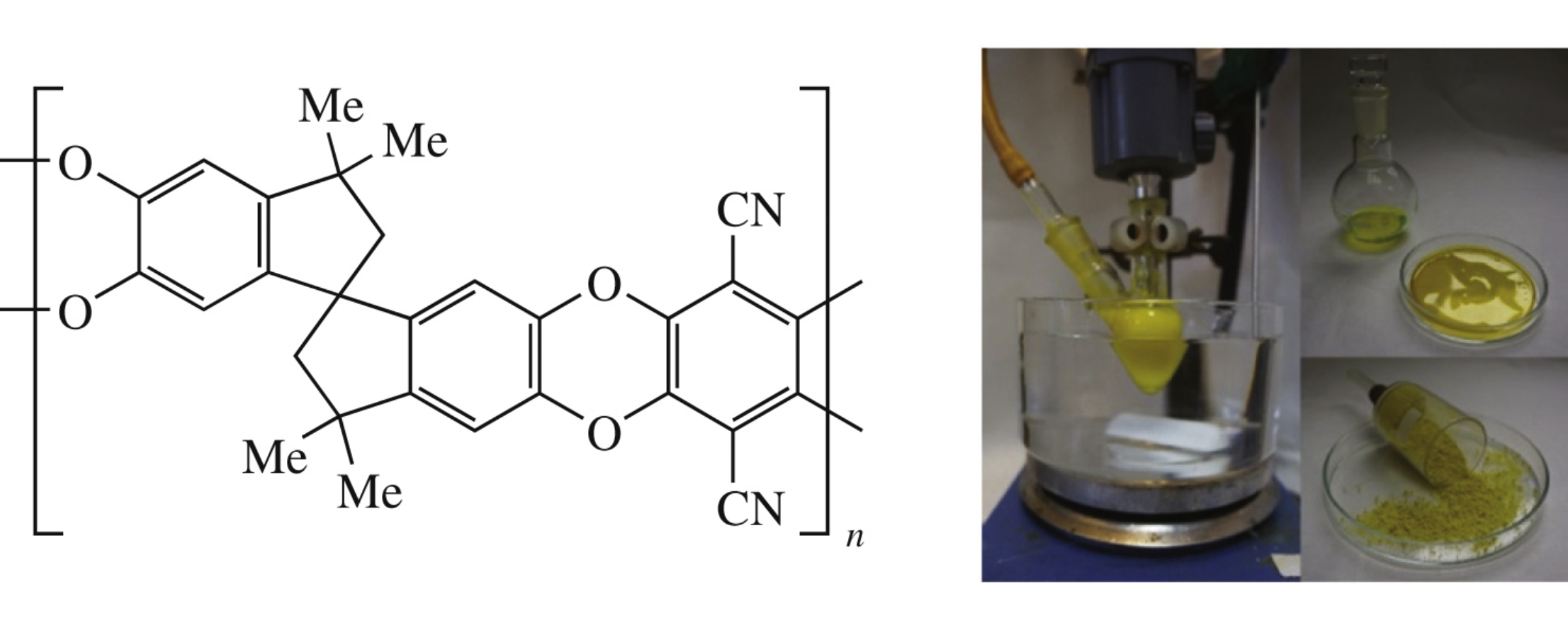
Abstract
A new method for the effective synthesis of high molecular weight polymer of intrinsic microporosity (PIM-1) powder widely employed for gas separation membranes was successfully developed using precipitative polyheterocyclization process in non-toxic DMSO.
References
1.

Budd P. ., Elabas E. ., Ghanem B. ., Makhseed S., McKeown N. ., Msayib K. ., Tattershall C. ., Wang D.
Advanced Materials,
2004
2.

Zhang J., Jin J., Cooney R., Zhang S.
Polymer,
2015
3.

Budd P.M., Ghanem B.S., Makhseed S., McKeown N.B., Msayib K.J., Tattershall C.E.
Chemical Communications,
2004
4.
Kurmaz S.V., Grubenko G.A., Knerelman E.I., Davydova G.I., Torbov V.I., Dremova N.N.
Mendeleev Communications,
2014
5.

6.
Yaroslavtsev A.B., Yampolskii Y.P.
Mendeleev Communications,
2014
7.

Du N., Song J., Robertson G.P., Pinnau I., Guiver M.D.
Macromolecular Rapid Communications,
2008
8.

Song J., Du N., Dai Y., Robertson G.P., Guiver M.D., Thomas S., Pinnau I.
Macromolecules,
2008
9.

Constable D.J., Jimenez-Gonzalez C., Henderson R.K.
Organic Process Research and Development,
2006
10.
10.1016/j.mencom.2016.07.033_bib0035
Ponomarev
1991
11.
10.1016/j.mencom.2016.07.033_bib0040
Ponomarev
Vysokomol. Soedin. A,
1992
12.

Jacobson H., Stockmayer W.H.
Journal of Chemical Physics,
1950
13.
10.1016/j.mencom.2016.07.033_bib0050
Flory
Principles of Polymer Chemistry,
1953
14.
10.1016/j.mencom.2016.07.033_bib0055
Blagodatskikh
e-Polymers,
2005
15.

Kricheldorf H.R., Lomadze N., Fritsch D., Schwarz G.
Journal of Polymer Science, Part A: Polymer Chemistry,
2006