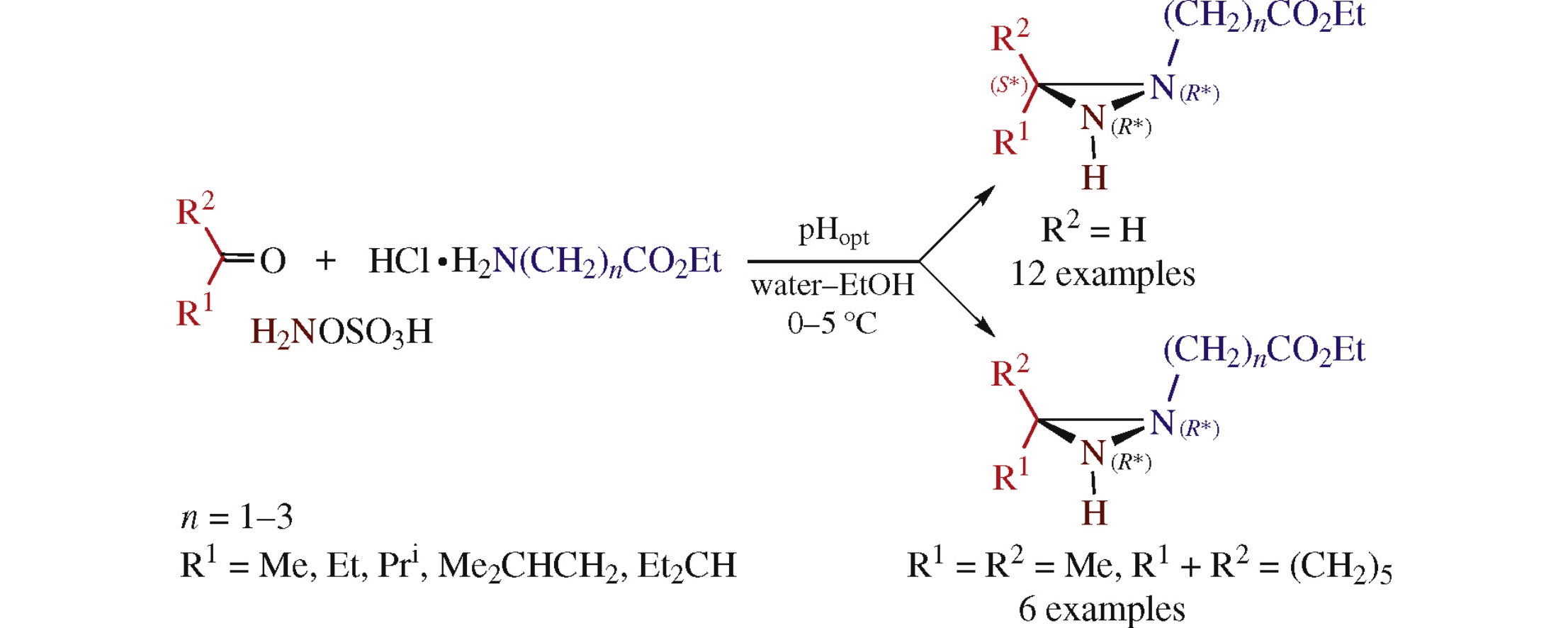
Abstract
The pH-controlled one-pot three-component condensation of carbonyl compounds, amino acid ethyl esters and hydroxylamine-O-sulfonic acid in water–ethanol mixture at 0–5 ̊C diastereoselectively affords conjugates of 1,3-di- or 1,3,3-trisubstituted diaziridines with neurotransmitter amino acids.
References
1.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
2.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
3.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
4.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
5.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
6.

Nortcliffe A., Botting N.P., O'Hagan D.
Organic and Biomolecular Chemistry,
2013
7.

Moody C.
Angewandte Chemie - International Edition,
2007
8.

Dondoni A., Massi A.
Accounts of Chemical Research,
2006
9.

Chakraborty T., Ghosh S., Jayaprakash S.
Current Medicinal Chemistry,
2002
10.

