Abstract
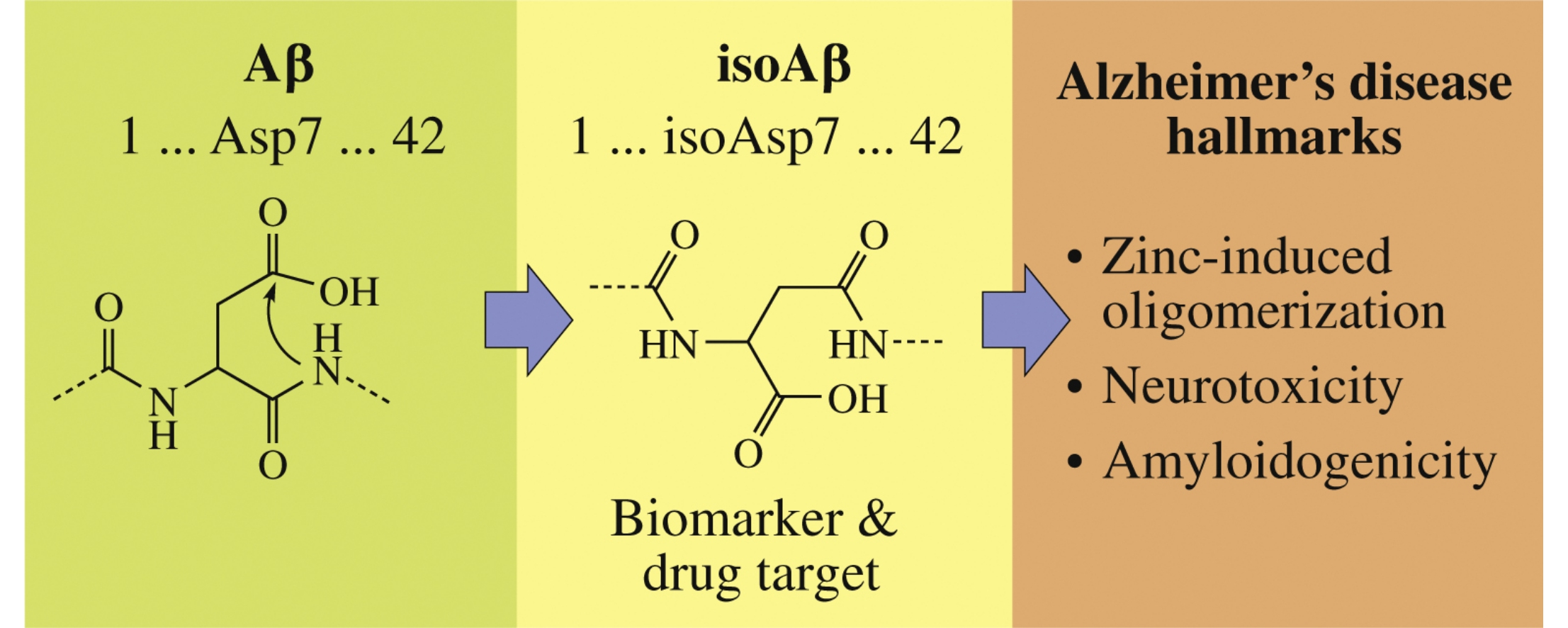
Recent data about structure, interaction with biometals, aggregation state, proteolysis by angiotensin converting enzyme, neurotoxicity and amyloidogenicity of amyloid-β containing isoaspartate 7 (isoAsp7-Aβ) in comparison with intact amyloid-β are summarized. Possible role of isoAsp7-Aβ as a prospective biomarker for molecular diagnostics and drug target in anti-amyloid therapy of Alzheimer's disease is discussed.
References
1.
10.1016/j.mencom.2016.07.001_bib0005
Alzheimer
Neurologisches Centralblatt,
1906
2.
10.1016/j.mencom.2016.07.001_bib0010
Alzheimer's & Dementia,
2014
3.
10.1016/j.mencom.2016.07.001_bib0015
Gavrilova
Farmakoterapiya bolezni Altsgeimera (Pharmacological Treatment of Alzheimer's Disease),
2007
4.

Querfurth H.W., LaFerla F.M.
New England Journal of Medicine,
2010
5.

6.

Cummings J.L., Morstorf T., Zhong K.
Alzheimer's Research and Therapy,
2014
7.

Karran E., Mercken M., Strooper B.D.
Nature Reviews Drug Discovery,
2011
8.

Schenk D., Basi G.S., Pangalos M.N.
Cold Spring Harbor perspectives in medicine,
2012
9.

Shankar G.M., Li S., Mehta T.H., Garcia-Munoz A., Shepardson N.E., Smith I., Brett F.M., Farrell M.A., Rowan M.J., Lemere C.A., Regan C.M., Walsh D.M., Sabatini B.L., Selkoe D.J.
Nature Medicine,
2008
10.

