Abstract
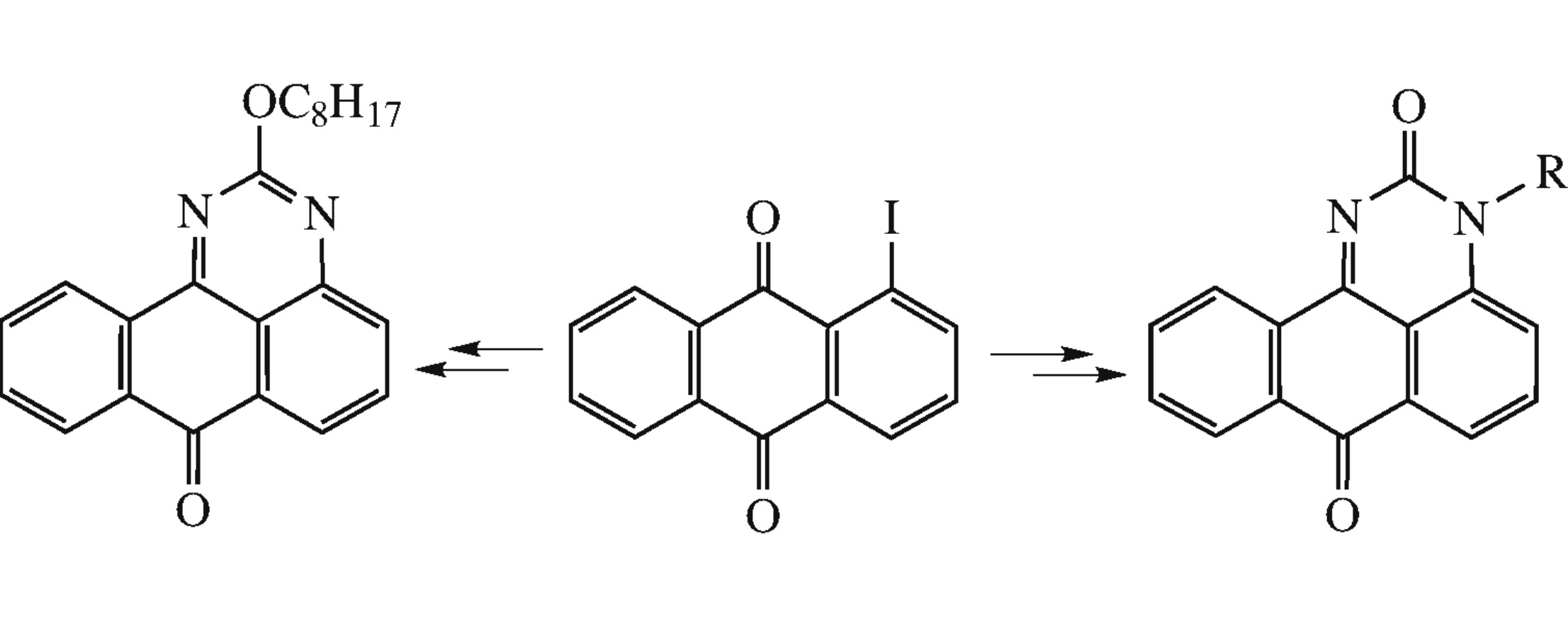
New 2-octyloxy-7H-benzo[e]perimidin-7-one and 3-benzylor 3-propargyl-3H-benzo[e]perimidine-2,7-diones were synthesized from 1-iodoanthraquinone. The propargyl derivative was subjected to Mannich and cycloaddition reactions.
References
1.
10.1016/j.mencom.2016.03.032_sbref0005a
Jones
Brit. J. Pharmacol.,
1952
2.
![6-[(Aminoalkyl)amino]-substituted 7H-benzo[e]perimidin-7-ones as novel antineoplastic agents. Synthesis and biological evaluation](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Stefanska B., Dzieduszycka M., Martelli S., Tarasiuk J., Bontemps-Gracz M., Borowski E.
Journal of Medicinal Chemistry,
1993
3.
D. R. Bhumralkar, R.A. Bychowski, D.S. Dhanoa, D.R. Luthin and A. K. Rabinovich, US Patent WO 1998008821 A1, 1998
4.
![8,11-Dihydroxy-6-[(aminoalkyl)amino]-7H-benzo[e]perimidin-7-ones with Activity in Multidrug-Resistant Cell Lines: Synthesis and Antitumor Evaluation](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Stefańska B., Dzieduszycka M., Bontemps-Gracz M.M., Borowski E., Martelli S., Supino R., Pratesi G., De Cesare M., Zunino F., Kuśnierczyk H., Radzikowski C.
Journal of Medicinal Chemistry,
1999
5.
![Synthesis and Cytotoxic Activity of 7-Oxo-7H-dibenz[f,ij]isoquinoline and 7-Oxo-7H-benzo[e]perimidine Derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Bu X., Deady L.W., Finlay G.J., Baguley B.C., Denny W.A.
Journal of Medicinal Chemistry,
2001
6.
![Effect of Modification of 6-[(Aminoalkyl)amino]-7H-benzo[e]-perimidin-7-ones on Their Cytotoxic Activity Toward Sensitive and Multidrug Resistant Tumor Cell Lines. Synthesis and Biological Evaluation](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dzieduszycka M., Martelli S., Arciemiuk M., Bontemps-Gracz M.M., Kupiec A., Borowski E.
Bioorganic and Medicinal Chemistry,
2002
7.
![Synthesis and cytotoxic activity of N-[(alkylamino)alkyl]carboxamide derivatives of 7-oxo-7H-benz[de]anthracene, 7-oxo-7H-naphtho[1,2,3-de]quinoline, and 7-oxo-7H-benzo[e]perimidine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bu X., Chen J., Deady L.W., Smith C.L., Baguley B.C., Greenhalgh D., Yang S., Denny W.A.
Bioorganic and Medicinal Chemistry,
2005
8.

