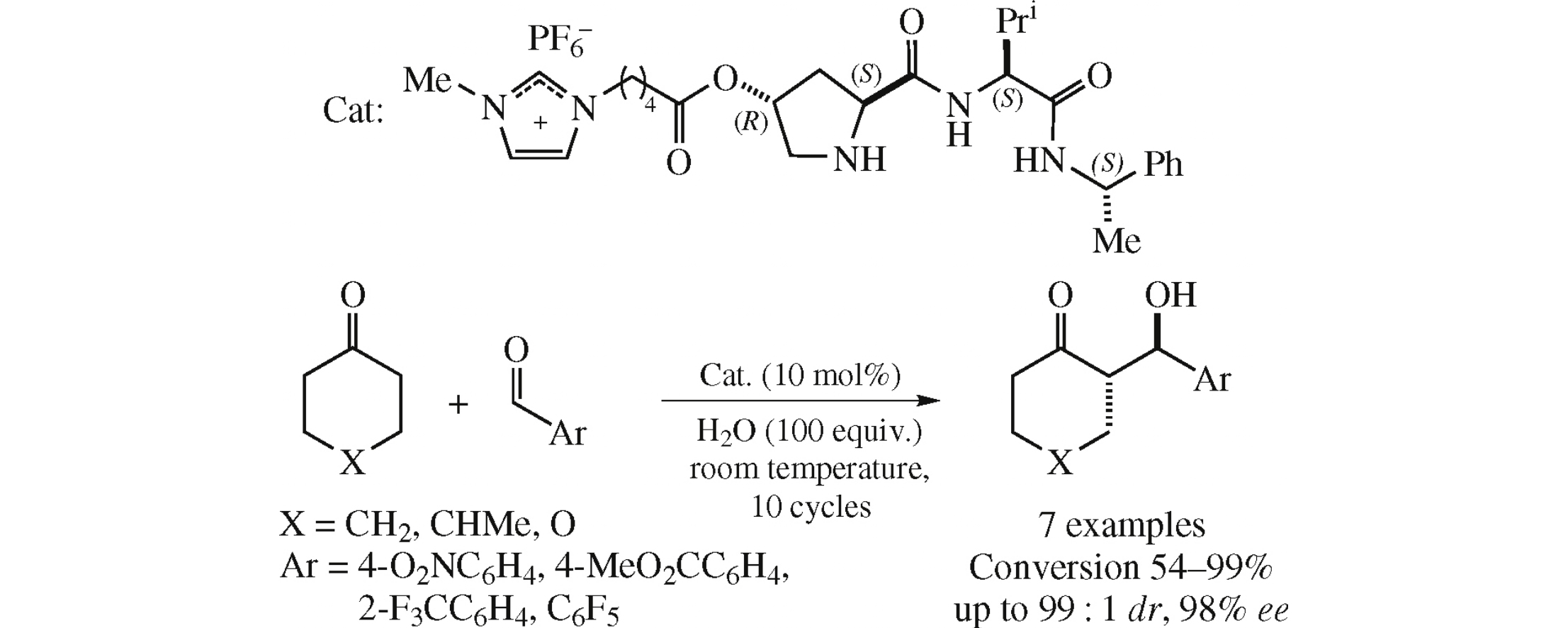
Abstract
Novel (4R)-HO-(2S)-Pro-(S)-Val derivative tagged to 1-methylimidazolium cation and PF6- anion efficiently catalyzes asymmetric aldol reactions of cyclic ketones with aromatic aldehydes in aqueous environment to afford the corresponding aldols in high yield with moderate to high anti-diastereo- (dr up to 99:1) and enantioselectivity (up to 98% ee). The catalyst was recycled 9 times in the same reaction without any decrease in selectivity values.
References
1.

Davie E.A., Mennen S.M., Xu Y., Miller S.J.
Chemical Reviews,
2007
2.

3.

Lewandowski B., Wennemers H.
Current Opinion in Chemical Biology,
2014
4.

Liu X., Lin L., Feng X.
Chemical Communications,
2009
5.

Chen X., Yu J., Gong L.
Chemical Communications,
2010
6.
10.1016/j.mencom.2016.09.007_bib0030
Yang
Synlett,
2010
7.
10.1016/j.mencom.2016.09.007_bib0035
Lu
Synlett,
2012
8.

Berdugo C., Escuder B., Miravet J.F.
Organic and Biomolecular Chemistry,
2015
9.

Wiesner M., Revell J., Wennemers H.
Angewandte Chemie - International Edition,
2008
10.

Rodríguez-Llansola F., Miravet J.F., Escuder B.
Chemical Communications,
2009
11.

Machuca E., Rojas Y., Juaristi E.
Asian Journal of Organic Chemistry,
2014
12.

