Abstract
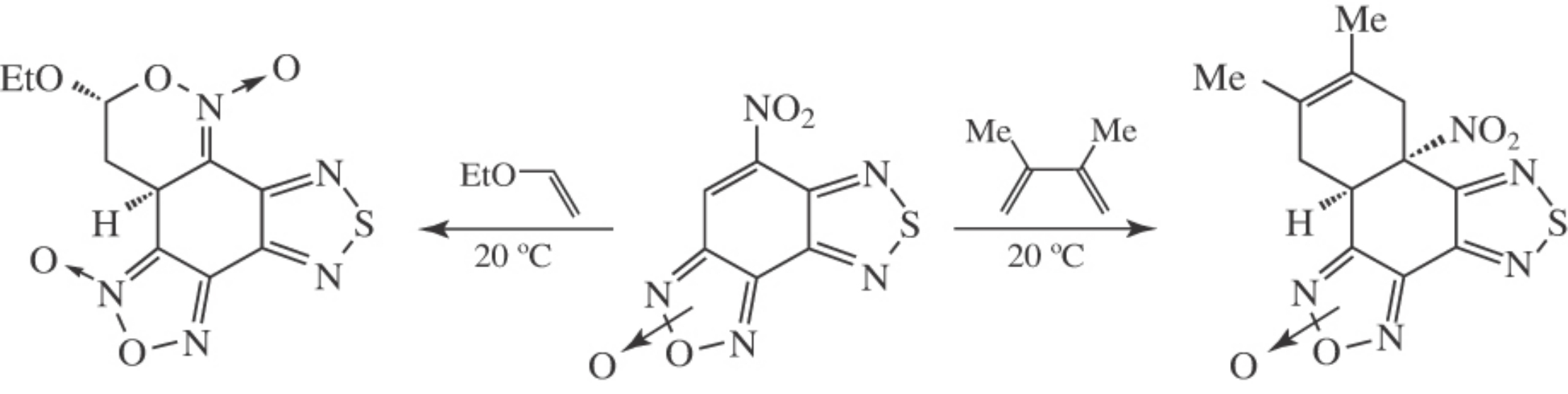
The Diels–Alder and Michael reactions of 5-nitro[1,2,5]-thiadiazolo[3,4-e]benzofuroxan afforded complex polycyclic compounds with potential NO-donor activity containing furoxan moiety along with another heterocyclic pharmacophoric fragments.
References
1.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
2.
Zlotin S.G., Churakov A.M., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2015
3.
![Facile Dearomatization of Nitroquinolines through [3+2] and [4+2] Cycloaddition Reactions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Fedyanin I.V., Shevelev S.A.
Asian Journal of Organic Chemistry,
2015
4.
Bastrakov M.A., Starosotnikov A.M., Fedyanin I.V., Kachala V.V., Shevelev S.A.
Mendeleev Communications,
2014
5.
![3-R-4-Nitro-6,7-furoxanobenzo[d]isoxazoles – a new type of condensed nitroarenes capable of Diels–Alder reaction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2015
6.

Cerecetto H., Porcal W.
Mini-Reviews in Medicinal Chemistry,
2005
7.

Cerecetto H., González M.
Topics in Heterocyclic Chemistry,
2007
8.

Jovene C., Chugunova E.A., Goumont R.
Mini-Reviews in Medicinal Chemistry,
2013
9.
Korolev S.P., Kondrashina O.V., Druzhilovsky D.S., Starosotnikov A.M., Dutov M.D., Bastrakov M.A., Dalinger I.L., Filimonov D.A., Shevelev S.A., Poroikov V.V., Agapkina Y.Y., Gottikh M.B.
Acta Naturae,
2013
10.

Granik V.G., Ryabova S.Y., Grigoriev N.B.
Russian Chemical Reviews,
1997
11.

Feelisch M.
Journal of Cardiovascular Pharmacology,
2008
12.

13.

Feelisch M., Schönafingeri K., Noack H.
Biochemical Pharmacology,
1992
14.

Medana C., Ermondi G., Fruttero R., Di Stilo A., Ferretti C., Gasco A.
Journal of Medicinal Chemistry,
1994
15.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
16.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
17.

Ghosh P.B., Everitt B.J.
Journal of Medicinal Chemistry,
1974
18.

Rai D., Chen W., Zhan P., Liu H., Tian Y., Liang X., De Clercq E., Pannecouque C., Balzarini J., Liu X.
Chemical Biology and Drug Design,
2014
19.

Ismail Z.H., Ghorab M.M., Mohamed E.M., Aly H.M., El-Gaby M.S.
Phosphorus, Sulfur and Silicon and the Related Elements,
2008
20.

Sauerberg P., Olesen P.H., Sheardown M.J., Suzdak P.D., Shannon H.E., Bymaster F.P., Calligaro D.O., Mitch C.H., Ward J.S., Swedberg M.D.
Life Sciences,
1995
21.

Sauerberg P., Jeppesen L., Olesen P.H., Rasmussen T., Swedberg M.D., Sheardown M.J., Fink-Jensen A., Thomsen C., Thøgersen H., Rimvall K., Ward J.S., Calligaro D.O., DeLapp N.W., Bymaster F.P., Shannon H.E., et. al.
Journal of Medicinal Chemistry,
1998
22.
10.1016/j.mencom.2016.04.013_bib0065
Pesin
Zh. Obshch. Khim.,
1964
23.

Kurbatov S., Goumont R., Marrot J., Terrier F.
Tetrahedron Letters,
2004
24.

Lakhdar S., Goumont R., Berionni G., Boubaker T., Kurbatov S., Terrier F.
Chemistry - A European Journal,
2007
25.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015