Abstract
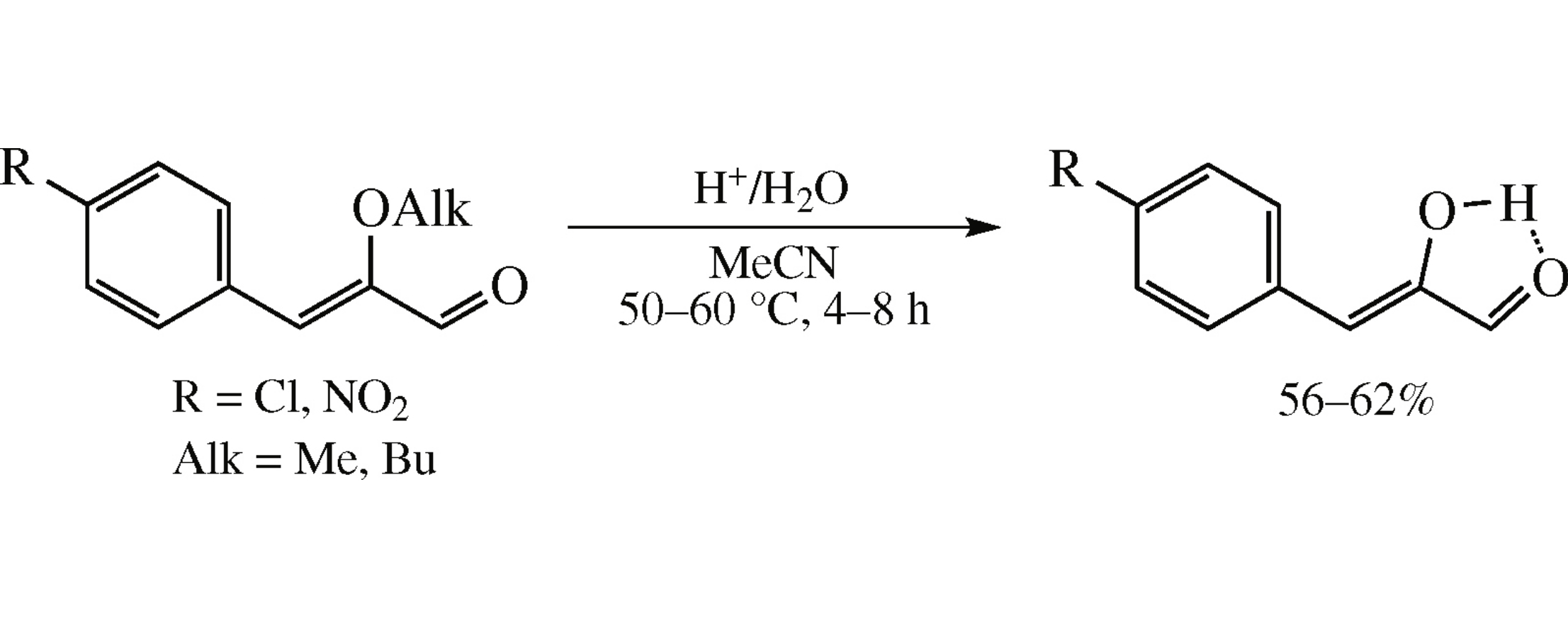
Hydrolysis of the C=C bond in 2-alkoxy-3-aryl(hetaryl)-propenals, depending upon substituents in the molecule, can proceed according to Markovnikov or Michael rule or with heterolytic opening of the cycle (in the case of 2-alkoxy-3-furylpropenal). Hydrolysis of 3-phenyl substituted alkoxypropenals bearing electron-withdrawing substituents (Cl, NO2) in the p-position has allowed a method for the preparation of corresponding benzylglyoxals (stable in enol form) to be developed.
References
1.

Sy L., Brown G.D.
Phytochemistry,
1999
2.

Kim H., Ralph J., Lu F., Ralph S.A., Boudet A., MacKay J.J., Sederoff R.R., Ito T., Kawai S., Ohashi H., Higuchi T.
Organic and Biomolecular Chemistry,
2002
3.

Grant A.J., Lerner L.M.
Biochemistry,
1979
4.

Mamashvili T.N., Keiko N.A., Sarapulova G.I., Voronkov M.G.
Russian Chemical Bulletin,
1998
5.

Zbiral E.
Tetrahedron Letters,
1965
6.
Keiko N.A., Stepanova L.G., Verochkina E.A., Larina L.I.
Arkivoc,
2010
7.

Eftekhari-Sis B., Zirak M., Akbari A.
Chemical Reviews,
2013
8.

Fröhlich L.G., Kotsonis P., Traub H., Taghavi-Moghadam S., Al-Masoudi N., Hofmann H., Strobel H., Matter H., Pfleiderer W., Schmidt H.H.
Journal of Medicinal Chemistry,
1999
9.

Girotra N.N., Patchett A.A., Zimmerman S.B., Achimov D.L., Wendler N.L.
Journal of Medicinal Chemistry,
1980
10.

Molina P., Fresneda P.M., García-Zafra S.
Tetrahedron Letters,
1996
11.

Mosrin M., Bresser T., Knochel P.
Organic Letters,
2009
12.

Lu Y., Hendra R., Oakley A.J., Keller P.A.
Tetrahedron Letters,
2014
13.

Layachi K., Ariès-Gautron I., Guerro M., Robert A.
Tetrahedron,
1992