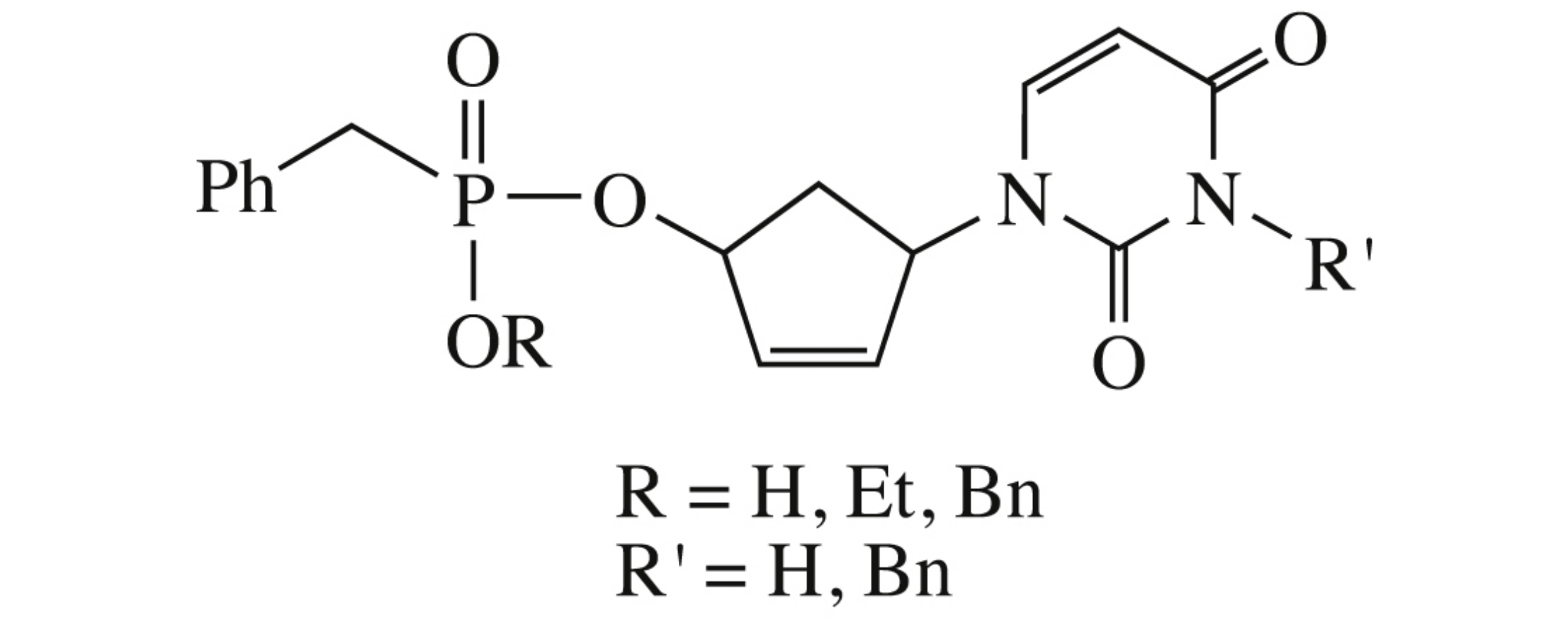
Abstract
Benzylphosphonates of 5’-norcarbocyclic analogue of 2’,3’-dideoxy-2’,3’-didehydrouridine and its N3-benzyl derivatives with different substituents at the phosphorus atom were designed and synthesized in attempt to improve solubility of potential non-nucleoside reverse transcriptase inhibitor. Solubility of the most hydrophilic representative was 400 mg in 100 ml of 1% DMSO solution in water. The target phosphonates were examined against HIV-1 reverse transcriptase with KI for all compounds being higher than 100 μM.
References
1.
http://www.who.int/mediacentre/factsheets/fs360/ru.
2.
10.1016/j.mencom.2016.03.009_bib0010
Levy
HIV and the Pathogenesis of AIDS,
2010
3.

Song Y., Fang Z., Zhan P., Liu X.
Current Medicinal Chemistry,
2013
4.

De Clercq E.
Biochemical Pharmacology,
2011
5.

Shirokova E.A., Jasko M.V., Khandazhinskaya A.L., Ivanov A.V., Yanvarev D.V., Skoblov Y.S., Mitkevich V.A., Bocharov E.V., Pronyaeva T.R., Fedyuk N.V., Kukhanova M.K., Pokrovsky A.G.
Journal of Medicinal Chemistry,
2004
6.

Khandazhinskaya A.L., Yanvarev D.V., Jasko M.V., Shipitsin A.V., Khalizev V.A., Shram S.I., Skoblov Y.S., Shirokova E.A., Kukhanova M.K.
Drug Metabolism and Disposition,
2008
7.

Shirokova E.A., Jasko M.V., Khandazhinskaya A.L., Yanvarev D.V., Skoblov Y.S., Pronayeva T.R., Fedyuk N.V., Pokrovsky A.G., Kukhanova M.K.
Nucleosides, Nucleotides and Nucleic Acids,
2003
8.

Khandazhinskaya A., Matyugina E., Shirokova E.
Expert Opinion on Drug Metabolism and Toxicology,
2010
9.
Khandazhinskaya А.L., Shirokova E.A.
Acta Naturae,
2013
10.

Staake M., Chauhan J., Zhou D., Shanker A., Chatterjee A.D., Das S., Patterson S.E.
Organic Letters,
2010
11.

Zhou D., Staake M., Patterson S.E.
Organic Letters,
2008
12.

Suk D., Rejman D., Dykstra C.C., Pohl R., Pankiewicz K.W., Patterson S.E.
Bioorganic and Medicinal Chemistry Letters,
2007
13.

Suk D., Bonnac L., Dykstra C.C., Pankiewicz K.W., Patterson S.E.
Bioorganic and Medicinal Chemistry Letters,
2007
14.

Rejman D., Olesiak M., Chen L., Patterson S.E., Wilson D., Jayaram H.N., Hedstrom L., Pankiewicz K.W.
Journal of Medicinal Chemistry,
2006
15.

Matyugina E.S., Valuev-Elliston V.T., Babkov D.A., Novikov M.S., Ivanov A.V., Kochetkov S.N., Balzarini J., Seley-Radtke K.L., Khandazhinskaya A.L.
MedChemComm,
2013
16.

Matyugina E.S., Valuev-Elliston V.T., Geisman A.N., Novikov M.S., Chizhov A.O., Kochetkov S.N., Seley-Radtke K.L., Khandazhinskaya A.L.
MedChemComm,
2013
17.

Hegde V.R., Seley K.L., Schneller S.W.
Nucleosides, Nucleotides and Nucleic Acids,
2000
18.

Matyugina E., Khandazhinskaya A., Chernousova L., Andreevskaya S., Smirnova T., Chizhov A., Karpenko I., Kochetkov S., Alexandrova L.
Bioorganic and Medicinal Chemistry,
2012
19.

Dixon M.
Biochemical Journal,
1953