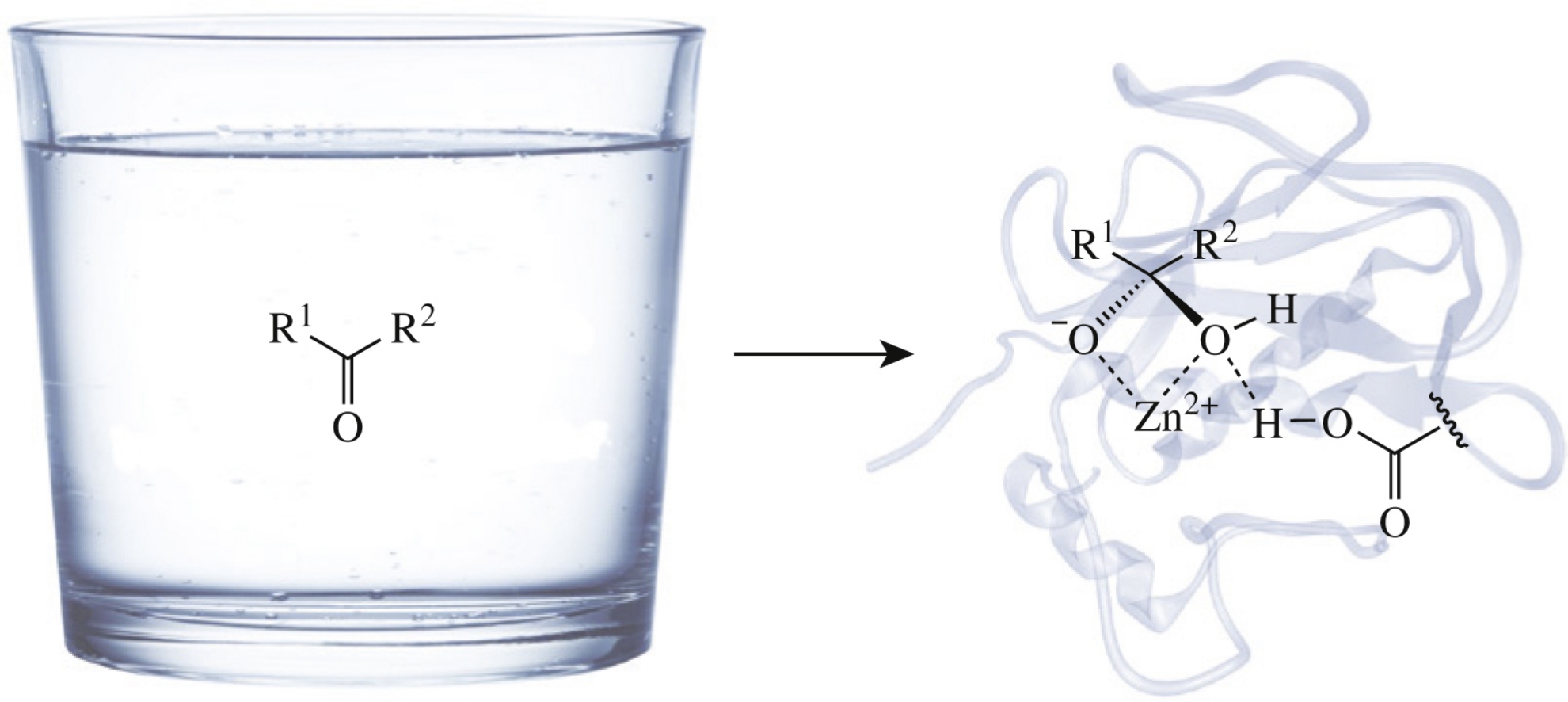
Abstract
A mimetic of a natural substrate of the matrix metalloproteinase MMP-2, in which the amide group with the scissile peptide bond is replaced with the oxoethylene moiety, inhibits the enzyme.
References
1.
Matrix Metalloproteinase Inhibitors. Specificity of Binding and Structure–Activity Relationships, ed. S. P. Gupta, Springer, Basel, 2012.
2.

Dormán G., Cseh S., Hajdú I., Barna L., Kónya D., Kupai K., Kovács L., Ferdinandy P.
Drugs,
2010
3.

Vandenbroucke R.E., Libert C.
Nature Reviews Drug Discovery,
2014
4.

Brinckerhoff C.E., Matrisian L.M.
Nature Reviews Molecular Cell Biology,
2002
5.

Ndinguri M., Bhowmick M., Tokmina-Roszyk D., Robichaud T., Fields G.
Molecules,
2012
6.

Higashi S., Miyazaki K.
Journal of Biological Chemistry,
2003
7.

Hashimoto H., Takeuchi T., Komatsu K., Miyazaki K., Sato M., Higashi S.
Journal of Biological Chemistry,
2011
8.

Khrenova M.G., Nemukhin A.V., Savitsky A.P.
Journal of Physical Chemistry B,
2014
9.

Netzel-Arnett S., Mallya S.K., Nagase H., Birkedal-Hansen H., Van Wart H.E.
Analytical Biochemistry,
1991
10.

Kovacs J., Kisfaludy L., Ceprini M.Q.
Journal of the American Chemical Society,
1967
11.

BENOITON N.L., LEE Y.C., STEINAUR R., CHEN F.M.
International journal of peptide & protein research,
2009
12.

EWENSON A., COHEN-SUISSA R., LEVIAN-TEITELBAUM D., SELINGER Z., CHOREV M., GILON C.
International journal of peptide & protein research,
2009