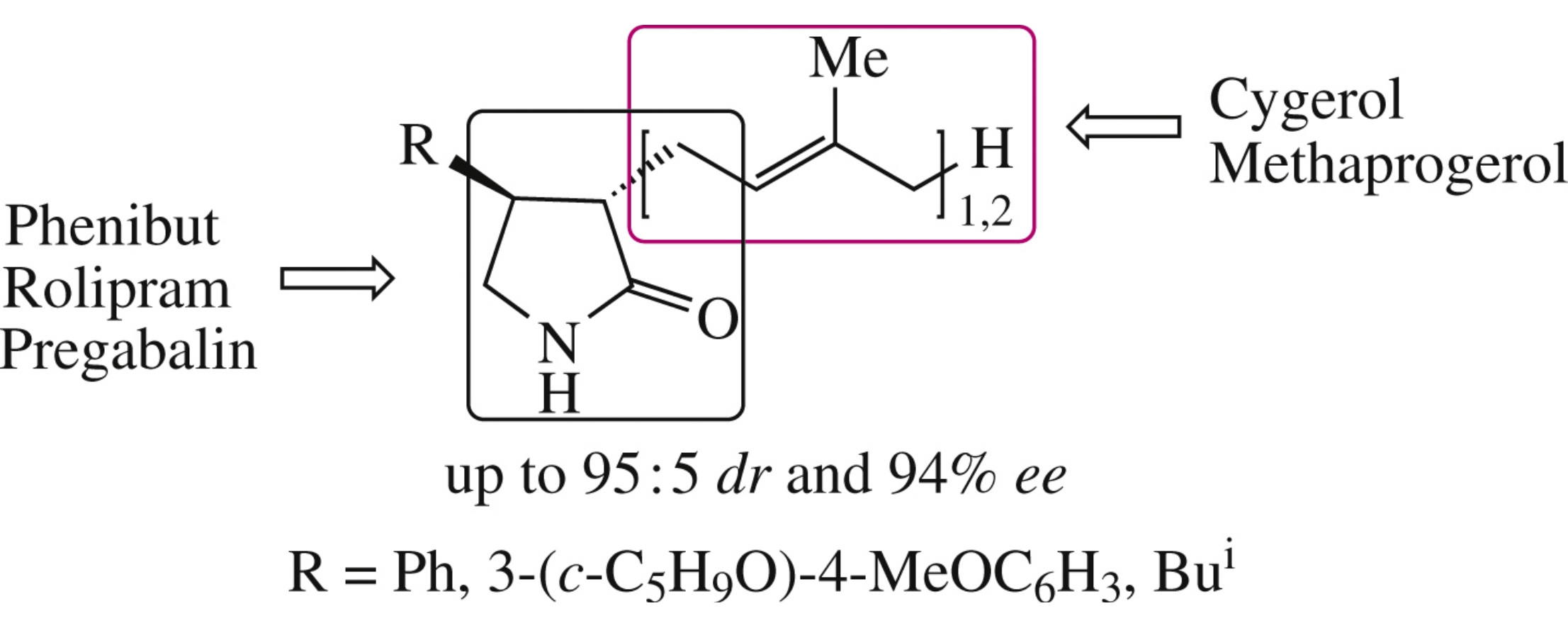
Abstract
Pharmacology-relevant compounds bearing structural fragments of racetam nootropics, wound-healing acyclic isoprenoids and neurotropic GABA analogues, were enantioselectively (up to 94% ee) synthesized from available and inexpensive precursors.
References
1.

Gouliaev A.H., Senning A.
Brain Research Reviews,
1994
2.

Giurgea C., Salama M.
Progress in Neuro-Psychopharmacology,
1977
3.

Winblad B.
CNS Drug Reviews,
2006
4.

Waegemans T., Wilsher C.R., Danniau A., Ferris S.H., Kurz A., Winblad B.
Dementia and Geriatric Cognitive Disorders,
2002
5.

Malykh A.G., Sadaie M.R.
Drugs,
2010
6.

Wheble P.C., Sena E.S., Macleod M.R.
Cerebrovascular Diseases,
2007
7.

Gualtieri F., Manetti D., Romanelli M., Ghelardini C.
Current Pharmaceutical Design,
2002
8.

De Reuck J., Van Vleymen B.
Pharmacopsychiatry,
1999
9.

Koskiniemi M., Van Vleymen B., Hakamies L., Lamusuo S., Taalas J.
Journal of Neurology, Neurosurgery and Psychiatry,
1998
10.

