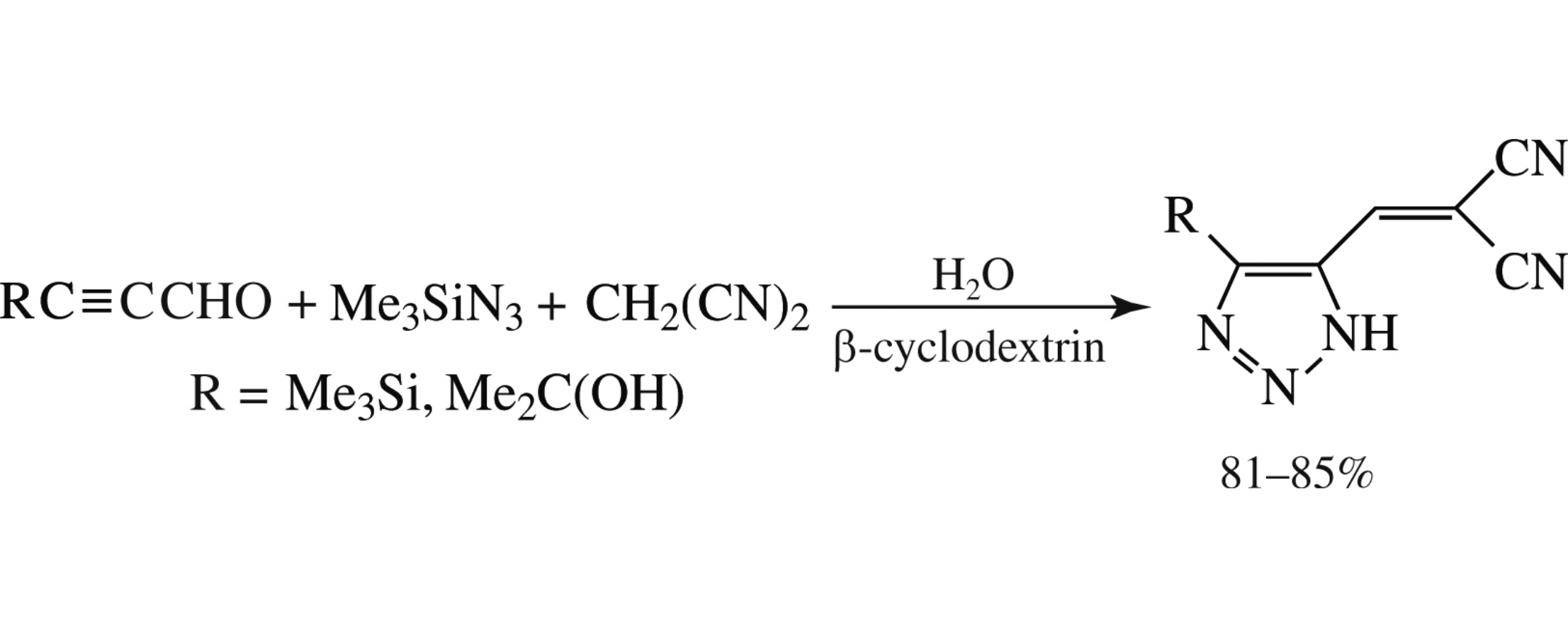Abstract
An efficient green method for the selective synthesis of new 4,5-disubstituted 1H-1,2,3-triazoloalkylidenes via the β-cyclodextrin-catalyzed three-component reaction between substituted propynals, trimethylsilyl azide and malononitrile in water at room temperature has been developed.
References
1.

Agalave S.G., Maujan S.R., Pore V.S.
Chemistry - An Asian Journal,
2011
2.

Tron G.C., Pirali T., Billington R.A., Canonico P.L., Sorba G., Genazzani A.A.
Medicinal Research Reviews,
2008
3.

Krivopalov V.P., Shkurko O.P.
Russian Chemical Reviews,
2005
4.
10.1016/j.mencom.2016.07.020_bib0010
Bakulev
2015
5.
![Bipyridyl-substituted benzo[1,2,3]triazoles as a thermally stable electron transporting material for organic light-emitting devices](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ichikawa M., Mochizuki S., Jeon H., Hayashi S., Yokoyama N., Taniguchi Y.
Journal of Materials Chemistry A,
2011
6.

7.

Gimeno N., Martín-Rapún R., Rodríguez-Conde S., Serrano J.L., Folcia C.L., Pericás M.A., Ros M.B.
Journal of Materials Chemistry A,
2012
8.
Fershtat L.L., Ashirbaev S.S., Kulikov A.S., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2015
9.
Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Nosova E.V., Kovalev I.S., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2015
10.