Villemagne V.L., Perez K.A., Pike K.E., Kok W.M., Rowe C.C., White A.R., Bourgeat P., Salvado O., Bedo J., Hutton C.A., Faux N.G., Masters C.L., Barnham K.J.
Journal of Neuroscience,
2010
11.

Friedlich A.L.
Journal of Neuroscience,
2004
12.

Faller P., Hureau C.
Dalton Transactions,
2009
13.

Kozin S.A., Zirah S., Rebuffat S., Hui Bon Hoa G., Debey P.
Biochemical and Biophysical Research Communications,
2001
14.

Kozin S.A., Mezentsev Y.V., Kulikova A.A., Indeykina M.I., Golovin A.V., Ivanov A.S., Tsvetkov P.O., Makarov A.A.
Molecular BioSystems,
2011
15.

Miller Y., Ma B., Nussinov R.
Proceedings of the National Academy of Sciences of the United States of America,
2010
16.

Lührs T., Ritter C., Adrian M., Riek-Loher D., Bohrmann B., Döbeli H., Schubert D., Riek R.
Proceedings of the National Academy of Sciences of the United States of America,
2005
17.

Dulin F., Léveillé F., Ortega J.B., Mornon J., Buisson A., Callebaut I., Colloc'h N.
FEBS Letters,
2008
18.

Walsh D.M., Klyubin I., Fadeeva J.V., Cullen W.K., Anwyl R., Wolfe M.S., Rowan M.J., Selkoe D.J.
Nature,
2002
19.

Mattson M.P.
Nature Structural and Molecular Biology,
1995
20.

Jiménez J.S.
Journal of Alzheimer's Disease,
2010
21.

B�ckig A., Tikkanen R., Herzog V., Schmitz A.
Histochemistry and Cell Biology,
2002
22.

Johnstone E.M., Babbey L.E., Stephenson D., Paul D.C., Santerre R.F., Clemens J.A., Williams D.C., Little S.P.
Biochemical and Biophysical Research Communications,
1996
23.

Ohyagi Y., Asahara H., Chui D., Tsuruta Y., Sakae N., Miyoshi K., Yamada T., Kikuchi H., Taniwaki T., Murai H., Ikezoe K., Furuya H., Kawarabayashi T., Shoji M., Checler F., et. al.
FASEB Journal,
2004
24.

Khmeleva S.A., Mezentsev Y.V., Kozin S.A., Tsvetkov P.O., Ivanov A.S., Bodoev N.V., Makarov A.A., Radko S.P.
Journal of Alzheimer's Disease,
2013
25.

Baker H.F., Ridley R.M., Duchen L.W., Crow T.J., Bruton C.J.
Molecular Neurobiology,
1994
26.

Ridley R.M., Baker H.F., Windle C.P., Cummings R.M.
Journal of Neural Transmission,
2005
27.

Langer F., Eisele Y.S., Fritschi S.K., Staufenbiel M., Walker L.C., Jucker M.
Journal of Neuroscience,
2011
28.

Morales R., Duran-Aniotz C., Castilla J., Estrada L.D., Soto C.
Molecular Psychiatry,
2011
29.

Rosen R.F., Fritz J.J., Dooyema J., Cintron A.F., Hamaguchi T., Lah J.J., LeVine H., Jucker M., Walker L.C.
Journal of Neurochemistry,
2011
30.

Watts J.C., Giles K., Grillo S.K., Lemus A., DeArmond S.J., Prusiner S.B.
Proceedings of the National Academy of Sciences of the United States of America,
2011
31.

Eisele Y.S., Bolmont T., Heikenwalder M., Langer F., Jacobson L.H., Yan Z., Roth K., Aguzzi A., Staufenbiel M., Walker L.C., Jucker M.
Proceedings of the National Academy of Sciences of the United States of America,
2009
32.

Eisele Y.S., Obermüller U., Heilbronner G., Baumann F., Kaeser S.A., Wolburg H., Walker L.C., Staufenbiel M., Heikenwalder M., Jucker M.
Science,
2010
33.

Meyer-Luehmann M., Coomaraswamy J., Bolmont T., Kaeser S., Schaefer C., Kilger E., Neuenschwander A., Abramowski D., Frey P., Jaton A.L., Vigouret J., Paganetti P., Walsh D.M., Mathews P.M., Ghiso J., et. al.
Science,
2006
34.

Stöhr J., Watts J.C., Mensinger Z.L., Oehler A., Grillo S.K., DeArmond S.J., Prusiner S.B., Giles K.
Proceedings of the National Academy of Sciences of the United States of America,
2012
35.

