Abstract
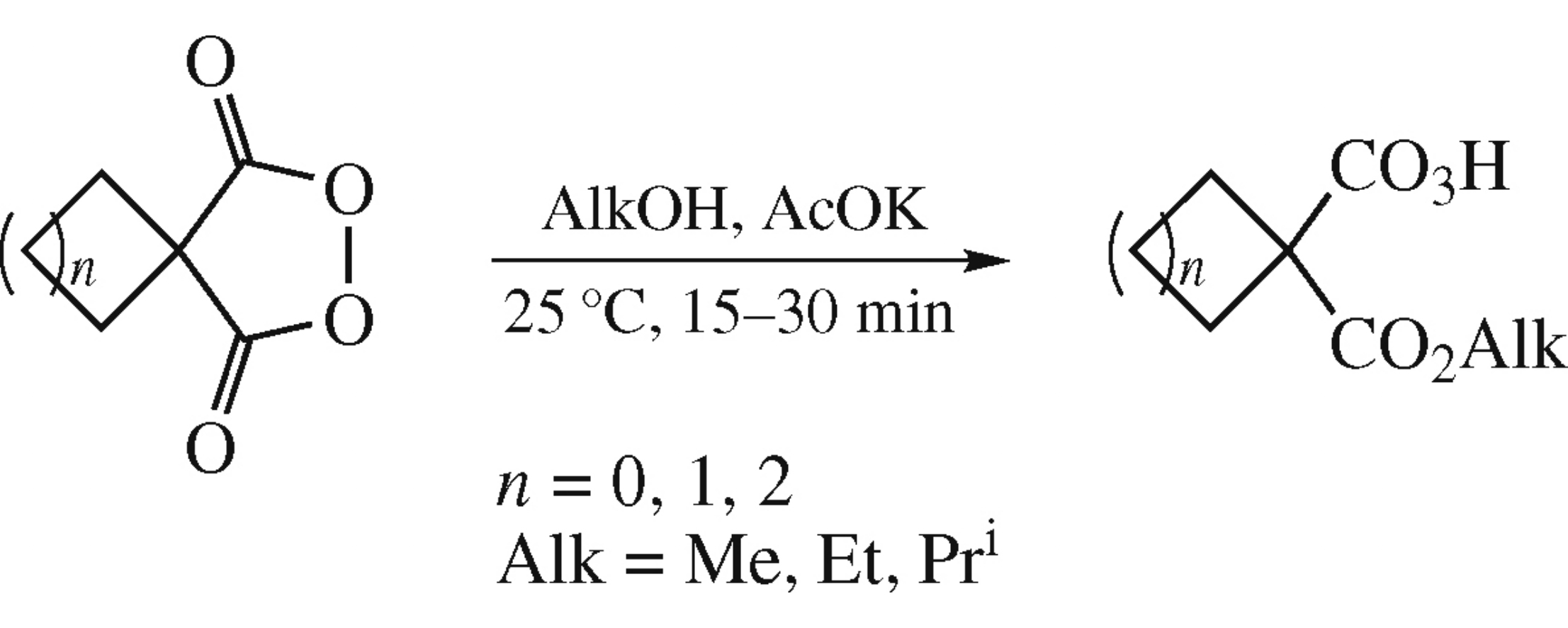
Potassium acetate catalyzed alcoholysis of spirocycloalkyl malonyl peroxides affords 1-alkoxycarbonylcycloalkane-1-percarboxylic acids which are suitable for epoxidation of olefins.
References
1.

Griffith J.C., Jones K.M., Picon S., Rawling M.J., Kariuki B.M., Campbell M., Tomkinson N.C.
Journal of the American Chemical Society,
2010
2.

Schwarz M., Reiser O.
Angewandte Chemie - International Edition,
2011
3.

Picon S., Rawling M., Campbell M., Tomkinson N.C.
Organic Letters,
2012
4.

Jones K.M., Tomkinson N.C.
Journal of Organic Chemistry,
2012
5.

Rawling M.J., Tomkinson N.C.
Organic and Biomolecular Chemistry,
2013
6.

Rawling M.J., Rowley J.H., Campbell M., Kennedy A.R., Parkinson J.A., Tomkinson N.C.
Chemical Science,
2014
7.
Terent’ev A.O., Vil’ V.A., Mulina O.M., Pivnitsky K.K., Nikishin G.I.
Mendeleev Communications,
2014
8.

Adam W., Rucktaeschel R.
Journal of the American Chemical Society,
1971
9.

Chapman O.L., Wojtkowski P.W., Adam W., Rodriguez O., Rucktaeschel R.
Journal of the American Chemical Society,
1972
10.

Adam W., Liu J., Rodriguez O.
Journal of Organic Chemistry,
1973
11.

Adam W., Diehl J.W.
Journal of the Chemical Society Chemical Communications,
1972
12.

Terent’ev A., Vil’ V., Nikishin G., Adam W.
Synlett,
2015
13.

Adam W., Rucktaeschel R.
Journal of Organic Chemistry,
1972
14.

Photolysis and thermolysis of di-n-butylmalonyl peroxide. Evidence for .alpha.-lactone intermediates
Adam W., Rucktaeschel R.
Journal of Organic Chemistry,
1978
15.
K. M. Jones, PhD Thesis, Cardiff University, 2010, p. 129.
16.
K. M. Jones, PhD Thesis, Cardiff University, 2010, p. 51.
17.
![Reactions of γδ-unsaturated ketones with peracids. 2,7-Dioxabicyclo-[2,2,1]heptanes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Gaoni Y.
Journal of the Chemical Society C Organic,
1968
18.

Anderson W.K., Veysoglu T.
Journal of Organic Chemistry,
1973
19.

Rebek J., McCready R., Wolf S., Mossman A.
Journal of Organic Chemistry,
1979
20.

Ceruti M., Balliano G., Viola F., Grosa G., Rocco F., Cattel L.
Journal of Medicinal Chemistry,
1992