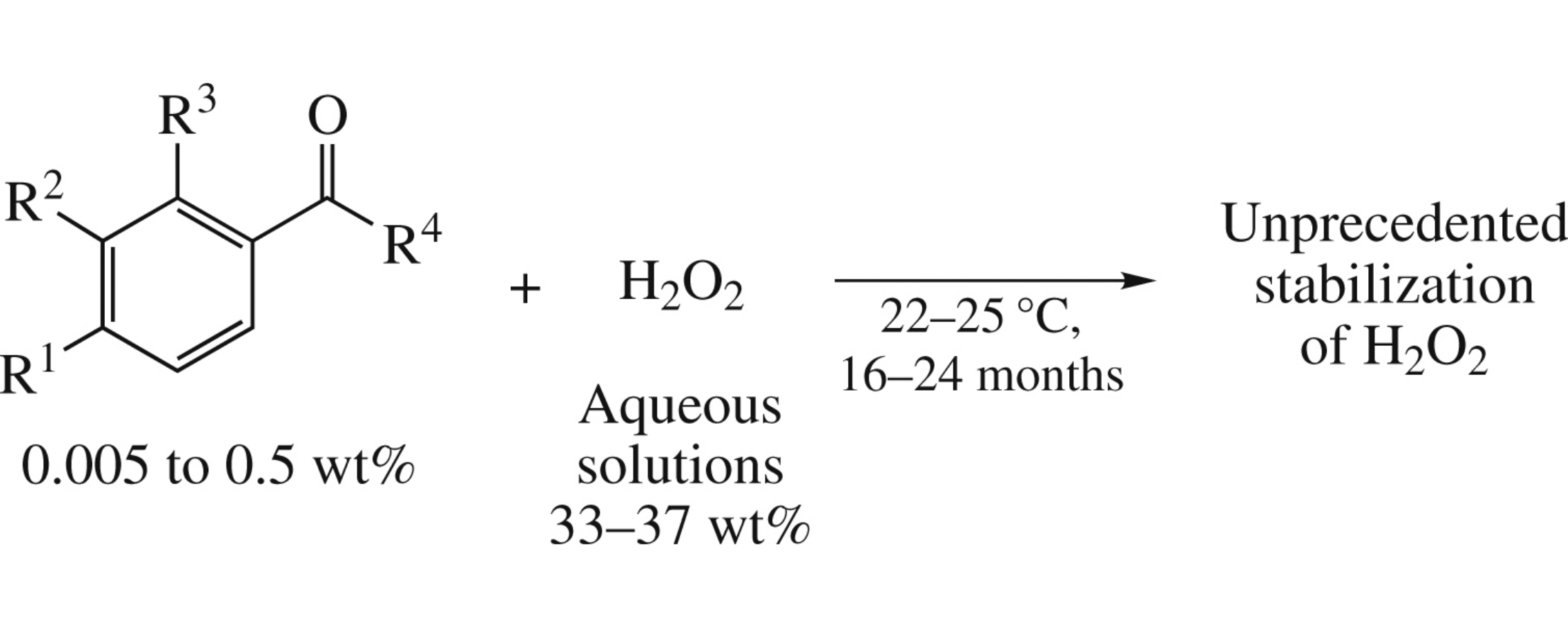
Abstract
Aryl alkyl ketones with substituents in the aromatic ring taken in an amount from 0.005 to 0.5% efficiently stabilize hydrogen peroxide in an aqueous solution during storage at 22–25°C for 16–24 months.
References
1.

Campos-Martin J.M., Blanco-Brieva G., Fierro J.L.
Angewandte Chemie - International Edition,
2006
2.

Samanta C.
Applied Catalysis A: General,
2008
3.

Dittmeyer R., Grunwaldt J.-., Pashkova A.
Catalysis Today,
2015
4.
10.1016/j.mencom.2016.07.021_sbref0005d
Jones
Applications of Hydrogen Peroxide and Derivatives,
1999
5.
10.1016/j.mencom.2016.07.021_sbref0010a
Goor
Catalytic Oxidations with Hydrogen Peroxide as Oxidant,
1992
6.
10.1016/j.mencom.2016.07.021_sbref0010b
Goor
2000
7.

