Abstract
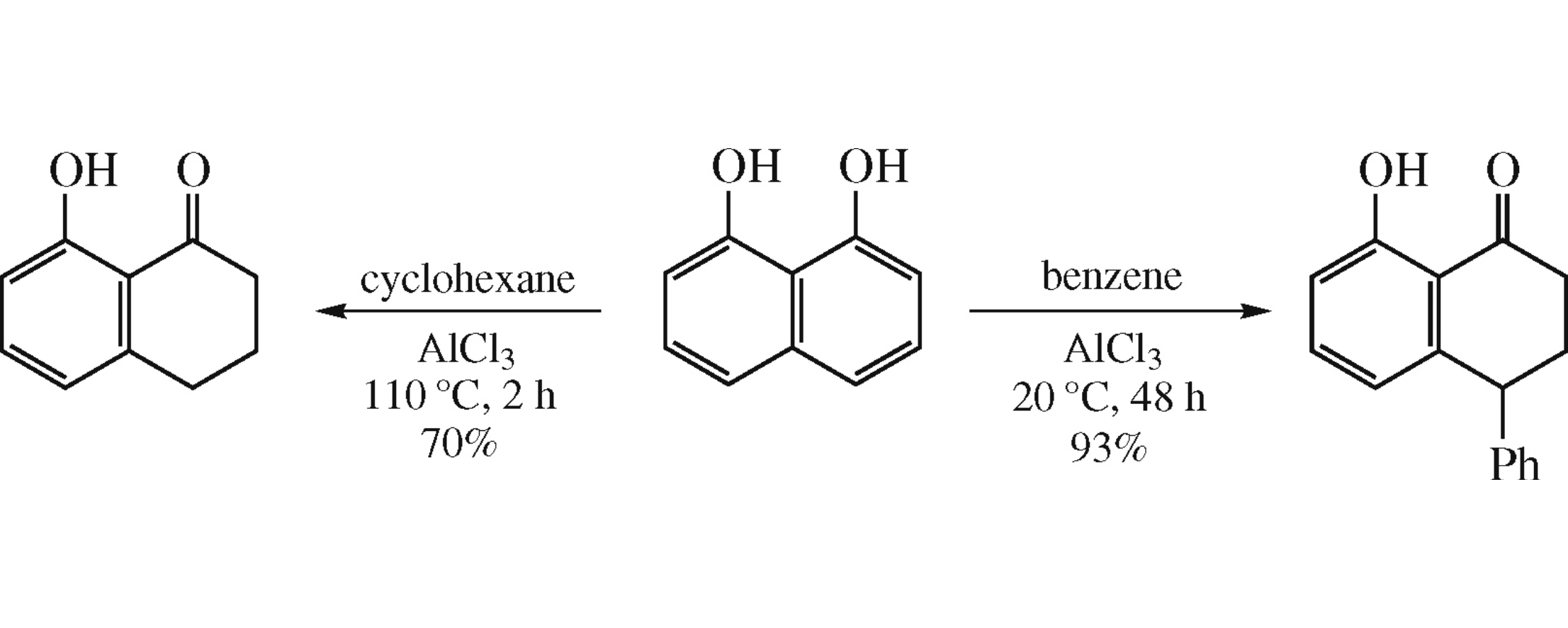
Naphthalene-1,8-diol on superelectrophilic activation with aluminium halides smoothly reacts with cyclohexane and benzene to afford 8-hydroxy-1-tetralone and 8-hydroxy-4-phenyl-1-tetralone, respectively.
References
1.
10.1016/j.mencom.2016.01.031_sbref0005a
Kaye
J. Chem. Soc.,
1964
2.

Yang R., Kizer D., Wu H., Volckova E., Miao X., Ali S.M., Tandon M., Savage R.E., Chan T.C., Ashwell M.A.
Bioorganic and Medicinal Chemistry,
2008
3.

Cabrera E.V., Banerjee A.K.
Organic Preparations and Procedures International,
2010
4.

Cai Y., Guo Y., Krohn K.
Natural Product Reports,
2010
5.

Couché E., Fkyerat A., Tabacchi R.
Helvetica Chimica Acta,
2003
6.
10.1016/j.mencom.2016.01.031_bib0010
Olah
Superelectrophiles and Their Chemistry,
2008
7.
10.1016/j.mencom.2016.01.031_sbref0015a
Repinskaya
Russ. J. Org. Chem.,
1993
8.
10.1016/j.mencom.2016.01.031_sbref0015b
Koltunov
Russ. J. Org. Chem.,
1994
9.
10.1016/j.mencom.2016.01.031_sbref0015c
Koltunov
Russ. J. Org. Chem.,
1997
10.
10.1016/j.mencom.2016.01.031_sbref0015d
Koltunov
Russ. J. Org. Chem.,
1998
11.

12.

Koltunov K.Y., Chernov A.N., Surya Prakash G.K., Olah G.A.
Chemical and Pharmaceutical Bulletin,
2012
13.
Koltunov K.Y., Chernov A.N., Ostashevskaya L.A., Gribov P.A.
Mendeleev Communications,
2014
14.

Zhu Z., Ostashevskaya L.A., Koltunov K.Y.
Tetrahedron Letters,
2015
15.
10.1016/j.mencom.2016.01.031_bib0020
Repinskaya
J. Org. Chem. USSR,
1988
16.

Koltunov K.Y., Prakash G.K., Rasul G., Olah G.A.
Journal of Organic Chemistry,
2002