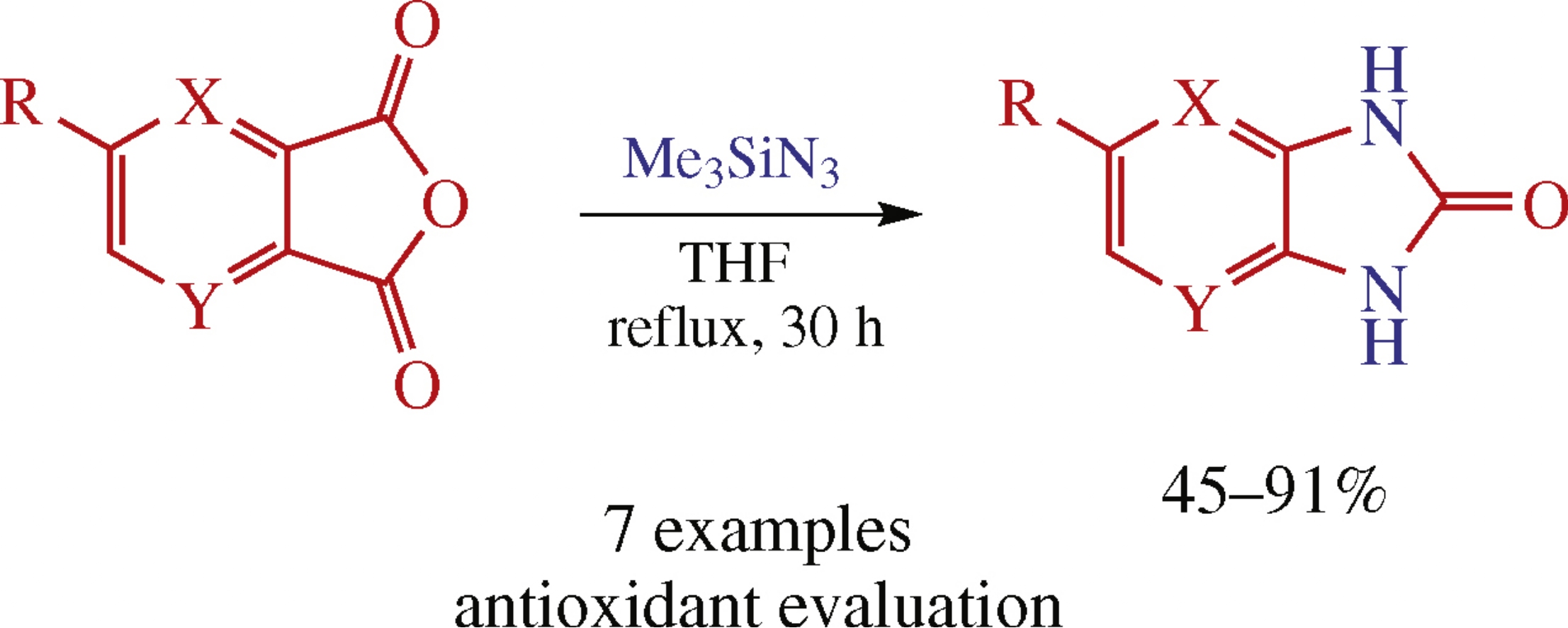
Abstract
Treatment of phthalic anhydride derivatives with trimethylsilyl azide affords benzimidazolin-2-ones in 45–91% yield, which is the result of two consecutive Curtius reactions. Within the series obtained, 1,3-dihydro-2H-imidazo[4,5-b]pyridin-2-one showed highest antioxidant activity.
References
1.

Lee S., Zhang Y.J., Song C.E., Lee J.K., Choi J.H.
Angewandte Chemie - International Edition,
2002
2.

Lee S., Yoon J., Chung S., Lee Y.
Tetrahedron,
2001
3.

Verniest G., Padwa A.
Organic Letters,
2008
4.

Rendler S., MacMillan D.W.
Journal of the American Chemical Society,
2010
5.

Rinnová M., Nefzi A., Houghten R.A.
Tetrahedron Letters,
2002
6.
Komogortsev A.N., Lichitsky B.V., Krylov K.S., Dudinov A.A., Purygin P.P., Krayushkin M.M.
Mendeleev Communications,
2014
7.
Gazieva G.A., Poluboyarov P.A., Kolotyrkina N.G., Lubuzh E.D., Kravchenko A.N.
Mendeleev Communications,
2014
8.

