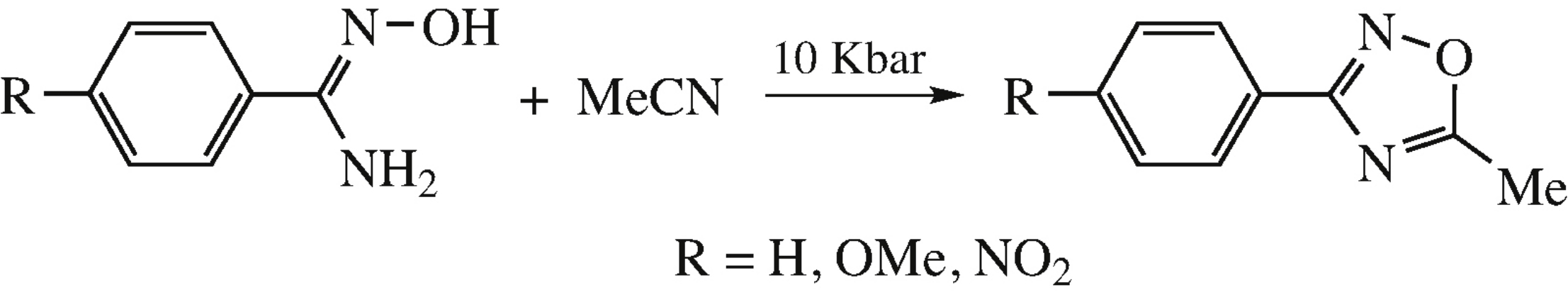Abstract
Reaction of amidoximes with acetonitrile giving 1,2,4-oxadiazoles occurs at 80–100°C under a pressure of 10 Kbar without catalysts.
References
1.

Yarovenko V.N., Zavarzin I.V., Krayushkin M.M.
Russian Chemical Bulletin,
1986
2.

Yarovenko V.N., Taralashvili V.K., Zavarzin I.V., Krayushkin M.M.
Tetrahedron,
1990
3.

Yarovenko V.N., Shirinyan V.Z., Zavarzin I.V., Krayushkin M.M.
Russian Chemical Bulletin,
1994
4.

Yarovenko V.N., Ugrak B.I., Krayushkin M.M., Shirinyan V.Z., Zavarzin I.V.
Russian Chemical Bulletin,
1994
5.

Augustine J.K., Akabote V., Hegde S.G., Alagarsamy P.
Journal of Organic Chemistry,
2009
6.

Redina T.N., Zharov A.A., Yarosh A.A., Ponomarenko V.A.
Russian Chemical Bulletin,
1995
7.

Zavarzin I.V., Zhulin V.M., Yarovenko V.N., Krayushkin M.M.
Russian Chemical Bulletin,
1988
8.
10.1016/j.mencom.2016.05.015_bib0040
Moskvin
2008
9.

Tadikonda R., Nakka M., Gajula M.B., Rayavarapu S., Gollamudi P.R., Vidavalur S.
Synthetic Communications,
2014
10.

Chiou S., Hoque A.K., Shine H.J.
Journal of Organic Chemistry,
1990
11.

Nicolaides D.N., Litinas K.E., Vrasidas I., Fylaktakidou K.C.
Journal of Heterocyclic Chemistry,
2004
12.

Otaka H., Ikeda J., Tanaka D., Tobe M.
Tetrahedron Letters,
2014
13.

Rostamizadeh S., Ghaieni H.R., Aryan R., Amani A.
Synthetic Communications,
2010
14.

Outirite M., Lebrini M., Lagrenée M., Bentiss F.
Journal of Heterocyclic Chemistry,
2007
15.
10.1016/j.mencom.2016.05.015_bib0075
Herrera
Synthesis,
2004