Abstract
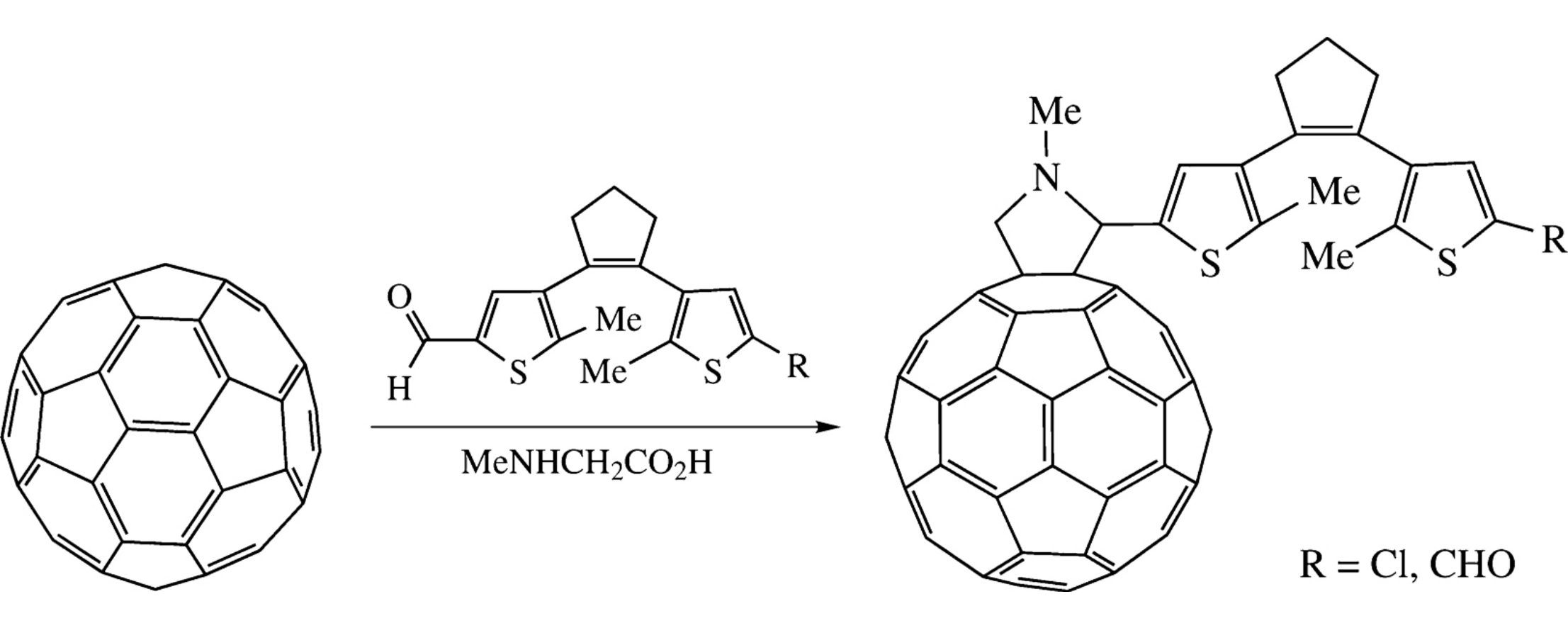
Dithienylethene-containing fulleropyrrolidines were synthesized by 1,3-dipolar cycloaddition of in situ generated appropriately derivatized azomethine ylides to C60. The photochromic properties of these cycloadducts were predicted resorting to DFT calculations.
References
1.

Light-controlled molecular switches based on bistable spirocyclic organic and coordination compounds
Minkin V.I.
Russian Chemical Reviews,
2013
2.

3.

Shirinian V.Z., Lonshakov D.V., Lvov A.G., Krayushkin M.M.
Russian Chemical Reviews,
2013
4.
Korshin D.E., Nastapova N.V., Kharlamov S.V., Nasybullina G.R., Sergeeva T.Y., Krasnova E.G., Sultanova E.D., Mukhitova R.K., Latypov S.K., Yanilkin V.V., Ziganshina A.Y., Konovalov A.I.
Mendeleev Communications,
2013
5.
Lonshakov D.V., Shirinian V.Z., Lvov A.G., Krayushkin M.M.
Mendeleev Communications,
2013
6.
Mumyatov A.V., Leshanskaya L.I., Anokhin D.V., Dremova N.N., Troshin P.A.
Mendeleev Communications,
2014
7.

Kroto H.W., Heath J.R., O’Brien S.C., Curl R.F., Smalley R.E.
Nature,
1985
8.

Liddell P.A., Kodis G., Andréasson J., de la Garza L., Bandyopadhyay S., Mitchell R.H., Moore T.A., Moore A.L., Gust D.
Journal of the American Chemical Society,
2004
9.

Straight S.D., Andréasson J., Kodis G., Moore A.L., Moore T.A., Gust D.
Journal of the American Chemical Society,
2005
10.

El-Khouly M., Kim J., Kay K., Choi C., Ito O., Fukuzumi S.
Chemistry - A European Journal,
2009
11.
A. R. Tuktarov, A. R. Akhmetov and U. M. Dzhemilev, In: S. B. Ellis (ed.), Fullerene: Chemistry, Natural Sources and Technological Applications, Nova Publishers, New York, 2014, pp. 123-166.
12.

Jeong J., Yun E., Choi Y., Jung H., Chung S.J., Song N.W., Chung B.H.
Chemical Communications,
2011
13.

Frey J., Kodis G., Straight S.D., Moore T.A., Moore A.L., Gust D.
Journal of Physical Chemistry A,
2013
14.

Castellanos S., Vieira A.A., Illescas B.M., Sacchetti V., Schubert C., Moreno J., Guldi D.M., Hecht S., Martín N.
Angewandte Chemie - International Edition,
2013
15.

Prato M., Maggini M., Giacometti C., Scorrano G., Sandona G., Farnia G.
Tetrahedron,
1996
16.

Lucas L., Jong J. ., Esch J. ., Kellogg R., Feringa B.
European Journal of Organic Chemistry,
2003
17.

Da Ros T., Prato M., Novello F., Maggini M., Banfi E.
Journal of Organic Chemistry,
1996
18.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
19.

Laikov D.N.
Chemical Physics Letters,
1997
20.
10.1016/j.mencom.2015.11.025_bib0100
Laikov
Russ. Chem. Bull., Int.,
2005
21.
D. N. Laikov, PhD Thesis, Moscow State University, 2000
22.

Kellogg R.M., Groen M.B., Wynberg H.
Journal of Organic Chemistry,
1967
23.

Irie M., Sakemura K., Okinaka M., Uchida K.
Journal of Organic Chemistry,
1995
24.
![Thermally Irreversible Photochromic Systems. Reversible Photocyclization of 1,2-Bis(benzo[b]thiophen-3-yl)ethene Derivatives](/storage/images/resized/yNSijlgQghQF53VZuyFLA30CKDe4j3HK74Vtpnxa_small_thumb.webp)
Uchida K., Nakayama Y., Irie M.
Bulletin of the Chemical Society of Japan,
1990
25.

Ramamurthy V., Venkatesan K.
Chemical Reviews,
1987
26.
10.1016/j.mencom.2015.11.025_bib0130
Frisch
Gaussian 09, Revision D.01,
2009
27.

Hu F., Hu M., Liu W., Yin J., Yu G., Liu S.H.
Tetrahedron Letters,
2015