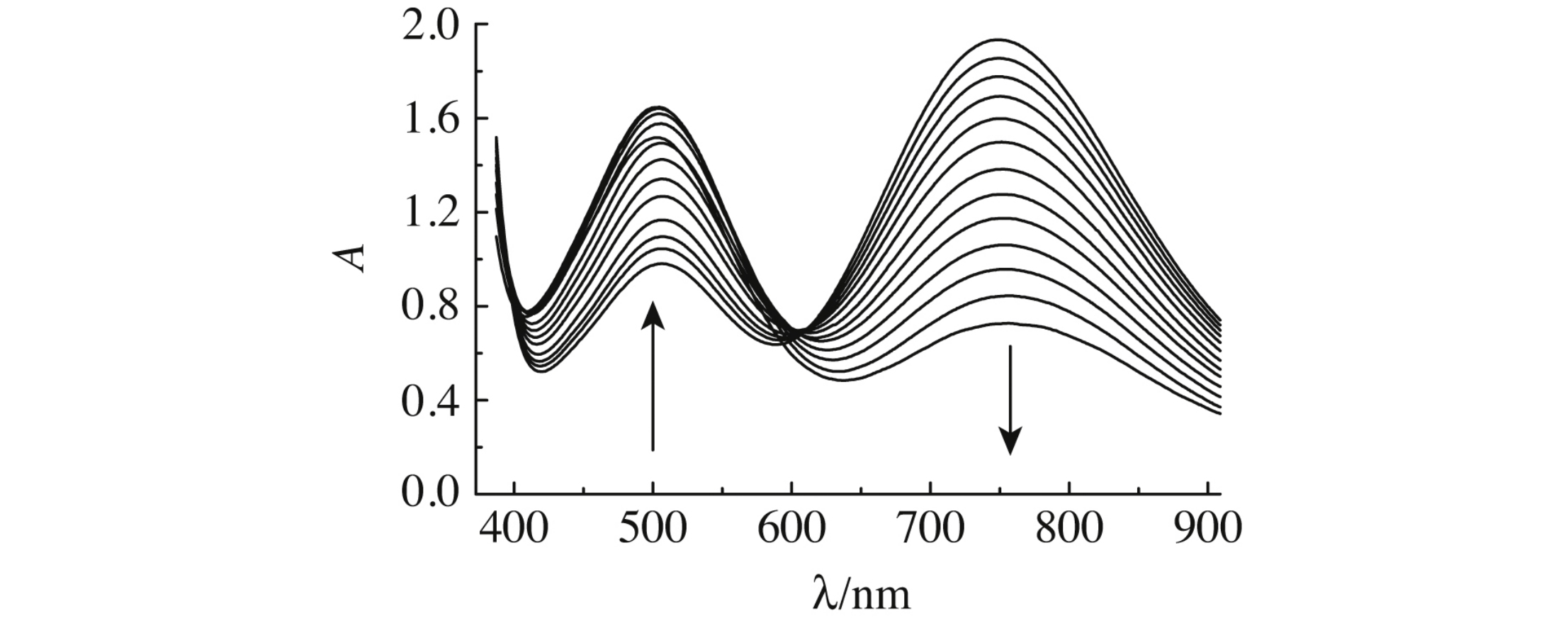
Abstract
The ion associates [Fe(CN)6]4–, M+, [Cu edta]2–, which are observed in the UV-VIS spectra of K4[Fe(CN)6] + Na2[Cu edta] solutions as an absorption band at 500 nm, contain alkali metal cations, and they are more stable in water-organic solutions than in aqueous solutions.
References
1.
10.1016/j.mencom.2016.03.025_bib0005
Basolo
Mechanisms of Inorganic Reactions,
1967
2.

Fuoss R.M.
Journal of Solution Chemistry,
1986
3.

Hemmes P.
Journal of the American Chemical Society,
1972
4.
10.1016/j.mencom.2016.03.025_bib0020
Zaidi
J. Chem. Soc. Pak.,
1991
5.
Nikol'skii A.B., Kotov V.Y.
Mendeleev Communications,
1995
6.
10.1016/j.mencom.2016.03.025_bib0030
Kotov
Russ. Chem. Bull,
1999
7.

Billing R., Khoshtariya D.E.
Inorganic Chemistry,
1994
8.

Kershaw M.R., Prue J.E.
Transactions of the Faraday Society,
1967
9.

Chlebek R.W., Lister M.W.
Canadian Journal of Chemistry,
1966
10.
![Ion association in the Na[CoEdta]-KI-sucrose system](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Kotov V.Y., Gordova A.F., Ivannikova Y.I., Ilyukhin A.B., Laurinavichute V.K.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2009
11.

Hemmes P., Costanzo J.N., Jordan F.
The Journal of Physical Chemistry,
1978
12.
Birin K.P., Dunin D.S., Tolstykh M.V., Kotov V.Y.
Mendeleev Communications,
2012
13.
Vannerberg N., Pedersen E., Romanowska E., Rudén U., Pilotti Å.
Acta Chemica Scandinavica,
2008
14.
10.1016/j.mencom.2016.03.025_bib0070
Porai-Koshits
Kristallografiya,
1973
15.
Y. Y. Akhadov, Dielectric Properties of Binary Solutions, Pergamon, Oxford, 1981.
16.
Kotov V.Y.
Mendeleev Communications,
2001
17.
Kostina S.A., Ilyukhin A.B., Lokshin B.V., Kotov V.Y.
Mendeleev Communications,
2001
18.

Benoit R.L., Buisson C.
Inorganica Chimica Acta,
1973