Yu B., Meng L., Chen J., Zhou T., Cheng K., Ding J., Qin G.
Journal of Natural Products,
2001
9.

Min Y.D., Choi S.U., Lee K.R.
Archives of Pharmacal Research,
2006
10.

Derivatives of oxoisoaporphine alkaloids: A novel class of selective acetylcholinesterase inhibitors
Tang H., Ning F., Wei Y., Huang S., Huang Z., Chan A.S., Gu L.
Bioorganic and Medicinal Chemistry Letters,
2007
11.

Tang H., Wang X., Wei Y., Huang S., Huang Z., Tan J., An L., Wu J., Sun-Chi Chan A., Gu L.
European Journal of Medicinal Chemistry,
2008
12.

Castro-Castillo V., Rebolledo-Fuentes M., Theoduloz C., Cassels B.K.
Journal of Natural Products,
2010
13.

Li Y., Ning F., Yang M., Li Y., Nie M., Ou T., Tan J., Huang S., Li D., Gu L., Huang Z.
European Journal of Medicinal Chemistry,
2011
14.

Castro-Castillo V., Suárez-Rozas C., Pabón A., Pérez E.G., Cassels B.K., Blair S.
Bioorganic and Medicinal Chemistry Letters,
2013
15.
M. Battegay, French Patent 736174, 1931.
16.
I. G. Farbenind, UK Patent 385295, 1931.
17.
10.1016/j.mencom.2016.03.032_sbref0015c
Battegay
Compt. Rend.,
1932
18.
I. G. Farbenind, UK Patent 401731, 1933
19.
H. Weidinger, H. Eilingsfeld and G. Haese, German Patent 1159456, 1963
20.
W. Hohmann, German Patent 1232296, 1967
21.

Adam J., Winkler T., Rihs G.
Helvetica Chimica Acta,
1982
22.
Z. Golubski and R. Kowal, Polish Patent 192093, 2006
23.

Wang Z.D., Eilander J., Yoshida M., Wang T.
European Journal of Organic Chemistry,
2014
24.
J. F. Ryley and G. J. Stacey, UK Patent 876719, 1959.
25.
S. I. Popov, I.B. Krasnova and N. S. Dokunichin, SU Patent 388557, 1974.
26.

NISHIO K., KASAI T., TSURUOKA S.
The Journal of the Society of Chemical Industry Japan,
1968
27.

Brown D.J., Hoerger E., Mason S.F.
Journal of the Chemical Society (Resumed),
1955
28.

Brown D.J., Foster R.V.
Journal of the Chemical Society (Resumed),
1965
29.

Lanni E.L., Bosscher M.A., Ooms B.D., Shandro C.A., Ellsworth B.A., Anderson C.E.
Journal of Organic Chemistry,
2008
30.
10.1016/j.mencom.2016.03.032_sbref0045a
Acetylene Chemistry: Chemistry Biology and Material Science,
2005
31.

Chinchilla R., Nájera C.
Chemical Reviews,
2013
32.

Tron G.C., Pirali T., Billington R.A., Canonico P.L., Sorba G., Genazzani A.A.
Medicinal Research Reviews,
2008
33.

Thirumurugan P., Matosiuk D., Jozwiak K.
Chemical Reviews,
2013
34.
gNMR 5.0 software, http://home.cc.umanitoba.ca/∼budzelaa/gNMR/gNMR.html.