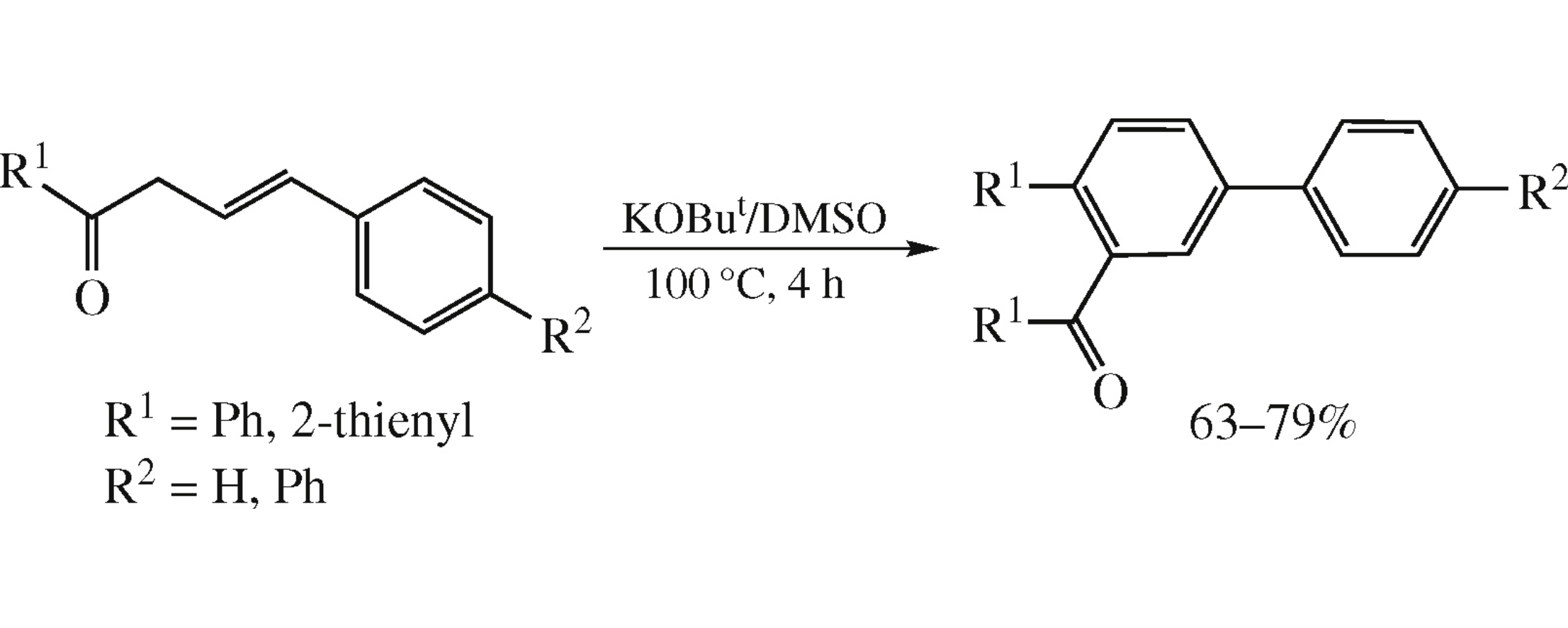
Abstract
4-Aryl-1-(het)arylbut-3-en-1-ones dimerize smoothly in the ButOK/DMSO superbase system at 100°C for 4 h to afford acylated terphenyls in up to 79% yield. The dimerization is accompanied by elimination of the methylarene molecule.
References
1.

Trofimov B., Schmidt E., Ushakov I., Zorina N., Skital'tseva E., Protsuk N., Mikhaleva A.
Chemistry - A European Journal,
2010
2.

Trofimov B.A., Schmidt E.Y., Zorina N.V., Ivanova E.V., Ushakov I.A., Mikhaleva A.I.
Advanced Synthesis and Catalysis,
2012
3.

Trofimov B.A., Schmidt E.Y., Zorina N.V., Ivanova E.V., Ushakov I.A.
Journal of Organic Chemistry,
2012
4.
Schmidt E.Y., Zorina N.V., Tarasova O.A., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2013
5.

Undeela S., Ramchandra J.P., Menon R.S.
Tetrahedron Letters,
2014
6.

Schmidt E.Y., Tatarinova I.V., Ivanova E.V., Zorina N.V., Ushakov I.A., Trofimov B.A.
Organic Letters,
2012
10.
Schmidt E.Y., Zorina N.V., Ivanova E.V., Tatarinova I.V., Ushakov I.A., Mikhaleva A.I., Trofimov B.A.
Mendeleev Communications,
2013
11.
12.
![Base-Promoted Formal [4 + 3] Annulation between 2-Fluorophenylacetylenes and Ketones: A Route to Benzoxepines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ouyang L., Qi C., He H., Peng Y., Xiong W., Ren Y., Jiang H.
Journal of Organic Chemistry,
2016
14.
15.
10.1016/j.mencom.2016.09.003_sbref0040c
Electronic Materials: The Oligomer Approach,
1998