Abstract
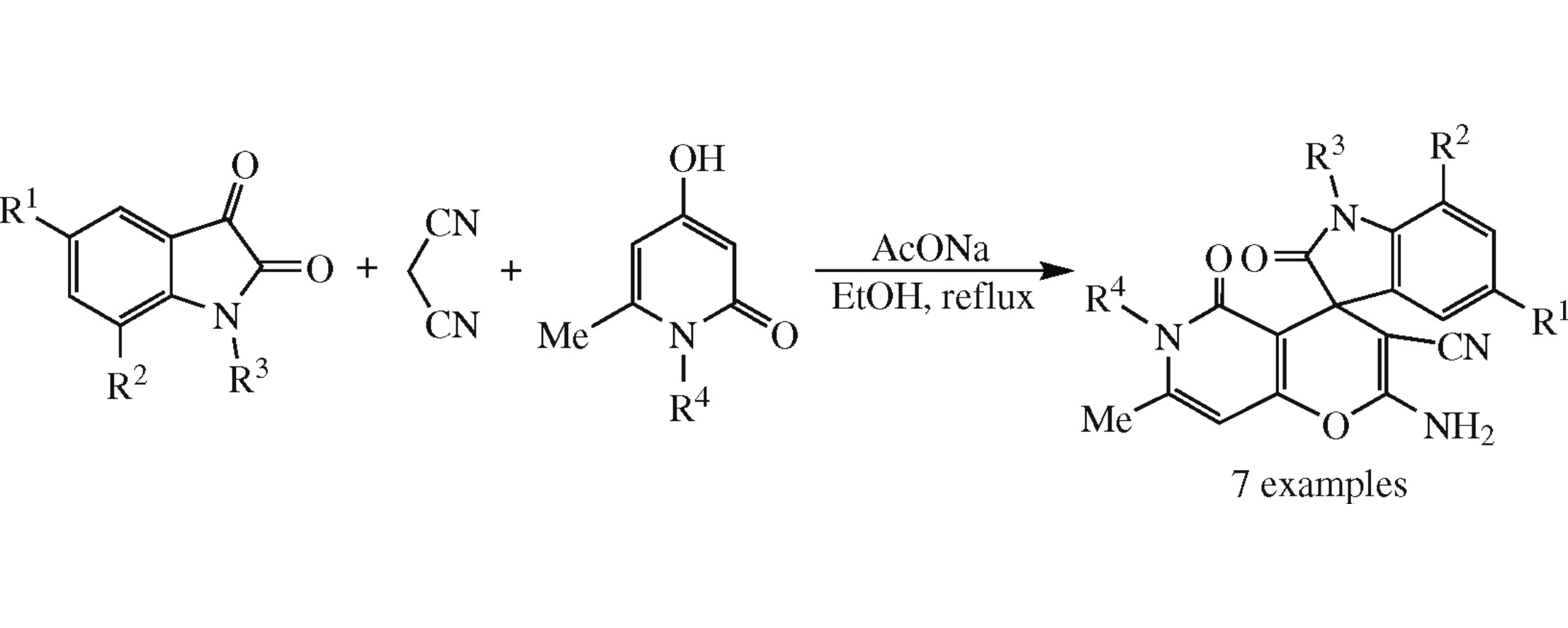
The new one-pot multicomponent assembling of isatins, malononitrile and 4 hydroxy-6-methylpyridin-2(1H)-ones into previously unknown 2’-amino-7’-methyl-2,5’-dioxo-5’,6’-dihydrospiro[indoline-3,4’-pyrano[3,2-c]pyridine]-3’-carbonitriles in 80–98% yields has been accomplished by their reflux in ethanol in the presence of sodium acetate.
References
1.

2.

Gu Y.
Green Chemistry,
2012
3.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
4.

Evans B.E., Rittle K.E., Bock M.G., DiPardo R.M., Freidinger R.M., Whitter W.L., Lundell G.F., Veber D.F., Anderson P.S., Chang R.S., Lotti V.J., Cerino D.J., Chen T.B., Kling P.J., Kunkel K.A., et. al.
Journal of Medicinal Chemistry,
1988
5.

Shanthi G., Perumal P.T.
Journal of Chemical Sciences,
2010
6.

Sun S.H., Zheng M., Ding K., Wang S., Sun Y.
Cancer Biology and Therapy,
2008
7.