Kim T.H., Lee G.
Journal of Organic Chemistry,
1999
9.

Guirado A., Andreu R., Martiz B., Bautista D., Ramírez de Arellano C., Jones P.G.
Tetrahedron,
2006
10.

DasGupta S., Murumkar P.R., Giridhar R., Yadav M.R.
Bioorganic and Medicinal Chemistry,
2009
11.

Bautista R., Bernal P., Herrera R., Santoyo B.M., Lazcano-Seres J.M., Delgado F., Tamariz J.
Journal of Organic Chemistry,
2011
12.

Dong C., Alper H.
Tetrahedron Asymmetry,
2004
13.

Zhang K., Chopade P.R., Louie J.
Tetrahedron Letters,
2008
14.

Fritz J.A., Wolfe J.P.
Tetrahedron,
2008
15.

De Clercq P.J.
Chemical Reviews,
1997
16.

Cumming J.N., Le T.X., Babu S., Carroll C., Chen X., Favreau L., Gaspari P., Guo T., Hobbs D.W., Huang Y., Iserloh U., Kennedy M.E., Kuvelkar R., Li G., Lowrie J., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
17.

Hutchinson J.H., Halczenko W., Brashear K.M., Breslin M.J., Coleman P.J., Duong L.T., Fernandez-Metzler C., Gentile M.A., Fisher J.E., Hartman G.D., Huff J.R., Kimmel D.B., Leu C., Meissner R.S., Merkle K., et. al.
Journal of Medicinal Chemistry,
2003
18.

Catto B.A., Tracy J.W., Webster L.T.
Molecular and Biochemical Parasitology,
1984
19.

Shia K., Li W., Chang C., Hsu M., Chern J., Leong M.K., Tseng S., Lee C., Lee Y., Chen S., Peng K., Tseng H., Chang Y., Tai C., Shih S., et. al.
Journal of Medicinal Chemistry,
2002
20.

Peretto I., Fossati C., Giardina G.A., Giardini A., Guala M., La Porta E., Petrillo P., Radaelli S., Radice L., Raveglia L.F., Santoro E., Scudellaro R., Scarpitta F., Cerri A., Menegon S., et. al.
Journal of Medicinal Chemistry,
2007
21.

Peretto I., Forlani R., Fossati C., Giardina G.A., Giardini A., Guala M., La Porta E., Petrillo P., Radaelli S., Radice L., Raveglia L.F., Santoro E., Scudellaro R., Scarpitta F., Bigogno C., et. al.
Journal of Medicinal Chemistry,
2007
22.

Peretto I., Petrillo P., Imbimbo B.P.
Medicinal Research Reviews,
2009
23.

Moussa Z., El-Sharief M.A., El-Sharief A.M.
European Journal of Medicinal Chemistry,
2011
24.

Robert J.H., Sabourin C., Alvarez N., Robert-Piessard S., Le Baut G., Le Pape P.
European Journal of Medicinal Chemistry,
2003
25.

Alvarez N., Robledo S., Velez I.D., Robert J.M., Baut G.L., Pape P.L.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2002
26.
10.1016/j.mencom.2016.01.027_sbref0075c
Abdala
Arzneim. -Forsch.,
2000
27.
10.1016/j.mencom.2016.01.027_bib0080
Sarmiento-Sánchez
Quim. Nova,
2014
28.

Snyder D.C.
Journal of Organic Chemistry,
1995
29.

Jones R.C., Schofield J.
Journal of the Chemical Society Perkin Transactions 1,
1990
30.
10.1016/j.mencom.2016.01.027_sbref0095a
Maffei
Ann. Chim.,
1959
31.
10.1016/j.mencom.2016.01.027_sbref0095b
Caronna
Gazz. Chim. Ital.,
1941
32.

Wiss J., Fleury C., Heuberger C., Onken U., Glor M.
Organic Process Research and Development,
2007
33.

Estimation of antiradical properties of antioxidants using DPPH assay: A critical review and results
Mishra K., Ojha H., Chaudhury N.K.
Food Chemistry,
2012