Abstract
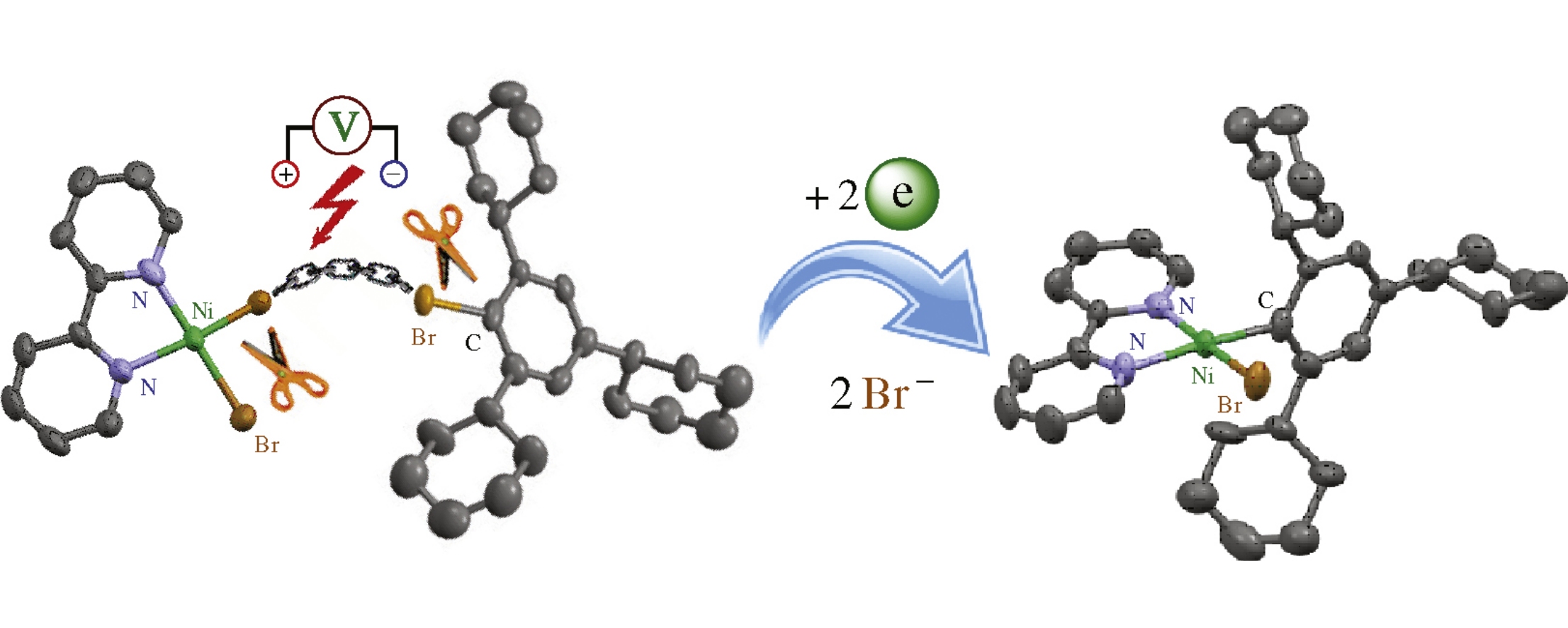
The electrochemical reduction of [NiBr2(bpy)] (bpy is 2,2’-bipyridine) complex in the presence of 2,4,6-tricyclohexylphenylbromide (TchpBr) in undivided electrochemical cell supplied with a sacrificial nickel anode results in the one-pot formation of an organonickel complex [NiBr(Tchp)(bpy)] bearing three cyclic substituents in the s-bonded aromatic ring. The synthesized complex was characterized by various methods including single crystal X-ray analysis.
References
1.
10.1016/j.mencom.2016.03.016_sbref0005a
2006
2.
10.1016/j.mencom.2016.03.016_sbref0005b
Nédeléc
1997
3.
10.1016/j.mencom.2016.03.016_sbref0005c
Tuck
1993
4.

Klein A., Budnikova Y.H., Sinyashin O.G.
Journal of Organometallic Chemistry,
2007
5.

Budnikova Y.H.
Russian Chemical Reviews,
2002
6.

Budnikova Y.H., Perichon J., Yakhvarov D.G., Kargin Y.M., Sinyashin O.G.
Journal of Organometallic Chemistry,
2001
7.

Yakhvarov D.G., Budnikova Y.G., Sinyashin O.G.
Russian Journal of Electrochemistry,
2003
8.
10.1016/j.mencom.2016.03.016_sbref0015a
Jolly
The Organic Chemistry of Nickel,
1975
9.
10.1016/j.mencom.2016.03.016_sbref0015b
Smith
1982
10.
10.1016/j.mencom.2016.03.016_sbref0015c
2005
11.
10.1016/j.mencom.2016.03.016_sbref0015d
Cámpora
2007
12.

Yakhvarov D.G., Khusnuriyalova A.F., Sinyashin O.G.
Organometallics,
2014
13.

Yakhvarov D.G., Kvashennikova S.V., Sinyashin O.G.
Russian Chemical Bulletin,
2013
14.

Yakhvarov D.G., Gorbachuk E.V., Sinyashin O.G.
European Journal of Inorganic Chemistry,
2013
15.

