Abstract
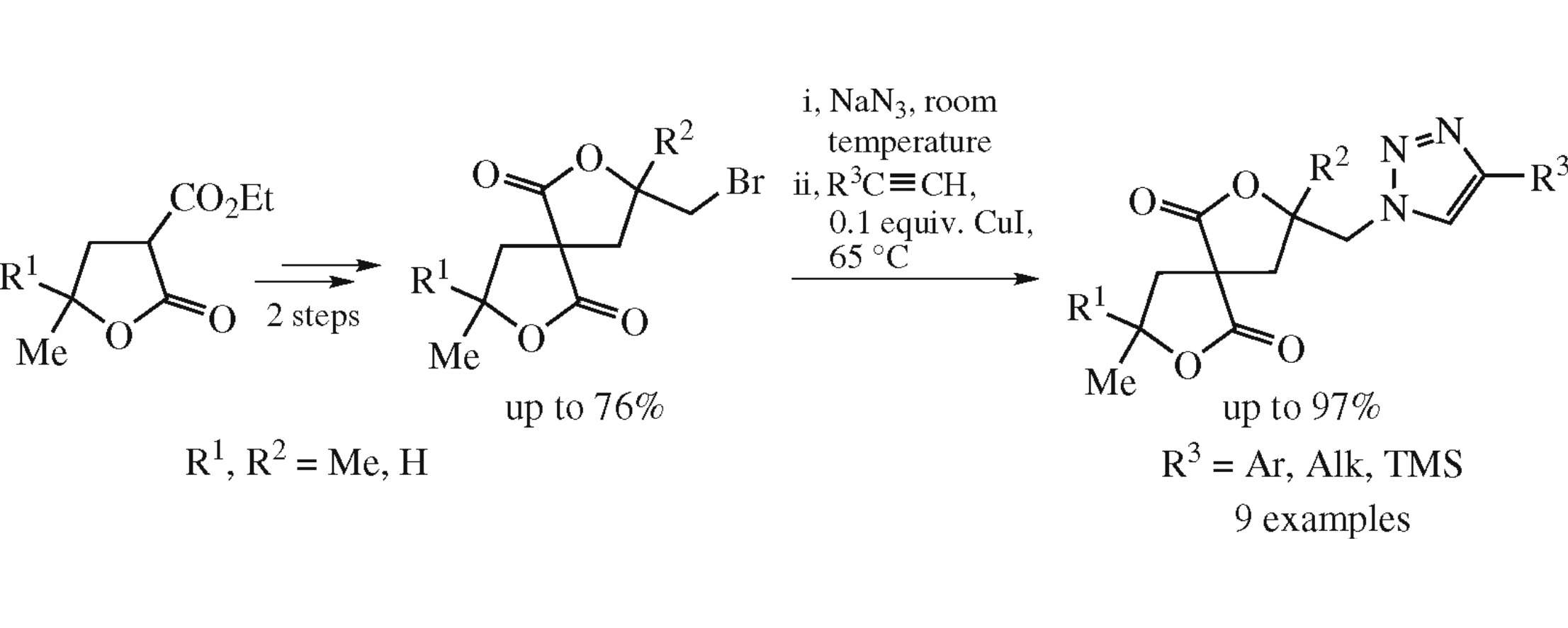
3-Bromomethyl-2,8-dioxaspiro[4.4]nonane-1,6-diones were efficiently prepared from 3-ethoxycarbonyltetrahydrofuran-2-ones. Subsequent conversion of the bromomethyl group into azidomethyl one followed by click reaction with alkynes afforded the multifunctional triazole-containing spiro dilactones in almost quantitative yields.
References
1.

Horton D.A., Bourne G.T., Smythe M.L.
Chemical Reviews,
2003
2.

Welsch M.E., Snyder S.A., Stockwell B.R.
Current Opinion in Chemical Biology,
2010
3.