Sheriff T.
Journal of Chemical Technology and Biotechnology,
2000
8.

Hug S.J., Leupin O.
Environmental Science & Technology,
2003
9.

Kwan W.P., Voelker B.M.
Environmental Science & Technology,
2002
10.

Kwan W.P., Voelker B.M.
Environmental Science & Technology,
2003
11.

Petigara B.R., Blough N.V., Mignerey A.C.
Environmental Science & Technology,
2002
12.

Lin S., Gurol M.D.
Environmental Science & Technology,
1998
13.

Watts R.J., Foget M.K., Kong S., Teel A.L.
Journal of Hazardous Materials,
1999
14.

Paciolla M.D., Davies G., Jansen S.A.
Environmental Science & Technology,
1999
15.
A. I. Dalton, Jr. and J. V. Bauer, US Patent 4320102 A, 1982.
16.
P. C. Wegner, US Patent 20030151024 A1, 2003.
17.
10.1016/j.mencom.2016.07.021_bib0025
Ippolitov
Russ. J. Inorg. Chem.,
2001
18.
D. J. Bonislawski and D. C. Lovetro, US Patent 20100261636 A1, 2010.
19.
J. C. Watts, US Patent 4070442 A, 1978.
20.
M. M. (McKay) Rauhut and A. M. Semsel, US Patent 4064064 A, 1977.
21.
M. Colvin, US Patent 6803354 B2, 2004.
22.
V. M. Zacska, C.L. Orr, D.L. Prater and J. Lockridge, US Patent 8357356 B2, 2013.
23.
D. A. Shiraeff, US Patent 3376110 A, 1968.
24.
P. Klug, M.F. Pilz and U. Back, US Patent 20130236383 A1, 2013.
25.
M. W. Mcmillen, Patent EP0351772A3, 1990.
26.
H. Müller and Y. Lissner, Patent WO2008077688 A1, 2008.
27.

Saravanakumar K., Baskaran R., Kubendran T.R.
E-Journal of Chemistry,
2012
28.
10.1016/j.mencom.2016.07.021_bib0075
Fenaroli's Handbook of Flavor Ingredients,
2009
29.

Terent'ev A.O., Platonov M.M., Sonneveld E.J., Peschar R., Chernyshev V.V., Starikova Z.A., Nikishin G.I.
Journal of Organic Chemistry,
2007
30.

Terent'ev A.O., Platonov M.M., Ogibin Y.N., Nikishin G.I.
Synthetic Communications,
2007
31.

Terent'ev A.O., Platonov M.M., Kashin A.S., Nikishin G.I.
Tetrahedron,
2008
32.
![Synthesis and antimalarial activity of sixteen dispiro-1,2,4, 5-tetraoxanes: alkyl-substituted 7,8,15,16-tetraoxadispiro[5.2.5. 2]hexadecanes.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vennerstrom J.L., Dong Y., Andersen S.L., Ager, A.L., Fu H., Miller R.E., Wesche D.L., Kyle D.E., Gerena L., Walters S.M., Wood J.K., Edwards G., Holme A.D., McLean W.G., et. al.
Journal of Medicinal Chemistry,
2000
33.

McCullough K.J., Wood J.K., Bhattacharjee A.K., Dong Y., Kyle D.E., Milhous W.K., Vennerstrom J.L.
Journal of Medicinal Chemistry,
2000
34.
10.1016/j.mencom.2016.07.021_sbref0095a
Sanderson
Synthesis,
1975
35.

SANDERSON J.R., ZEILER A.G.
Synthesis,
1975
36.

Terent’ev A.O., Yaremenko I.A., Chernyshev V.V., Dembitsky V.M., Nikishin G.I.
Journal of Organic Chemistry,
2012
37.

Ogibin Y.N., Terent"ev A.O., Ananikov V.P., Nikishin G.I.
Russian Chemical Bulletin,
2001
38.

Ogibin Y.N., Terent'ev A.O., Kutkin A.V., Nikishin G.I.
Tetrahedron Letters,
2002