Abstract
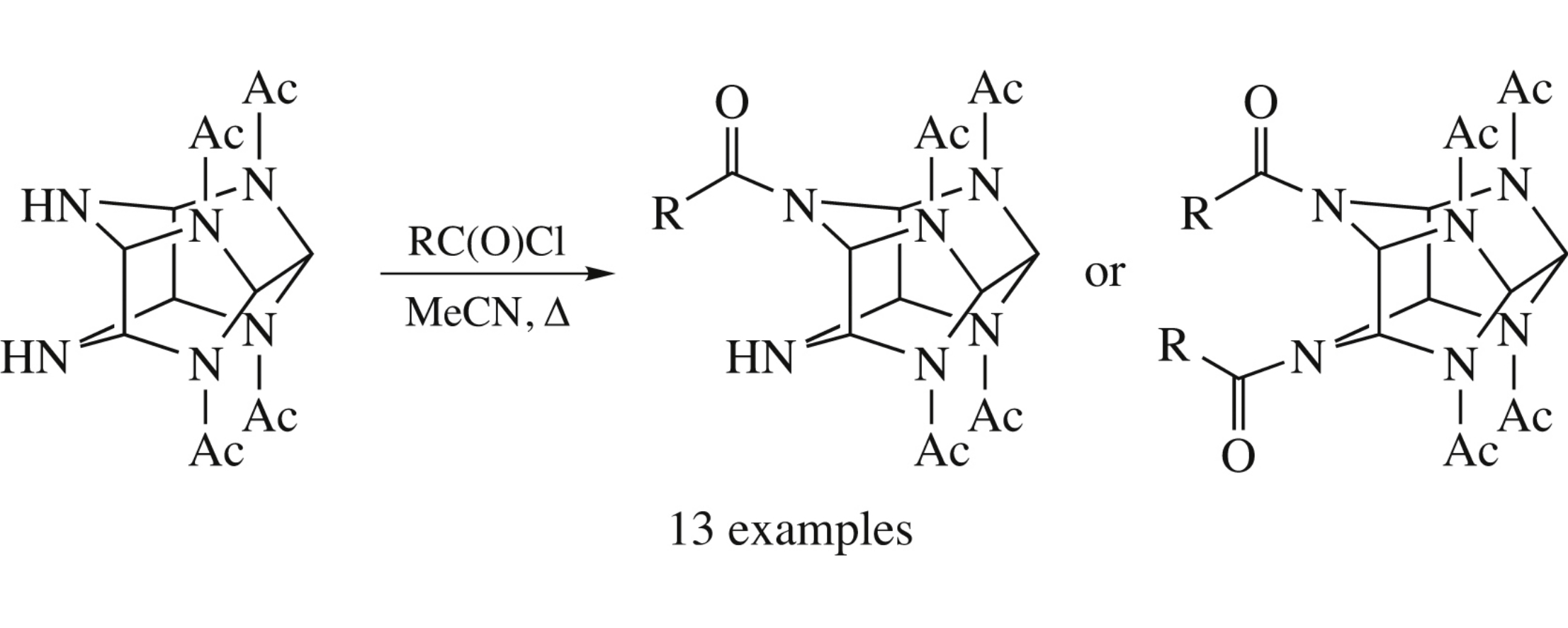
4-Mono- and 4,10-diacyl derivatives of 2,6,8,12-tetraacetyl-2,4,6,8,10,12-hexaazatetracyclo[5.5.0.03,11.05,9]dodecane were obtained by its treatment with acid chlorides in boiling acetonitrile.
References
1.

Bellamy A., MacCuish A., Golding P., Mahon M.
Propellants, Explosives, Pyrotechnics,
2007
2.
10.1016/j.mencom.2016.03.019_bib0010
Fang
Chin. J. Energ. Mater,
2008
3.
R. B. Wardle and J. C. Hinshaw, US Patent 6147209 (C06B 25/00), 1992.
4.

Sysolyatin S.V., Lobanova A.A., Chernikova Y.T., Sakovich G.V.
Russian Chemical Reviews,
2005
5.
10.1016/j.mencom.2016.03.019_bib0025
Qian
J. Indian Chem. Soc.,
2008
6.

Dong K., Wang Y., Gong X., Zhang J., Sun C., Pang S.
New Journal of Chemistry,
2013
7.
10.1016/j.mencom.2016.03.019_bib0035
Sysolyatin
Polzunovskii Vestnik,
2013
8.

Dong K., Sun C.H., Song J.W., Wei G.X., Pang S.P.
Organic Process Research and Development,
2014