Fujii H.
Topics in Current Chemistry,
2010
11.
10.1016/j.mencom.2016.11.003_bib0055
Contreras
The Practice of Medicinal Chemistry,
2008
12.
L. S. Bondar and R. A. Okunev, British Patent GB 1 173 419, 1969.(Chem. Abstr., 1970, 72, 54770f).
13.
L. S. Bondar and R. A. Okunev, German Patent DE 1 801 868, 1970.(Chem. Abstr., 1970, 73, 34814r).
14.
L. S. Bondar and R. A. Okunev, Avtorskoe svidetel'stvo SSSR 333162, 1972.(Chem. Abstr., 1972, 77, 100910e).
15.
J. Nichols and G. F. Bulbenko, German Patent DE 2 335 067, 1974 (Chem. Abstr., 1976, 80, 146380x).
16.
L. S. Bondar and R. A. Okunev, German Patent DE 2 153 832, 1973.(Chem. Abstr., 1973, 79, 42708y).
17.
L. S. Bondar and R. A. Okunev, US Patent 3991086, 1976.(Chem. Abstr., 1976, 86, 105925j).
18.
L. S. Bondar, R.A. Okunev, L.V. Polezhaev, S.P. Kolchin, L.V. Cherkasova and L. F. Nikolaeva, US Patent 4 495 201, 1985.(Chem. Abstr., 1985, 102, 172652e).
19.
B. B. Smirnov, G.V. Kryshtal, A.G. Konopljannikov, S.G. Zlotin and G. M. Zhdankina, RF Patent RU 2 301 667, 2007.(Chem. Abstr., 2007, 147, 110262).
20.

Kryshtal G.V., Zhdankina G.M., Konoplyannikov A.G., Tartakovsky V.A., Serebryakov E.P., Smirnov B.B., Konoplyannikova O.A., Agaeva E.V., Zlotina S.G.
Russian Chemical Bulletin,
2012
21.

Hamandi K., Sander J.W.
Seizure : the journal of the British Epilepsy Association,
2006
22.

Lapin I.
CNS Drug Reviews,
2006
23.

Zhu J., Mix E., Winblad B.
CNS Drug Reviews,
2001
24.

Ohsawa F., Yamauchi M., Nagaso H., Murakami S., Baba J., Sawa A.
The Japanese Journal of Pharmacology,
1998
25.

Taylor C.P., Vartanian M.G., Po-Wai Y., Bigge C., Suman-Chauhan N., Hill D.R.
Epilepsy Research,
1993
26.
![Efficient synthesis of 3-mono and disubstituted lactams using meerwein eschenmoser [3,3] sigmatropic rearrangements.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Coates B., Montgomery D.J., Stevenson P.J.
Tetrahedron,
1994
27.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
28.

Berner O., Tedeschi L., Enders D.
European Journal of Organic Chemistry,
2002
29.
10.1016/j.mencom.2016.11.003_sbref0120a
Halimehjani
RSC Adv.,
2014
30.
10.1016/j.mencom.2016.11.003_sbref0120b
Halimehjani
RSC Adv.,
2014
31.

Halimehjani A.Z., Namboothiri I.N., Hooshmand S.E.
RSC Advances,
2014
32.

Sukhorukov A.Y., Sukhanova A.A., Zlotin S.G.
Tetrahedron,
2016
33.

Okino T., Hoashi Y., Furukawa T., Xu X., Takemoto Y.
Journal of the American Chemical Society,
2004
34.

Okino T., Hoashi Y., Takemoto Y.
Journal of the American Chemical Society,
2003
35.

Hestericová M., Šebesta R.
Tetrahedron,
2014
36.

Liu J., Wang X., Ge Z., Sun Q., Cheng T., Li R.
Tetrahedron,
2011
37.

Jakubec P., Cockfield D.M., Helliwell M., Raftery J., Dixon D.J.
Beilstein Journal of Organic Chemistry,
2012
38.
L. Thijs, US Patent 312 560 A1, 2009.
39.

Li F., Li Y., Jia Z., Xu M., Tian P., Lin G.
Tetrahedron,
2011
40.

41.

Perlikowska R., Piekielna J., Mazur M., Koralewski R., Olczak J., do Rego J., Fichna J., Modranka J., Janecki T., Janecka A.
Bioorganic and Medicinal Chemistry,
2014
42.

Hynes P.S., Stupple P.A., Dixon D.J.
Organic Letters,
2008
43.

Padwa A., Price A.T.
Journal of Organic Chemistry,
1998
44.

Civitavecchia A., Martelli G., Orena M., Rinaldi S.
Amino Acids,
2014