Prasanna P., Balamurugan K., Perumal S., Yogeeswari P., Sriram D.
European Journal of Medicinal Chemistry,
2010
8.
![Regioselective synthesis of dispiro[1H-indene-2,3′-pyrrolidine-2′,3″-[3H]indole]-1,2″(1″H)-diones of potential anti-tumor properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Girgis A.S.
European Journal of Medicinal Chemistry,
2009
9.

Bhaskar G., Arun Y., Balachandran C., Saikumar C., Perumal P.T.
European Journal of Medicinal Chemistry,
2012
10.

Kornet M.J., Thio A.P.
Journal of Medicinal Chemistry,
1976
11.

Beecham A., Hart N., Johns S., Lamberton J.
Australian Journal of Chemistry,
1968
12.

Pandey R., Singh S.C., Gupta M.M.
Phytochemistry,
2006
13.

Heitzman Mary E, Neto Catherine C, Winiarz Elizabeth, Vaisberg Abraham J, Hammond Gerald B
Phytochemistry,
2005
14.

Chen I., Wu S., Tsai I., Wu T., Pezzuto J.M., Lu M.C., Chai H., Suh N., Teng C.
Journal of Natural Products,
1994
15.
![Structural Simplification of Bioactive Natural Products with Multicomponent Synthesis. 2. Antiproliferative and Antitubulin Activities of Pyrano[3,2-c]pyridones and Pyrano[3,2-c]quinolones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Magedov I.V., Manpadi M., Ogasawara M.A., Dhawan A.S., Rogelj S., Van slambrouck S., Steelant W.F., Evdokimov N.M., Uglinskii P.Y., Elias E.M., Knee E.J., Tongwa P., Antipin M.Y., Kornienko A.
Journal of Medicinal Chemistry,
2008
16.
![Electrochemically induced multicomponent assembling of isatins, 4-hydroxyquinolin-2(1H)-one and malononitrile: a convenient and efficient way to functionalized spirocyclic [indole-3,4′-pyrano[3,2-c]quinoline] scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Merkulova V.M., Ilovaisky A.I., Demchuk D.V., Belyakov P.A., Nikishin G.I.
Molecular Diversity,
2009
17.
![The electrocatalytic cascade assembling of isatins, malononitrile and N-alkyl barbiturates: An efficient multicomponent approach to the spiro[indole-3,5′-pyrano[2,3-d]pyrimidine] framework](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Demchuk D.V., Belyakov P.A., Ogibin Y.N., Nikishin G.I.
Electrochimica Acta,
2008
18.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Barba F., Batanero B.
Tetrahedron,
2013
19.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2012
20.

Elinson M.N., Merkulova V.M., Ilovaisky A.I., Barba F., Batanero B.
Electrochimica Acta,
2011
21.
Elinson M.N., Ryzhkov F.V., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2015
22.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Belyakov P.A., Barba F., Batanero B.
Tetrahedron,
2012
23.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
24.
![One-Pot ‘On-solvent’ Multicomponent Protocol for the Synthesis of Medicinally Relevant 4H-Pyrano[3,2-c]quinoline Scaffold](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Vereshchagin A.N., Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Bobrovsky S.I., Bushmarinov I.S., Egorov M.P.
Helvetica Chimica Acta,
2015
25.

Narayan S., Muldoon J., Finn M.G., Fokin V.V., Kolb H.C., Sharpless K.B.
Angewandte Chemie - International Edition,
2005
26.
Elinson M.N., Ryzhkov F.V., Nasybullin R.F., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2014
27.

Elinson M.N., Nasybullin R.F., Sokolova O.O., Zaimovskaya T.A., Egorov M.P.
Monatshefte fur Chemie,
2015
28.
10.1016/j.mencom.2016.09.011_bib0140
Rosen
1991
29.

Elinson M.N., Vereshchagin A.N., Feducovich S.K., Zaimovskaya T.A., Starikova Z.A., Belyakov P.A., Nikishin G.I.
Tetrahedron Letters,
2007
30.
![Electrocatalytic multicomponent assembling of isatins, 3-methyl-2-pyrazolin-5-ones and malononitrile: facile and convenient way to functionalized spirocyclic [indole-3,4′-pyrano[2,3-c]pyrazole] system](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Dorofeev A.S., Miloserdov F.M., Nikishin G.I.
Molecular Diversity,
2008
31.

Elinson M.N., Vereshchagin A.N., Nasybullin R.F., Bobrovsky S.I., Ilovaisky A.I., Merkulova V.M., Bushmarinov I.S., Egorov M.P.
RSC Advances,
2015
32.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960