Kumar S., Rezaei-Ghaleh N., Terwel D., Thal D.R., Richard M., Hoch M., Mc Donald J.M., Wüllner U., Glebov K., Heneka M.T., Walsh D.M., Zweckstetter M., Walter J.
EMBO Journal,
2011
36.

Nussbaum J.M., Schilling S., Cynis H., Silva A., Swanson E., Wangsanut T., Tayler K., Wiltgen B., Hatami A., Rönicke R., Reymann K., Hutter-Paier B., Alexandru A., Jagla W., Graubner S., et. al.
Nature,
2012
37.

Tsvetkov P.O., Popov I.A., Nikolaev E.N., Archakov A.I., Makarov A.A., Kozin S.A.
ChemBioChem,
2008
38.

Kulikova A.A., Makarov A.A., Kozin S.A.
Molecular Biology,
2015
39.

Kozin S.A., Makarov A.A.
Zhurnal Nevrologii i Psikhiatrii imeni S.S. Korsakova,
2015
40.

Kostyukevich Y., Kononikhin A., Popov I., Indeykina M., Kozin S.A., Makarov A.A., Nikolaev E.
Journal of Mass Spectrometry,
2015
41.

Mekmouche Y., Coppel Y., Hochgräfe K., Guilloreau L., Talmard C., Mazarguil H., Faller P.
ChemBioChem,
2005
42.

Zirah S., Rebuffat S., Kozin S.A., Debey P., Fournier F., Lesage D., Tabet J.
International Journal of Mass Spectrometry,
2003
43.

Zirah S., Kozin S.A., Mazur A.K., Blond A., Cheminant M., Ségalas-Milazzo I., Debey P., Rebuffat S.
Journal of Biological Chemistry,
2006
44.

Tsvetkov P.O., Kulikova A.A., Golovin A.V., Tkachev Y.V., Archakov A.I., Kozin S.A., Makarov A.A.
Biophysical Journal,
2010
45.

Kozin S.A., Kulikova A.A., Istrate A.N., Tsvetkov P.O., Zhokhov S.S., Mezentsev Y.V., Kechko O.I., Ivanov A.S., Polshakov V.I., Makarov A.A.
Metallomics,
2015
46.

Kulikova A.A., Tsvetkov P.O., Indeykina M.I., Popov I.A., Zhokhov S.S., Golovin A.V., Polshakov V.I., Kozin S.A., Nudler E., Makarov A.A.
Molecular BioSystems,
2014
47.

Liu S., Howlett G., Barrow C.J.
Biochemistry,
1999
48.

Nisbet R.M., Nuttall S.D., Robert R., Caine J.M., Dolezal O., Hattarki M., Pearce L.A., Davydova N., Masters C.L., Varghese J.N., Streltsov V.A.
Proteins: Structure, Function and Genetics,
2013
49.
10.1016/j.mencom.2016.07.001_bib0245
Adzhubei
J. Biomol. Struct. Dyn.,
2016
50.

Adzhubei A.A., Sternberg M.J., Makarov A.A.
Journal of Molecular Biology,
2013
51.

De Strooper B., Simons M., Multhaup G., Van Leuven F., Beyreuther K., Dotti C.G.
EMBO Journal,
1995
52.

Shivers B.D., Hilbich C., Multhaup G., Salbaum M., Beyreuther K., Seeburg P.H.
EMBO Journal,
1988
53.

Istrate A., Tsvetkov P., Mantsyzov A., Kulikova A., Kozin S., Makarov A., Polshakov V.
Biophysical Journal,
2012
54.

Popov I.A., Indeikina M.I., Kononikhin A.S., Starodubtseva N.L., Kozin S.A., Makarov A.A., Nikolaev E.N.
Molecular Biology,
2013
55.

Istrate A.N., Kozin S.A., Zhokhov S.S., Mantsyzov A.B., Kechko O.I., Pastore A., Makarov A.A., Polshakov V.I.
Scientific Reports,
2016
56.
10.1016/j.mencom.2016.07.001_bib0280
Mezentsev
J. Biomol. Struct. Dyn.,
2016
57.

Shimizu T., Matsuoka Y., Shirasawa T.
Biological and Pharmaceutical Bulletin,
2005
58.

Reissner K.J., Aswad D.W.
Cellular and Molecular Life Sciences,
2003
59.

Shimizu T., Watanabe A., Ogawara M., Mori H., Shirasawa T.
Archives of Biochemistry and Biophysics,
2000
60.

Roher A.E., Lowenson J.D., Clarke S., Wolkow C., Wang R., Cotter R.J., Reardon I.M., Zürcher-Neely H.A., Heinrikson R.L., Ball M.J.
Journal of Biological Chemistry,
1993
61.

Roher A.E., Lowenson J.D., Clarke S., Woods A.S., Cotter R.J., Gowing E., Ball M.J.
Proceedings of the National Academy of Sciences of the United States of America,
1993
62.
10.1016/j.mencom.2016.07.001_bib0310
Tsvetkov
Biofizika,
2009
63.

