Abstract
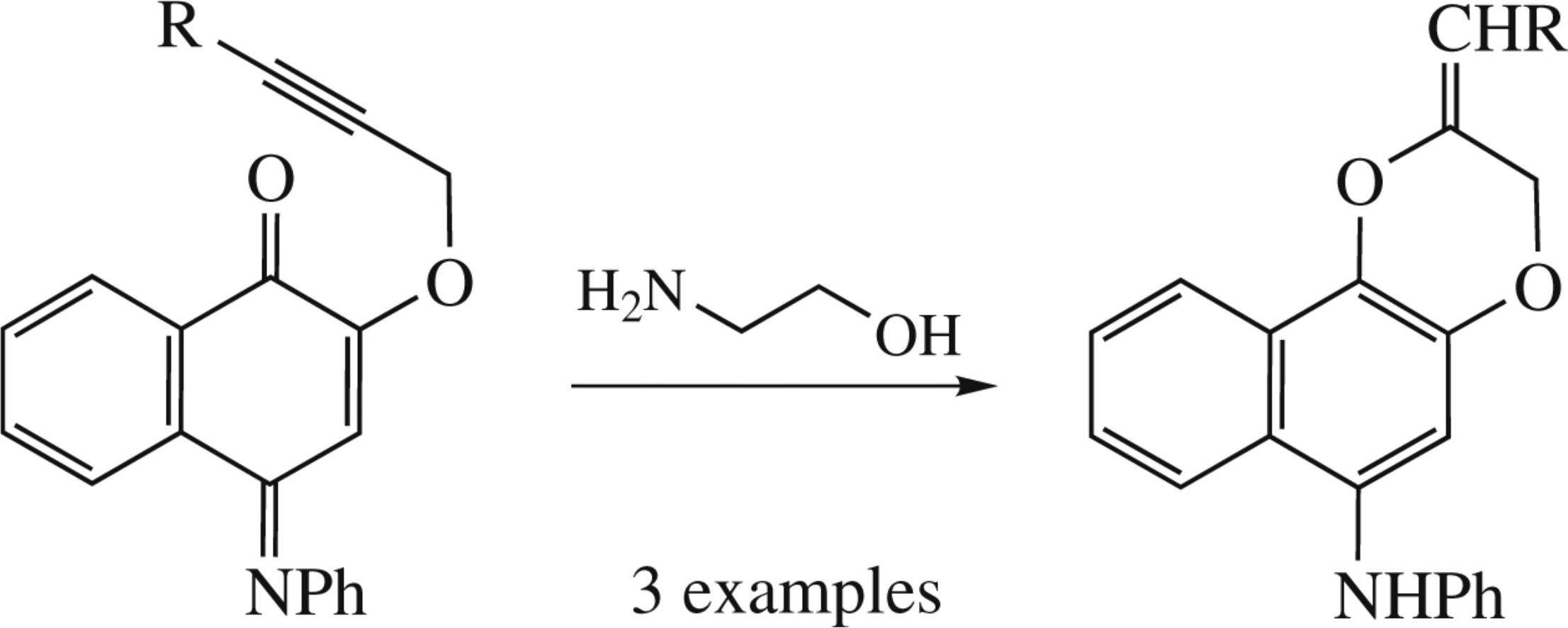
2-(3-R-prop-2-ynyloxy)-4-phenyliminonaphthalen-1(4H)- ones on heating with ethanolamine undergo cyclization into 2-R-methylidene-6-phenylamino-2,3-dihydronaphtho[2,1-b]- 1,4-dioxines.
References
1.

Zhou R., Luo G., Ewing A.
Journal of Neuroscience,
1994
2.

Fang Q.K., Grover P., Han Z., McConville F.X., Rossi R.F., Olsson D.J., Kessler D.W., Wald S.A., Senanayake C.H.
Tetrahedron Asymmetry,
2001
3.

Rouf A., Gupta P., Aga M.A., Kumar B., Chaubey A., Parshad R., Taneja S.C.
Tetrahedron Asymmetry,
2012
4.

Pinder R.M., Wieringa J.H.
Medicinal Research Reviews,
1993
5.

Hobbs J.J., King F.E.
Journal of the Chemical Society (Resumed),
1960
6.

Taniguchi E., Oshima Y.
Agricultural and Biological Chemistry,
1972
7.

Ishibashi F., Taniguchi E.
Phytochemistry,
1998
8.

