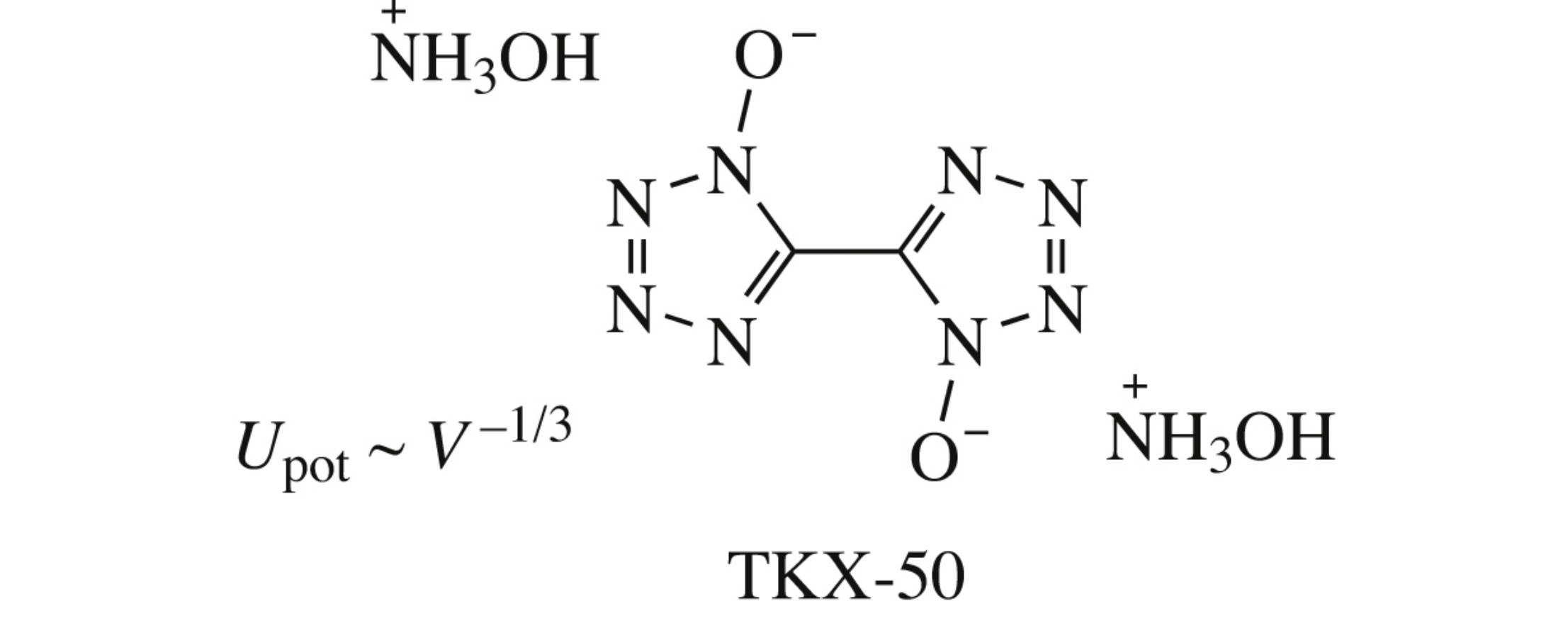
Abstract
The enthalpies of formation were calculated for a number of bitetrazole salts using classical quantum chemistry methods and an improved method of isodesmic reactions, and the latter was found most accurate in the assessment of this characteristic.
References
1.

2.
10.1016/j.mencom.2016.03.017_bib0010
Klapötke
Chemistry of High-energy Materials,
2012
3.

Jenkins H.D., Roobottom H.K., Passmore J., Glasser L.
Inorganic Chemistry,
1999
4.

Jenkins H.D., Glasser L.
Inorganic Chemistry,
2006
5.

Khakimov D., Dalinger I., Pivina T.
Computational and Theoretical Chemistry,
2015
6.

McKee M.L., Buehl M., Schleyer P.V.
Inorganic Chemistry,
1993
7.

Csonka G.I., Hencsei P.
Journal of Molecular Structure THEOCHEM,
1996
8.
10.1016/j.mencom.2016.03.017_bib0035
Frisch
Gaussian 98,
1998
9.
10.1016/j.mencom.2016.03.017_bib0040
Fischer
J. Mater. Chem.,
2012
10.

Sinditskii V.P., Filatov S.A., Kolesov V.I., Kapranov K.O., Asachenko A.F., Nechaev M.S., Lunin V.V., Shishov N.I.
Thermochimica Acta,
2015
11.
T. S. Kon’kova, Yu. N. Matyushin, A.I. Vakhtina, E.A. Miroshnichenko, A.F. Asachenko, P.B. Dzevakov and N. I. Shishov, Proceedings of the All-Russian Scientific and Technical Conference ‘Advances Special Chemistry and Chemical Technology”, the Russian University of Chemical Technology, 2015.(in press).
12.

Glasser L., Jenkins H.D., Klapötke T.M.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2014