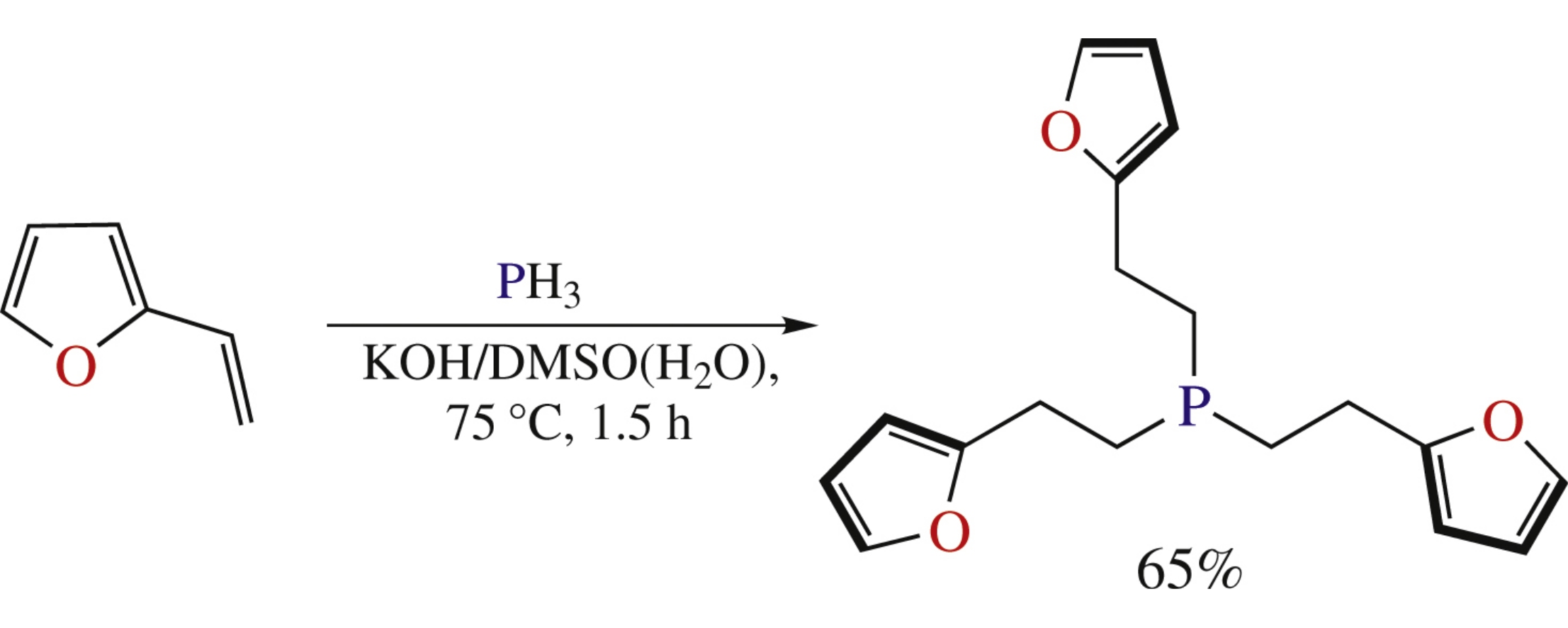
Abstract
Nucleophilic addition of PH3 to 2-vinylfuran in the KOH/DMSO(H2O) suspension affords 65% of tris[2-(2-furyl)ethyl]-phosphine, whose treatment with H2O2, Se or PdCl gives the corresponding phosphine oxide, selenide and complex of trans-[Pd(L)2Cl2] type in good to excellent yields.
References
1.

Trofimov B.A., Arbuzova S.N., Gusarova N.K.
Russian Chemical Reviews,
1999
2.

Baillie C., Xiao J.
Current Organic Chemistry,
2003
3.

Delacroix O., Gaumont A.
Current Organic Chemistry,
2005
4.
10.1016/j.mencom.2016.07.016_sbref0010a
Quin
A Guide to Organophosphorus Chemistry,
2000
5.
10.1016/j.mencom.2016.07.016_sbref0010b
Organophosphorus Reagents,
2004
6.
Trofimov B.A., Gusarova N.K.
Mendeleev Communications,
2009
7.

Gusarova N.K., Arbuzova S.N., Trofimov B.A.
Pure and Applied Chemistry,
2012
8.
Gusarova N.K., Malysheva S.F., Kuimov V.A., Belogorlova N.A., Mikhailenko V.L., Trofimov B.A.
Mendeleev Communications,
2008
9.

Malysheva S.F., Gusarova N.K., Artem'ev A.V., Belogorlova N.A., Smirnov V.I., Shagun V.A., Kuimov V.A., Trofimov B.A.
Synthetic Communications,
2012
10.

Artem’ev A.V., Malysheva S.F., Gusarova N.K., Korocheva A.O., Timokhina L.V., Trofimov B.A.
Russian Chemical Bulletin,
2013
11.
10.1016/j.mencom.2016.07.016_sbref0035a
Gusarova
Russ. J. Gen. Chem.,
1997
12.
10.1016/j.mencom.2016.07.016_sbref0035b
Trofimov
Russ. J. Gen. Chem.,
2000
13.

Malysheva S.F., Artem’ev A.V., Gusarova N.K., Timokhin B.V., Tatarinova A.A., Trofimov B.A.
Russian Journal of General Chemistry,
2009
14.

Trofimov B.A., Brandsma L., Arbuzova S.N., Malysheva S.F., Gusarova N.K.
Tetrahedron Letters,
1994
15.

Brandsma L., Arbuzova S., Lang R.D., Gusarova N., Trofimov B.
Phosphorus, Sulfur and Silicon and the Related Elements,
1997
16.

Nawara A.J., Shima T., Hampel F., Gladysz J.A.
Journal of the American Chemical Society,
2006
17.
Artem’ev A.V., Gusarova N.K., Malysheva S.F., Belogorlova N.A., Kazheva O.N., Alexandrov G.G., Dyachenko O.A., Trofimov B.A.
Mendeleev Communications,
2015
18.

Huang X., Peng B., Luparia M., Gomes L.F., Veiros L.F., Maulide N.
Angewandte Chemie - International Edition,
2012
19.

Cui Z., Li Y., Ling Y., Huang J., Cui J., Wang R., Yang X.
European Journal of Medicinal Chemistry,
2010
20.

21.

Mortensen D.S., Rodriguez A.L., Carlson K.E., Sun J., Katzenellenbogen B.S., Katzenellenbogen J.A.
Journal of Medicinal Chemistry,
2001
22.
10.1016/j.mencom.2016.07.016_sbref0060a
Keay
in Comprehensive Heterocyclic Chemistry III,
2008
23.

24.

25.
10.1016/j.mencom.2016.07.016_sbref0065a
Sheldrick
Program for the Solution of Crystal Structures SHELXS-97,
1997
26.
10.1016/j.mencom.2016.07.016_sbref0065b
Sheldrick
Program for Refinement of Crystal Structures SHELXL-97,
1997