Rostovtsev V.V., Green L.G., Fokin V.V., Sharpless K.B.
Angewandte Chemie - International Edition,
2002
11.
![Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tornøe C.W., Christensen C., Meldal M.
Journal of Organic Chemistry,
2002
12.

Meldal M., Tornøe C.W.
Chemical Reviews,
2008
13.
![Tricks with clicks: modification of peptidomimetic oligomers via copper-catalyzed azide-alkyne [3 + 2] cycloaddition](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Holub J.M., Kirshenbaum K.
Chemical Society Reviews,
2010
14.

Synthesis and investigation of tuberculosis inhibition activities of some 1,2,3-triazole derivatives
Dabak K., Sezer Ö., Akar A., Anaç O.
European Journal of Medicinal Chemistry,
2003
15.

16.

Komeda S., Lutz M., Spek A.L., Yamanaka Y., Sato T., Chikuma M., Reedijk J.
Journal of the American Chemical Society,
2002
17.

Harrison T., Owens A.P., Williams B.J., Swain C.J., Williams A., Carlson E.J., Rycroft W., Tattersall F.D., Cascieri M.A., Chicchi G.G., Sadowski S., Rupniak N.M., Hargreaves R.J.
Journal of Medicinal Chemistry,
2001
18.

Weide T., Saldanha S.A., Minond D., Spicer T.P., Fotsing J.R., Spaargaren M., Frère J., Bebrone C., Sharpless K.B., Hodder P.S., Fokin V.V.
ACS Medicinal Chemistry Letters,
2010
19.

Aromí G., Barrios L.A., Roubeau O., Gamez P.
Coordination Chemistry Reviews,
2011
20.

Duan H., Sengupta S., Petersen J.L., Akhmedov N.G., Shi X.
Journal of the American Chemical Society,
2009
21.

Schulze B., Schubert U.S.
Chemical Society Reviews,
2014
22.

Jewett J.C., Bertozzi C.R.
Chemical Society Reviews,
2010
23.

Gaetke L.
Toxicology,
2003
24.

Demina M.M., Nguyen T.L., Shaglaeva N.S., Mareev A.V., Medvedeva A.S.
Russian Journal of Organic Chemistry,
2012
25.

Medvedeva A.S., Demina M.M., Nguyen T.L., Vu T.D., Bulanov D.A., Novokshonov V.V.
Russian Journal of Organic Chemistry,
2013
26.

Demina M.M., Novopashin P.S., Sarapulova G.I., Larina L.I., Smolin A.S., Fundamenskii V.S., Kashaev A.A., Medvedeva A.S.
Russian Journal of Organic Chemistry,
2004
27.
10.1016/j.mencom.2016.07.020_bib0060
Novokshonova
Synthesis,
2008
28.

Medvedeva A.S., Mareev A.V., Demina M.M.
Russian Chemical Bulletin,
2008
29.

Shagun V.A., Medvedeva A.S., Mareev A.V.
Tetrahedron,
2013
30.

Novokshonov V.V., Novokshonova I.A., Nguyen H.T., Medvedeva A.S.
Synthetic Communications,
2012
31.

Medvedeva A.S., Demina M.M., Konkova T.V., Vu T.D., Larina L.I.
Chemistry of Heterocyclic Compounds,
2014
32.

Borate H.B., Kudale A.S., Agalave S.G.
Organic Preparations and Procedures International,
2012
33.

Huang J., Zhou J., Song S., Song H., Chen Z., Yi W.
Tetrahedron,
2015
34.

Zonouz A.M., Moghani D.
Synthetic Communications,
2011
35.

Yang J., Li J., Hao P., Qiu F., Liu M., Zhang Q., Shi D.
Dyes and Pigments,
2015
36.

37.

Zhao W., Zhong Q.
Journal of Inclusion Phenomena and Macrocyclic Chemistry,
2011
38.

Medvedeva A.S., Mitroshina I.V., Afonin A.V., Demina M.M., Pavlov D.V., Mareev A.V.
Russian Journal of Organic Chemistry,
2010
39.
Shankar J., Satish G., Kumar B.S., Nageswar Y.V.
European Journal of Chemistry,
2014
40.

Ramesh K., Murthy S.N., Nageswar Y.V.
Tetrahedron Letters,
2011
41.

Shin J., Lim Y., Lee K.
Journal of Organic Chemistry,
2012
42.

Bulanov D.A., Novokshonova I.A., Safronova L.P., Ushakov I.A., Medvedeva A.S.
Tetrahedron Letters,
2016
43.

Jarowski P.D., Wu Y., Schweizer W.B., Diederich F.
Organic Letters,
2008
44.
Mareev A.V., Medvedeva A.S., Khatashkeev A.V., Afonin A.V.
Mendeleev Communications,
2005