Abstract
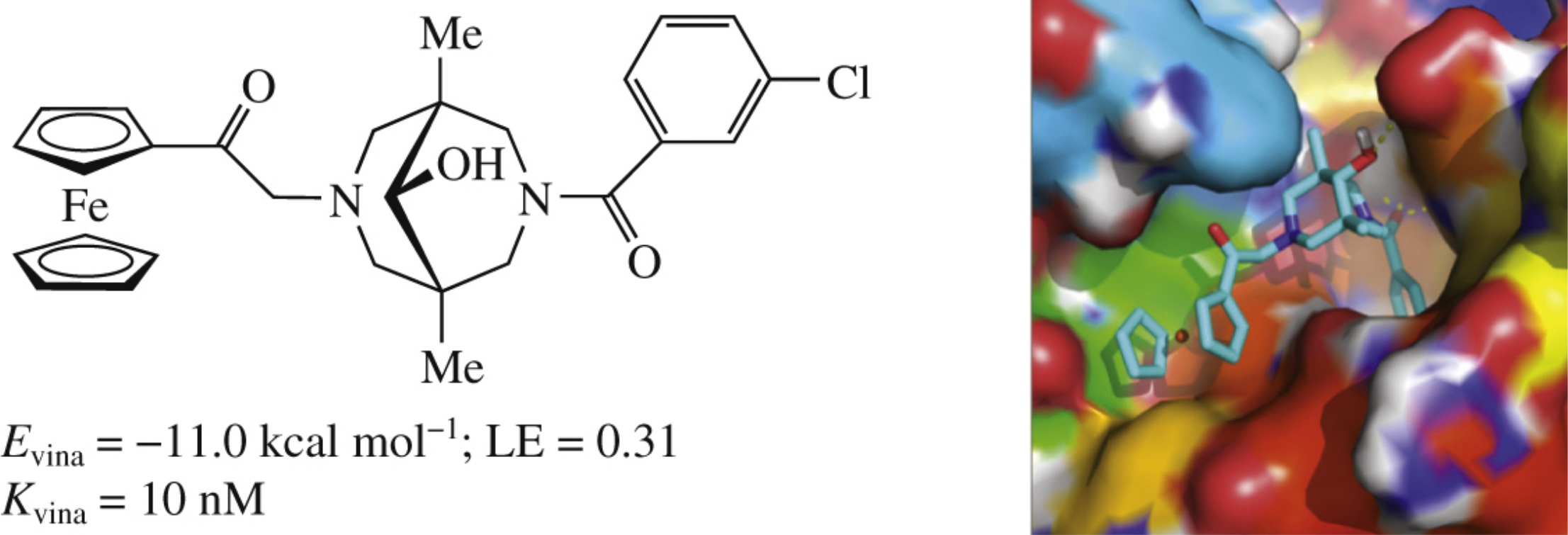
The docking study of ferrocene-substituted bispidines to binding sites of thrombin and factor Xa has shown that bispidine scaffold provides a 3D-arranegment of all substituents and a direction for the ferrocene group to fill the S4 pocket for both thrombin and factor Xa.
References
1.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
2.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
3.

Koutsoukas A., Simms B., Kirchmair J., Bond P.J., Whitmore A.V., Zimmer S., Young M.P., Jenkins J.L., Glick M., Glen R.C., Bender A.
Journal of Proteomics,
2011
4.

5.

Straub A., Roehrig S., Hillisch A.
Angewandte Chemie - International Edition,
2011
6.

Dubar F., Wintjens R., Martins-Duarte É.S., Vommaro R.C., de Souza W., Dive D., Pierrot C., Pradines B., Wohlkonig A., Khalife J., Biot C.
MedChemComm,
2011
7.

Hussain R.A., Badshah A., Sohail M., Lal B., Akbar K.
Journal of Molecular Structure,
2013
8.

Kudryavtsev K.V., Shulga D.A., Chupakhin V.I., Sinauridze E.I., Ataullakhanov F.I., Vatsadze S.Z.
Tetrahedron,
2014
9.

Politzer P., Murray J.S., Clark T.
Physical Chemistry Chemical Physics,
2013
10.

Bissantz C., Kuhn B., Stahl M.
Journal of Medicinal Chemistry,
2010
11.

Abel R., Salam N.K., Shelley J., Farid R., Friesner R.A., Sherman W.
ChemMedChem,
2011
12.
https://www.pymol.org/.
13.

Morris G.M., Huey R., Lindstrom W., Sanner M.F., Belew R.K., Goodsell D.S., Olson A.J.
Journal of Computational Chemistry,
2009
14.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
15.
https://www.chemaxon.com/products/marvin/marvinsketch.
16.

O'Boyle N.M., Banck M., James C.A., Morley C., Vandermeersch T., Hutchison G.R.
Journal of Cheminformatics,
2011
17.

Hanwell M.D., Curtis D.E., Lonie D.C., Vandermeersch T., Zurek E., Hutchison G.R.
Journal of Cheminformatics,
2012
18.

Baum B., Muley L., Heine A., Smolinski M., Hangauer D., Klebe G.
Journal of Molecular Biology,
2009
19.
![Discovery of the novel antithrombotic agent 5-chloro-N-({(5S)-2-oxo-3- [4-(3-oxomorpholin-4-yl)phenyl]-1,3-oxazolidin-5-yl}methyl)thiophene- 2-carboxamide (BAY 59-7939): an oral, direct factor Xa inhibitor.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Roehrig S., Straub A., Pohlmann J., Lampe T., Pernerstorfer J., Schlemmer K., Reinemer P., Perzborn E.
Journal of Medicinal Chemistry,
2005
20.

Bembenek S.D., Tounge B.A., Reynolds C.H.
Drug Discovery Today,
2009