Debenedetti S.L., Nadinic E.L., Coussio J.D., Kimpe N.D., Dupon J.F., Declercq J.P.
Phytochemistry,
1991
9.

Hänsel R., Schulz J., Pelter A.
Journal of the Chemical Society Chemical Communications,
1972
10.

Pelter A., Hänsel R.
Chemische Berichte,
1975
11.

Hibert M., Zimmermann A.
Journal of the Chemical Society Chemical Communications,
1986
12.

13.

Kuwabe S., Torraca K.E., Buchwald S.L.
Journal of the American Chemical Society,
2001
14.

Bao W., Liu Y., Lv X., Qian W.
Organic Letters,
2008
15.

Batanero B., Saez R., Barba F.
Electrochimica Acta,
2009
16.

Shvartsberg M.S., Barabanov I.I., Fedenok L.G.
Russian Chemical Reviews,
2004
17.

Vasilevskiy S.F., Baranov D.S.
Chemistry of Heterocyclic Compounds,
2013
18.
![Synthesis of benzo[g]quinoline-5,10-diones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kolodina E.A., Lebedeva N.I., Shvartsberg M.S.
Russian Chemical Bulletin,
2007
19.
Shvartsberg M.S., Kolodina E.A.
Mendeleev Communications,
2008
20.
Kolodina E.A., Shvartsberg M.S., Gritsan N.P.
Mendeleev Communications,
2008
21.
![Synthesis of benz[f]indole-4,9-diones via acetylenic derivatives of 1,4-naphthoquinone](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Shvartsberg M.S., Kolodina E.A., Lebedeva N.I., Fedenok L.G.
Tetrahedron Letters,
2009
22.

Shvartsberg M.S., Kolodina E.A., Lebedeva N.I., Fedenok L.G.
Russian Chemical Bulletin,
2012
23.
Kolodina E.A., Lebedeva N.I., Shvartsberg M.S.
Mendeleev Communications,
2012
24.

Baranov D.S., Gold B., Vasilevsky S.F., Alabugin I.V.
Journal of Organic Chemistry,
2012
25.
Baranov D.S., Popov A.G., Uvarov M.N., Kulik L.V.
Mendeleev Communications,
2014
26.
![Naphtho[4,3,2,1-lmn][2,9]phenanthrolines: Synthesis, сharacterization, optical properties and light-induced electron transfer in composites with the semiconducting polymer MEH-PPV](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Baranov D.S., Popov A.G., Uvarov M.N., Kazantsev M.S., Mostovich E.A., Glebov E.M., Kulik L.V.
Synthetic Metals,
2015
27.

Vasilevsky S.F., Baranov D.S., Mamatyuk V.I., Fadeev D.S., Gatilov Y.V., Stepanov A.A., Vasilieva N.V., Alabugin I.V.
Journal of Organic Chemistry,
2015
28.

Chowdhury C., Kundu N.G.
Chemical Communications,
1996
29.
![Palladium-Catalyzed Heteroannulation Leading to Heterocyclic Structures with Two Heteroatoms: A Highly Convenient and Facile Method for a Totally Regio- and Stereoselective Synthesis of (Z)-2,3-Dihydro-2-(ylidene)-1,4-benzo- and -naphtho[2,3-b]dioxins](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chowdhury C., Chaudhuri G., Guha S., Mukherjee A.K., Kundu N.G.
Journal of Organic Chemistry,
1998
30.
![A New Synthesis of 2,3-Dihydrobenzo[1,4]dioxine and 3,4-Dihydro-2H-benzo[1,4]oxazine Derivatives by Tandem Palladium-Catalyzed Oxidative Aminocarbonylation−Cyclization of 2-Prop-2-ynyloxyphenols and 2-Prop-2-ynyloxyanilines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Gabriele B., Salerno G., Veltri L., Mancuso R., Li Z., Crispini A., Bellusci A.
Journal of Organic Chemistry,
2006
31.

Hamasaka G., Uozumi Y.
Chemical Communications,
2014
32.

Yamamoto M.
Journal of the Chemical Society Perkin Transactions 1,
1979
33.

Meltsner M., Wohlberg C., Kleiner M.J.
Journal of the American Chemical Society,
1935
34.

Shvartsberg M.S., Ivanchikova I.D.
Tetrahedron Letters,
2000
35.

Harmon R.E., Phipps L.M., Howell J.A., Gupta S.K.
Tetrahedron,
1969