Steer D., Lew R., Perlmutter P., Smith A., Aguilar M.
Current Medicinal Chemistry,
2002
11.
10.1016/j.mencom.2016.09.008_bib0015
Mashkovskii
2000
12.
10.1016/j.mencom.2016.09.008_bib0020
Mashkovskii
2000
13.

Baichurina A.Z., Semina I.I., Garaev R.S.
Bulletin of Experimental Biology and Medicine,
1996
14.

Kostyanovskii R.G., Shustov G.V., Nabiev O.G., Denisenko S.N., Sukhanova S.A., Lavretskaya É.F.
Pharmaceutical Chemistry Journal,
1986
15.

Paget C.J., Davis C.S.
Journal of Medicinal Chemistry,
1964
16.

17.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2007
18.
![Synthesis and structure of 1-[ω-(3,3-dialkyldiaziridin-1-yl)alkyl]-3, 3-dialkyldiaziridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Petukhova V.Y., Strelenko Y.A., Lyssenko K.A., Makhova N.N.
Russian Chemical Bulletin,
2007
19.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2006
20.

Prokopov A.A., Kotlova L.I., Berlyand A.S.
Pharmaceutical Chemistry Journal,
2005
21.
Makhova N.N., Petukhova V.Y., Kuznetsov V.V.
Arkivoc,
2008
22.

Kuznetsov V.V., Syroeshkina J.S., Moskvin D.I., Struchkova M.I., Makhova N.N., Zharov A.A.
Journal of Heterocyclic Chemistry,
2008
23.

Syroeshkina Y.S., Kuznetsov V.V., Kachala V.V., Makhova N.N.
Journal of Heterocyclic Chemistry,
2009
24.

Zlotin S.G., Makhova N.N.
Russian Chemical Reviews,
2010
25.

Makhova N.N., Shevtsov A.V., Petukhova V.Y.
Russian Chemical Reviews,
2011
26.

Petukhova V.Y., Fershtat L.L., Kachala V.V., Kuznetsov V.V., Khakimov D.V., Pivina T.S., Makhova N.N.
Journal of Heterocyclic Chemistry,
2013
27.
![Conformational and Bonding Properties of 3,3-Dimethyl- and 6,6-Dimethyl-1,5-diazabicyclo[3.1.0]hexane: A Case Study Employing the Monte Carlo Method in Gas Electron Diffraction](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vishnevskiy Y.V., Schwabedissen J., Rykov A.N., Kuznetsov V.V., Makhova N.N.
Journal of Physical Chemistry A,
2015
28.
![CAN-mediated new, regioselective one-pot access to bicyclic cationic structures with 2,3-dihydro-1H-pyrazolo[1,2-a]pyrazol-4-ium core](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Pleshchev M.I., Das Gupta N.V., Kuznetsov V.V., Fedyanin I.V., Kachala V.V., Makhova N.N.
Tetrahedron,
2015
29.
10.1016/j.mencom.2016.09.008_sbref0040a
Schmitz
Dreiringe mit zwei Heteroatom: Oxaziridine, Diaziridine, cyclische Diazoverbindungen,
1967
30.
10.1016/j.mencom.2016.09.008_sbref0040b
Heine
1983
31.
10.1016/j.mencom.2016.09.008_sbref0040c
Schmitz
1984
32.
10.1016/j.mencom.2016.09.008_sbref0040d
Kostyanovsky
1996
33.
10.1016/j.mencom.2016.09.008_sbref0040e
McAllister
2008
34.

Kuznetsov V.V., Makhova N.N., Strelenko Y.A., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1991
35.

Kuznetsov V.V., Makhova N.N., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1997
36.

Trapp O., Schurig V., Kostyanovsky R.G.
Chemistry - A European Journal,
2004
37.

Jung M., Schurig V.
Journal of the American Chemical Society,
1992
38.
10.1016/j.mencom.2016.09.008_sbref0050c
Lehn
Top. Curr. Chem.,
1970
39.

Mannschreck A., Radeglia R., Gründemann E., Ohme R.
European Journal of Inorganic Chemistry,
1967