Abstract
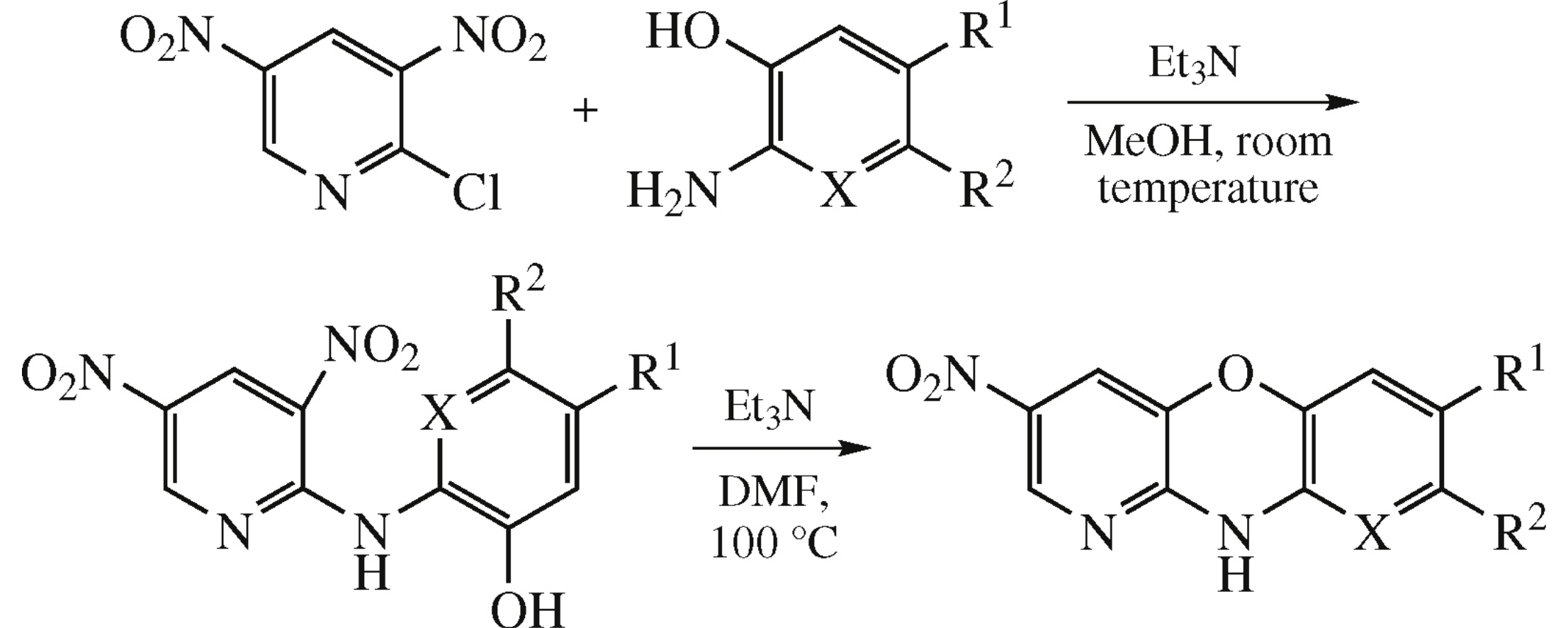
3-Nitro-10H-pyrido[3,2-b][1,4]benzoxazines and first representative of a new heterocyclic system, 10H-dipyrido[2,3-e:3’,2’-b][1,4]oxazine, were obtained by reaction of 2-chloro-3,5-dinitropyridine with various o-aminophenols followed by intramolecular substitution of the nitro group.
References
1.
10.1016/j.mencom.2016.09.005_sbref0005a
Terrier
Nucleophilic Aromatic Displacement. The Influence of the Nitro Group,
1991
2.
10.1016/j.mencom.2016.09.005_sbref0005b
Terrier
Modern Nucleophilic Aromatic Substitution,
2013
3.
10.1016/j.mencom.2016.09.005_sbref0010a
Chupakhin
Nucleophilic Aromatic Substitution of Hydrogen,
1994
4.

Charushin V.N., Chupakhin O.N.
Topics in Heterocyclic Chemistry,
2013
5.
10.1016/j.mencom.2016.09.005_sbref0015a
Bastrakov
ARKIVOC,
2009
6.
![Pericyclic [4+2] and [3+2] Cycloaddition Reactions of Nitroarenes in Heterocyclic Synthesis](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shevelev S.A., Starosotnikov A.M.
Chemistry of Heterocyclic Compounds,
2013
11.
10.1016/j.mencom.2016.09.005_sbref0025b
Mashkovskii
Lekarstvennye sredstva (Drugs),
2006
15.
19.
Z. Tu, R. Mach, L. Yu and P. Kotzbauer, Patent US 20130315825 A1, 2013.
20.
A. Sun, T. Ganesh, J. Min, P. Thepchatri, Y. Du, J.P. Snyder and D. C. Liotta, Patent US WO2010042489 A2, 2010.
21.
10.1016/j.mencom.2016.09.005_bib0055
Truce
1970
22.

Gerasimova T.N., Kolchina E.F.
Russian Chemical Reviews,
1995
24.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
25.
10.1016/j.mencom.2016.09.005_sbref0075a
Mąkosza
2014
26.
10.1016/j.mencom.2016.09.005_sbref0075b
Shevelev
2014