Abstract
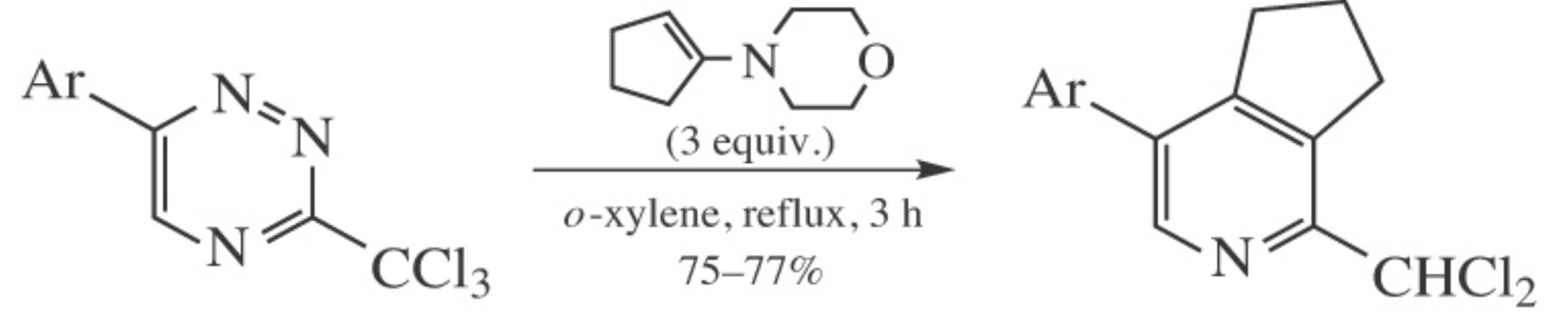
Reaction of 6-aryl-3-trichloromethyl-1,2,4-triazines with 1-morpholinocyclopentene affords 4-aryl-1-dichloromethyl- 6,7-dihydro-5H-cyclopenta[c]pyridines.
References
1.

Larsen A.F., Ulven T.
Organic Letters,
2011
2.

Mullins S.T., Sammes P.G., West R.M., Yahioglu G.
Journal of the Chemical Society Perkin Transactions 1,
1996
3.
![Reactivity ofβ-Carbolines and Cyclopenta[b]indolones Prepared from the Intramolecular Cyclization of 5(4H)-Oxazolones Derived fromL-Tryptophan](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Condie G., Bergman J.
European Journal of Organic Chemistry,
2004
4.

Langer P., Iaroshenko V., Mkrtchyan S., Ghazaryan G., Hakobyan A., Maalik A., Supe L., Villinger A., Tolmachev A., Ostrovskyi D., Sosnovskikh V., Ghochikyan T.
Synthesis,
2010
5.

Gasser J.K.
Journal of Agricultural Science,
1965
6.
Morris M.A., Page L.M.
Copeia,
1981
7.

LANG Y., CAO Z., JIANG X.
Talanta,
2005
8.

Gewald K., Hain U., Gruner M.
Chemische Berichte,
1985
9.

Varela J.A., Castedo L., Saá C.
Journal of Organic Chemistry,
2003
10.

Shvekhgeimer G.A., Kobrakov K.I., Toshkhodzhaev H.A.
Chemistry of Heterocyclic Compounds,
1994
11.

Sanchez J.P., Rogowski J.W.
Journal of Heterocyclic Chemistry,
1987
12.
10.1016/j.mencom.2016.04.014_sbref0040b
Graf
J. Prakt. Chem.,
1937
13.

Graf R., Zettl F.
Journal für praktische Chemie,
1936
14.

S. Ibrahim N., Hassan Mohamed M., Sobhy Ibrahim N., Hilmy Elnagdi M.
Heterocycles,
1987
15.

Ibrahim N.S., Mohamed M.H., Elnagdi M.H.
Archiv der Pharmazie,
1988
16.
![Syntheses of 3,4,7-triazaacenaphthylene and pyrido[3,4,5-de]cinnoline derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Elassar A.A., El-Kholy Y.M.
Heteroatom Chemistry,
2003
17.

Abdelrazek F.M., Ibrahim N.S., Kandeel Z.E., Elnagdi M.H.
Synthesis,
1984
18.