Yakhvarov D.G., Gorbachuk E.V., Kagirov R.M., Sinyashin O.G.
Russian Chemical Bulletin,
2012
16.
Gorbachuk E.V., Khayarov K.R., Sinyashin O.G., Yakhvarov D.G.
Mendeleev Communications,
2014
17.

Experimental evidence of phosphine oxide generation in solution and trapping by ruthenium complexes.
Yakhvarov D., Caporali M., Gonsalvi L., Latypov S., Mirabello V., Rizvanov I., Sinyashin O., Stoppioni P., Peruzzini M.
Angewandte Chemie - International Edition,
2011
18.

Chatt J., Shaw B.L.
Journal of the Chemical Society (Resumed),
1960
19.

Uchino M., Asagi K., Yamamoto A., Ikeda S.
Journal of Organometallic Chemistry,
1975
20.

Coronas J.M., Rossell O., Sales J.
Journal of Organometallic Chemistry,
1976
21.
![Reactivity of [NiR(R′)L2] compounds and the crystal structure of [Ni(C2Cl3)(C6H2Me3-2,4,6)(PMe2Ph)2]](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Reactivity of [NiR(R′)L2] compounds and the crystal structure of [Ni(C2Cl3)(C6H2Me3-2,4,6)(PMe2Ph)2]
Coronas J.M., Muller G., Rocamora M., Miravitlles C., Solans X.
Journal of the Chemical Society Dalton Transactions,
1985
22.

Yamamoto T., Kohara T., Osakada K., Yamamoto A.
Bulletin of the Chemical Society of Japan,
1983
23.
10.1016/j.mencom.2016.03.016_sbref0045a
Moss
Soc. A,
1966
24.

Cusumano M., Ricevuto V.
Journal of the Chemical Society Dalton Transactions,
1978
25.
10.1016/j.mencom.2016.03.016_sbref0045c
Fahey
Chim. Acta,
1979
26.

Klein A.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2001
27.

Feth M., Klein A., Bertagnolli H.
European Journal of Inorganic Chemistry,
2003
28.

Klein A., Feth M., Bertagnolli H., Záliš S.
European Journal of Inorganic Chemistry,
2004
29.
![π-Stacking Modulates the Luminescence of [(dppz)Ni(Mes)Br] (dppz = dipyrido[3,2-a:2′,3′-c]phenazine, Mes = 2,4,6-trimethylphenyl)†](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Klein A., Hurkes N., Kaiser A., Wielandt W.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2007
30.
10.1016/j.mencom.2016.03.016_bib0055
Seidel
Chem,
1985
31.

Walg S.P., Schmidt A.D., Schmitz M., Farsadpour S., Lang J., Niebergall M., Sun Y., Roesky P.W., Niedner-Schatteburg G., Thiel W.R.
Dalton Transactions,
2015
32.

Higgs A.T., Zinn P.J., Simmons S.J., Sanford M.S.
Organometallics,
2009
33.

Higgs A.T., Zinn P.J., Sanford M.S.
Organometallics,
2010
34.

Renz A.L., Pérez L.M., Hall M.B.
Organometallics,
2011
35.
10.1016/j.mencom.2016.03.016_sbref0060e
Tsybizova
Chem. A.,
2013
36.

Cheng J., Deming T.J.
Macromolecules,
1999
37.
![Synthesis and imaging validation of [¹⁸F]MDL100907 enabled by Ni-mediated fluorination.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ren H., Wey H., Strebl M., Neelamegam R., Ritter T., Hooker J.M.
ACS Chemical Neuroscience,
2014
38.

Klein A., Biewer C., Hamacher C., Hurkes N., Perez Outeiral J., Paniagua E.M., Schmieder A., Schüren A.O., Burma P.R., Ciszewski J.T., Vicic D.A.
European Journal of Inorganic Chemistry,
2012
39.

Laskowski C.A., Bungum D.J., Baldwin S.M., Del Ciello S.A., Iluc V.M., Hillhouse G.L.
Journal of the American Chemical Society,
2013
40.

Pestov A.V., Slepukhin P.A., Charushin V.N.
Russian Chemical Reviews,
2015
41.

Yakhvarov D.G., Trofimova E.A., Rizvanov I.K., Fomina O.S., Sinyashin O.G.
Russian Journal of Electrochemistry,
2011
42.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
43.

Klein A., Kaiser A., Wielandt W., Belaj F., Wendel E., Bertagnolli H., Záliš S.
Inorganic Chemistry,
2008
44.

Spek A.L.
Acta Crystallographica Section D Biological Crystallography,
2009
45.
![Electrochemical synthesis of the σ-aryl complex [NiBr(Mes)(bpy)] and its use as catalyst precursor for the oligomerization of ethylene (Mes = 2,4,6-trimethylphenyl, bpy = 2,2'-bipyridine)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yakhvarov D.G., Tazeev D.I., Sinyashin O.G., Giambastiani G., Bianchini C., Segarra A.M., Lönnecke P., Hey-Hawkins E.
Polyhedron,
2006
46.
![Synthesis, structure and electrochemical properties of the organonickel complex [NiBr(Mes)(phen)] (Mes = 2,4,6-trimethylphenyl, phen = 1,10-phenanthroline)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yakhvarov D.G., Petr A., Kataev V., Büchner B., Gómez-Ruiz S., Hey-Hawkins E., Kvashennikova S.V., Ganushevich Y.S., Morozov V.I., Sinyashin O.G.
Journal of Organometallic Chemistry,
2014