Szőllősi G., Csámpai A., Somlai C., Fekete M., Bartók M.
Journal of Molecular Catalysis A Chemical,
2014
13.

Murtinho D., Serra M.
Current Organocatalysis,
2014
14.

Chandrasekhar S., Kumar C.P., Kumar T.P., Haribabu K., Jagadeesh B., Lakshmi J.K., Mainkar P.S.
RSC Advances,
2014
15.

Psarra A., Kokotos C.G., Moutevelis-Minakakis P.
Tetrahedron,
2014
16.

Hofmann C., Schuler S.M., Wende R.C., Schreiner P.R.
Chemical Communications,
2014
17.

Akiyama M., Akagawa K., Seino H., Kudo K.
Chemical Communications,
2014
18.

Ötvös S.B., Mándity I.M., Fülöp F.
ChemSusChem,
2012
19.

Carpenter R., Fettinger J., Lam K., Kurth M.
Angewandte Chemie - International Edition,
2008
20.

Arakawa Y., Wiesner M., Wennemers H.
Advanced Synthesis and Catalysis,
2011
21.

Yan J., Wang L.
Chirality,
2009
22.

Scatena G.S., de la Torre A.F., Cass Q.B., Rivera D.G., Paixão M.W.
ChemCatChem,
2014
23.

Brunelli N.A., Jones C.W.
Journal of Catalysis,
2013
24.

Akagawa K., Sakai N., Kudo K.
Angewandte Chemie - International Edition,
2014
25.

Ni B., Headley A.
Chemistry - A European Journal,
2010
26.

Prechtl M., Scholten J., Neto B., Dupont J.
Current Organic Chemistry,
2009
27.

Kucherenko A.S., Siyutkin D.E., Maltsev O.V., Kochetkov S.V., Zlotin S.G.
Russian Chemical Bulletin,
2012
28.

Lombardo M., Pasi F., Easwar S., Trombini C.
Advanced Synthesis and Catalysis,
2007
29.

Lombardo M., Easwar S., Pasi F., Trombini C.
Advanced Synthesis and Catalysis,
2009
30.

Larionova N.A., Kucherenko A.S., Siyutkin D.E., Zlotin S.G.
Tetrahedron,
2011
31.

Maltsev O.V., Chizhov A.O., Zlotin S.G.
Chemistry - A European Journal,
2011
32.

Kucherenko A.S., Siyutkin D.E., Nigmatov A.G., Chizhov A.O., Zlotin S.G.
Advanced Synthesis and Catalysis,
2012
33.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
34.

Gruttadauria M., Giacalone F., Noto R.
Advanced Synthesis and Catalysis,
2009
35.

Raj M., Singh V.K.
Chemical Communications,
2009
36.

Mlynarski J., Baś S.
Chemical Society Reviews,
2014
37.

Bhowmick S., Mondal A., Ghosh A., Bhowmick K.C.
Tetrahedron Asymmetry,
2015
38.

Trombini C., Lombardo M., Pasi F., Easwar S.
Synlett,
2008
39.

Lombardo M., Easwar S., De Marco A., Pasi F., Trombini C.
Organic and Biomolecular Chemistry,
2008
40.

Siyutkin D.E., Kucherenko A.S., Struchkova M.I., Zlotin S.G.
Tetrahedron Letters,
2008
41.

Kucherenko A.S., Syutkin D.E., Zlotin S.G.
Russian Chemical Bulletin,
2008
42.
![(1R,2R)‐Bis[(S)‐prolinamido]cyclohexane Modified with Ionic Groups: The First C2‐Symmetric Immobilized Organocatalyst for Asymmetric Aldol Reactions in Aqueous Media](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kochetkov S.V., Kucherenko A.S., Zlotin S.G.
European Journal of Organic Chemistry,
2011
43.
Kochetkov S.V., Kucherenko A.S., Zlotin S.G.
Mendeleev Communications,
2015
44.

Machajewski T.D., Wong C.
Angewandte Chemie - International Edition,
2000
45.
10.1016/j.mencom.2016.09.007_sbref0135b
Fessner
in Asymmetric Organic Synthesis with Enzymes,
2008
46.
10.1016/j.mencom.2016.09.007_bib0140
Dodda
Synlett,
2007
47.
10.1016/j.mencom.2016.09.007_sbref0145a
Modern Methods in Stereoselective Aldol Reactions,
2013
48.

Mukherjee S., Yang J.W., Hoffmann S., List B.
Chemical Reviews,
2007
49.

Zlotin S.G., Kucherenko A.S., Beletskaya I.P.
Russian Chemical Reviews,
2009
50.

Trost B.M., Brindle C.S.
Chemical Society Reviews,
2010
51.

Scheffler U., Mahrwald R.
Chemistry - A European Journal,
2013