Rios R.
Chemical Society Reviews,
2012
4.
![Construction of Spiro[pyrrolidine‐3,3′‐oxindoles] − Recent Applications to the Synthesis of Oxindole Alkaloids](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Marti C., Carreira E.
European Journal of Organic Chemistry,
2003
5.

Hong L., Wang R.
Advanced Synthesis and Catalysis,
2013
6.

Galliford C., Scheidt K.
Angewandte Chemie - International Edition,
2007
7.

Wuts P., Anderson A., Ashford S., Goble M., White M., Beck D., Gilbert I., Hrab R.
Synlett,
2008
8.
10.1016/j.mencom.2016.01.005_sbref0015b
Xu
Synlett,
2010
9.
10.1016/j.mencom.2016.01.005_sbref0015c
Hejmanowska
Synlett,
2014
10.
Khafizova L.O., Gubaidullin R.R., Popod’ko N.R., Meshcheryakova E.S., Khalilov L.M., Dzhemilev U.M.
Mendeleev Communications,
2014
11.
Shabalin D.A., Dvorko M.Y., Schmidt E.Y., Protsuk N.I., Ushakov I.A., Mikhaleva A.I., Trofimov B.A.
Mendeleev Communications,
2015
12.
Laptev A.V., Lukin A.Y., Belikov N.E., Demina O.V., Khodonov A.A., Shvets V.I.
Mendeleev Communications,
2014
13.
![Syntheses based on 8-substituted 3-bromoacetyl-3,8-dimethyl-2,7-dioxaspiro[4,4]nonane-1,6-diones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kochikyan T.V., Harutyunyan E.V., Harutyunyan V.S., Avetisyan A.A.
Chemistry of Heterocyclic Compounds,
2006
14.

Kochikyan T.V., Samvelyan M.A., Arutyunyan V.S., Avetisyan A.A.
Russian Journal of Organic Chemistry,
2005
15.
10.1016/j.mencom.2016.01.005_sbref0020c
Harutyunyan
Arm. Khim. Zh.,
2000
16.
10.1016/j.mencom.2016.01.005_sbref0020d
Harutyunyan
Arm. Khim. Zh.,
1994
17.

Rostovtsev V.V., Green L.G., Fokin V.V., Sharpless K.B.
Angewandte Chemie - International Edition,
2002
18.
![Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tornøe C.W., Christensen C., Meldal M.
Journal of Organic Chemistry,
2002
19.

Sokolova N.V., Nenajdenko V.G.
RSC Advances,
2013
20.

Nenajdenko V.G., Gulevich A.V., Sokolova N.V., Mironov A.V., Balenkova E.S.
European Journal of Organic Chemistry,
2010
21.

Sokolova N.V., Latyshev G.V., Lukashev N.V., Nenajdenko V.G.
Organic and Biomolecular Chemistry,
2011
22.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
23.
Govdi A.I., Vasilevsky S.F., Sokolova N.V., Sorokina I.V., Tolstikova T.G., Nenajdenko V.G.
Mendeleev Communications,
2013
24.

Govdi A.I., Sokolova N.V., Sorokina I.V., Baev D.S., Tolstikova T.G., Mamatyuk V.I., Fadeev D.S., Vasilevsky S.F., Nenajdenko V.G.
MedChemComm,
2015
25.

Vasilevsky S.F., Govdi A.I., Sorokina I.V., Tolstikova T.G., Baev D.S., Tolstikov G.A., Mamatuyk V.I., Alabugin I.V.
Bioorganic and Medicinal Chemistry Letters,
2011
26.

Baldwin J.E.
Journal of the Chemical Society Chemical Communications,
1976
27.

Alabugin I.V., Gilmore K.
Chemical Communications,
2013
28.

Houk K.N., Jabbari A., Hall H.K., Alemán C.
Journal of Organic Chemistry,
2008
29.

Baroudi A., Mauldin J., Alabugin I.V.
Journal of the American Chemical Society,
2009