Abstract
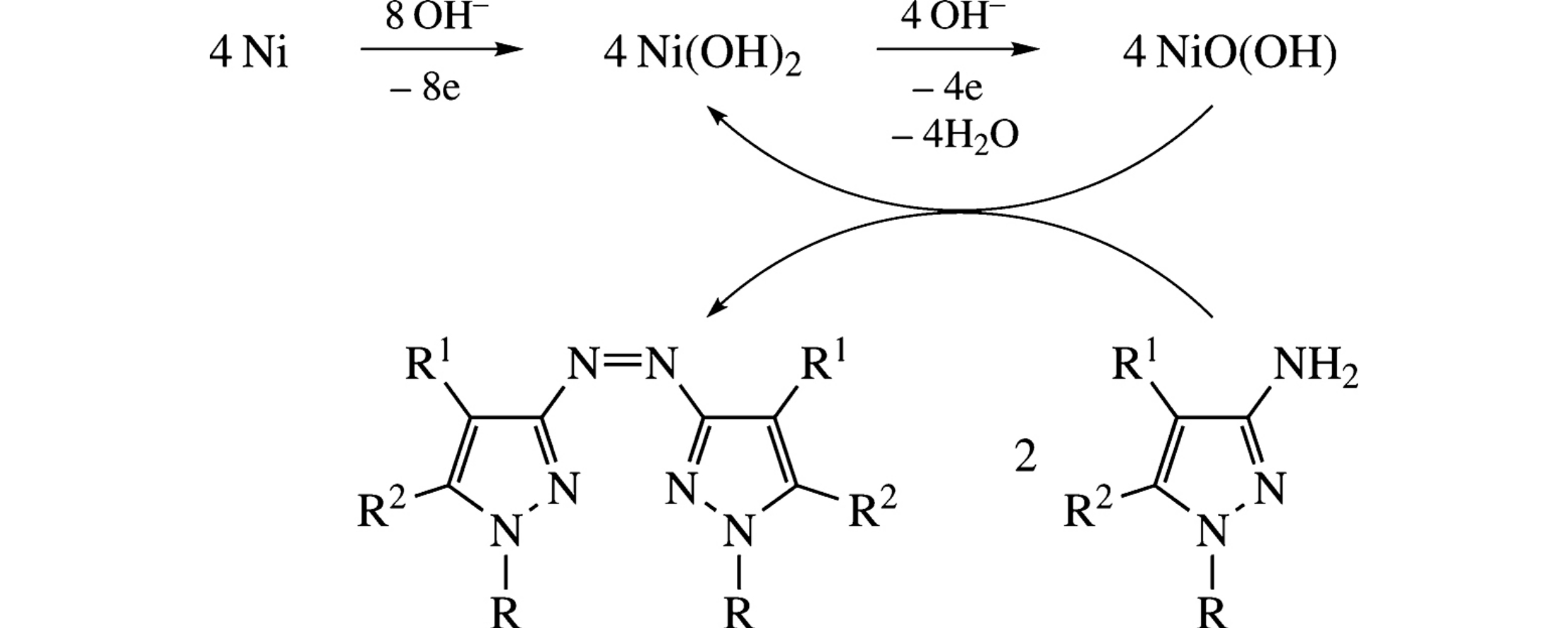
Oxidation of C-amino-N-alkylpyrazoles on a NiO(OH) electrode in an aqueous alkaline medium affords the corresponding azopyrazoles. The success in implementation of these processes is due to the structure of C-aminopyrazoles.
References
1.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
2.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
3.
10.1016/j.mencom.2015.11.028_bib0015
Hunger
Industrial Dyes: Chemistry, Properties, Applications,
2003
4.

Carlucci L., Ciani G., Proserpio D.M., Rizzato S.
New Journal of Chemistry,
2003
5.

Iranpoor N., Firouzabadi H., Khalili D., Motevalli S.
Journal of Organic Chemistry,
2008
6.