Toropygin I.Y., Kugaevskaya E.V., Mirgorodskaya O.A., Elisseeva Y.E., Kozmin Y.P., Popov I.A., Nikolaev E.N., Makarov A.A., Kozin S.A.
Rapid Communications in Mass Spectrometry,
2007
64.

Yurinskaya M.M., Mitkevich V.A., Kozin S.A., Evgen'ev M.B., Makarov A.A., Vinokurov M.G.
Cell Death and Disease,
2015
65.

Yurinskaya M.M., Mit’kevich V.A., Barykin E.P., Garbuz D.G., Evgen’ev M.B., Makarov A.A., Vinokurov M.G.
Molecular Biology,
2015
66.

Barykin E.P., Petrushanko I.Y., Burnysheva K.M., Makarov A.A., Mitkevich V.A.
Molecular Biology,
2016
67.

Mitkevich V.A., Petrushanko I.Y., Yegorov Y.E., Simonenko O.V., Vishnyakova K.S., Kulikova A.A., Tsvetkov P.O., Makarov A.A., Kozin S.A.
Cell Death and Disease,
2013
68.

Manczak M., Anekonda T.S., Henson E., Park B.S., Quinn J., Reddy P.H.
Human Molecular Genetics,
2006
69.

Yurinskaya M.M., Mit’kevich V.A., Evgen’ev M.B., Makarov A.A., Vinokurov M.G.
Molecular Biology,
2016
70.

Fukuda H., Shimizu T., Nakajima M., Mori H., Shirasawa T.
Bioorganic and Medicinal Chemistry Letters,
1999
71.

Parri H., Dineley K.
Current Alzheimer Research,
2010
72.

Jucker M., Walker L.C.
Nature,
2013
73.

Hosoda R., Saido T.C., Otvos L., Arai T., Mann D.M., Lee V.M., Trojanowski J.Q., Iwatsubo T.
Journal of Neuropathology and Experimental Neurology,
1998
74.

Jucker M., Walker L.C.
Annals of Neurology,
2011
75.

Kozin S.A., Cheglakov I.B., Ovsepyan A.A., Telegin G.B., Tsvetkov P.O., Lisitsa A.V., Makarov A.A.
Neurotoxicity Research,
2013
76.

Borchelt D.R., Ratovitski T., van Lare J., Lee M.K., Gonzales V., Jenkins N.A., Copeland N.G., Price D.L., Sisodia S.S.
Neuron,
1997
77.

Garcia-Alloza M., Robbins E.M., Zhang-Nunes S.X., Purcell S.M., Betensky R.A., Raju S., Prada C., Greenberg S.M., Bacskai B.J., Frosch M.P.
Neurobiology of Disease,
2006
78.

Gowing E., Roher A.E., Woods A.S., Cotter R.J., Chaney M., Little S.P., Ball M.J.
Journal of Biological Chemistry,
1994
79.

Portelius E., Price E., Brinkmalm G., Stiteler M., Olsson M., Persson R., Westman-Brinkmalm A., Zetterberg H., Simon A.J., Blennow K.
Neurobiology of Aging,
2011
80.

Kulikova A.A., Cheglakov I.B., Kukharsky M.S., Ovchinnikov R.K., Kozin S.A., Makarov A.A.
Neurotoxicity Research,
2016
81.

Indeykina M.I., Popov I.A., Kozin S.A., Kononikhin A.S., Kharybin O.N., Tsvetkov P.O., Makarov A.A., Nikolaev E.N.
Analytical Chemistry,
2011
82.

Moreth J., Mavoungou C., Schindowski K.
Immunity and Ageing,
2013
83.

Busche M.A., Grienberger C., Keskin A.D., Song B., Neumann U., Staufenbiel M., Förstl H., Konnerth A.
Nature Neuroscience,
2015
84.

Lawrence J.L., Tong M., Alfulaij N., Sherrin T., Contarino M., White M.M., Bellinger F.P., Todorovic C., Nichols R.A.
Journal of Neuroscience,
2014
85.

Tsvetkov P.O., Cheglakov I.B., Ovsepyan A.A., Mediannikov O.Y., Morozov A.O., Telegin G.B., Kozin S.A.
Journal of Alzheimer's Disease,
2015
86.
Aisen P.S., Gauthier S., Ferris S.H., Saumier D., Haine D., Garceau D., Duong A., Suhy J., Oh J., Lau W.C., Sampalis J.
Archives of Medical Science,
2011
87.

Gervais F., Paquette J., Morissette C., Krzywkowski P., Yu M., Azzi M., Lacombe D., Kong X., Aman A., Laurin J., Szarek W.A., Tremblay P.
Neurobiology of Aging,
2007