Yamamoto Y., Kinpara K., Nishiyama H., Itoh K.
Advanced Synthesis and Catalysis,
2005
19.

Clémence F., Le Martret O., Collard J.
Journal of Heterocyclic Chemistry,
1984
20.

Folli U., Goldoni F., Iarossi D., Sbardellati S., Taddei F.
Journal of the Chemical Society Perkin Transactions 2,
1995
21.

Newkome G.R., Puckett W.E., Kiefer G.E., Gupta V.D., Xia Y., Coreil M., Hackney M.A.
Journal of Organic Chemistry,
1982
22.

Julia L., Ballester M., Riera J., Castaner J., Ortin J.L., Onrubia C.
Journal of Organic Chemistry,
1988
23.

Chupp J.P., Smith L.R.
Journal of Heterocyclic Chemistry,
1988
24.
![Cp*RuCl-Catalyzed [2 + 2 + 2] Cycloadditions of α,ω-Diynes with Electron-Deficient Carbon−Heteroatom Multiple Bonds Leading to Heterocycles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yamamoto Y., Kinpara K., Saigoku T., Takagishi H., Okuda S., Nishiyama H., Itoh K.
Journal of the American Chemical Society,
2004
25.
![Ruthenium(ii)-catalyzed [2 + 2 + 2] cycloaddition of 1,6-diynes with electron-deficient nitriles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Yamamoto Y., Okuda S., Itoh K.
Chemical Communications,
2001
26.
![Efficient Generation of Pyridines by Ruthenium Carbene Mediated [2 + 2 + 2] Cyclotrimerization](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Medina S., Domínguez G., Pérez-Castells J.
Organic Letters,
2012
27.
![Novel Synthesis of 1,8-Alkanopyrido[3,4-d]pyridazine: a New Ring System](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elassar A.Z., Elkholy Y.M.
Chemistry of Heterocyclic Compounds,
2002
28.

Pabst G.R., Sauer J.
Tetrahedron Letters,
1998
29.

Rykowski A., Branowska D., Kielak J.
Tetrahedron Letters,
2000
30.

Kopchuk D.S., Khasanov A.F., Kovalev I.S., Zyryanov G.V., Kim G.A., Nikonov I.L., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
31.
10.1016/j.mencom.2016.04.014_sbref0090b
Catozzi
Synlett,
2007
32.
Kovalev I.S., Kopchuk D.S., Khasanov A.F., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2014
33.

Gonsalves A.M., Pinho e Melo T.M., Gilchrist T.L.
Tetrahedron,
1992
34.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
35.

Krinochkin A.P., Kopchuk D.S., Kozhevnikov D.N.
Polyhedron,
2015
36.

Kopchuk D.S., Chepchugov N.V., Kim G.A., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Russian Chemical Bulletin,
2015
37.

Kozhevnikov D.N., Kataeva N.N., Rusinov V.L., Chupakhin O.N.
Russian Chemical Bulletin,
2004
38.

Yamanaka H., Konno S., Yokoyama M.
Heterocycles,
1982
39.
![Regioselectivity in methylation and phenylation of the zwitterionic pyrido[2,1-f]-as-triazinium-1- and 3-olates and thiolates†](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bátori S., Messmer A.
Journal of Heterocyclic Chemistry,
1988
40.

KONNO S., SAGI M., TAKAHARU E., FUJIMURA S., HAYASHI K., YAMANAKA H.
Chemical and Pharmaceutical Bulletin,
2011
41.

Sawama Y., Imanishi T., Nakatani R., Fujiwara Y., Monguchi Y., Sajiki H.
Tetrahedron,
2014
42.

Guzmán A., Romero M., Talamás F.X., Muchowski J.M.
Tetrahedron Letters,
1992
43.

Cartwright D., Ferguson J.R., Giannopoulos T., Varvounis G., Wakefield B.J.
Journal of the Chemical Society Perkin Transactions 1,
1995
44.
Kopchuk D.S., Khasanov A.F., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhina O.N.
Mendeleev Communications,
2013
45.
10.1016/j.mencom.2016.04.014_sbref0135b
Danishefsky
Chem. Ind. (London),
1967