Iranpoor N., Firouzabadi H., Khalili D.
Organic and Biomolecular Chemistry,
2010
7.

Iranpoor N., Firouzabadi H., Shahin R., Khalili D.
Synthetic Communications,
2012
8.

Baumgarten H.E., Staklis A., Miller E.M.
Journal of Organic Chemistry,
1965
9.

Firouzabadi H., Mostafavipoor Z.
Bulletin of the Chemical Society of Japan,
1983
10.
10.1016/j.mencom.2015.11.028_bib0050
Birchall
J. Chem. Soc., Chem. Commun.,
1970
11.

Wenkert E., Wickberg B.
Journal of the American Chemical Society,
1962
12.
10.1016/j.mencom.2015.11.028_bib0060
Farbagi
Acta Chim. Slov.,
2007
13.

Wawzonek S., McIntyre T.W.
Journal of the Electrochemical Society,
2007
14.

Wawzonek S., McIntyre T.W.
Journal of the Electrochemical Society,
2007
15.

Schäfer H.
Topics in Current Chemistry,
1987
16.

Lyalin B.V., Petrosyan V.A.
Russian Journal of Electrochemistry,
2010
17.
10.1016/j.mencom.2015.11.028_bib0085
Sheldon
Green Chemistry and Catalysis,
2007
18.
10.1016/j.mencom.2015.11.028_bib0090
Lyalin
Russ. J. Electrochem.,
1996
19.

Lyalin B.V., Petrosyan V.A.
Russian Chemical Bulletin,
2004
20.

Lyalin B.V., Petrosyan V.A.
Russian Chemical Bulletin,
2009
21.

Lyalin B.V., Petrosyan V.A.
Russian Chemical Bulletin,
2007
22.

Lyalin B.V., Petrosyan V.A.
Russian Chemical Bulletin,
2012
23.
10.1016/j.mencom.2015.11.028_bib0115
Lyalin
in Proceedings of III All-Russian Scientific Conference ‘Advances in Synthesis and Complexing’,
2014
24.
10.1016/j.mencom.2015.11.028_bib0120
Sheremetev
RCS Adv.,
2015
25.

Salaheldin A.M., Oliveira-Campos A.M., Rodrigues L.M.
Tetrahedron Letters,
2007
26.

Okumura S., Lin C., Takeda Y., Minakata S.
Journal of Organic Chemistry,
2013
27.

da Silva D.B., Samadi A., Infantes L., Carreiras M.D., Marco-Contelles J.
Tetrahedron Letters,
2010
28.

Jiang B., Ning Y., Fan W., Tu S., Li G.
Journal of Organic Chemistry,
2014
29.
10.1016/j.mencom.2015.11.028_bib0145
Banerjee
Indian J. Chem., Sect. A,
1983
30.

Perevalov V.P., Baryshnenkova L.I., Andreeva M.A., Manaev Y.A., Denisova I.A., Stepanov B.I., Seraya V.I.
Chemistry of Heterocyclic Compounds,
1983
31.

Tret'yakov E.V., Vasitevsky S.F.
Russian Chemical Bulletin,
1996
32.
J. Craig, Mc. K. Darren and P. K. Gordon, Patent WO 2005/80359 A1, 2005.
33.
X. Dengming, C. Liang, L. Xijie, H. Yuandong, H. Xinhe, L. Zhihua, Z. Lipeng, W. Wei, W. Shylong, S. Yu, L. Gen, W. Yin, Z. Sheng, L. Chonglong, T. Jia and Y. Honghao, Patent WO 2012/92880 A1, 2012.
34.

ADACHI I., YAMAMORI T., HIRAMATSU Y., SAKAI K., SATO H., KAWAKAMI M., UNO O., UEDA M.
Chemical and Pharmaceutical Bulletin,
2011
35.

Zheng K., Iqbal S., Hernandez P., Park H., LoGrasso P.V., Feng Y.
Journal of Medicinal Chemistry,
2014
36.

Kaulen J., Schäfer H.
Tetrahedron,
1982