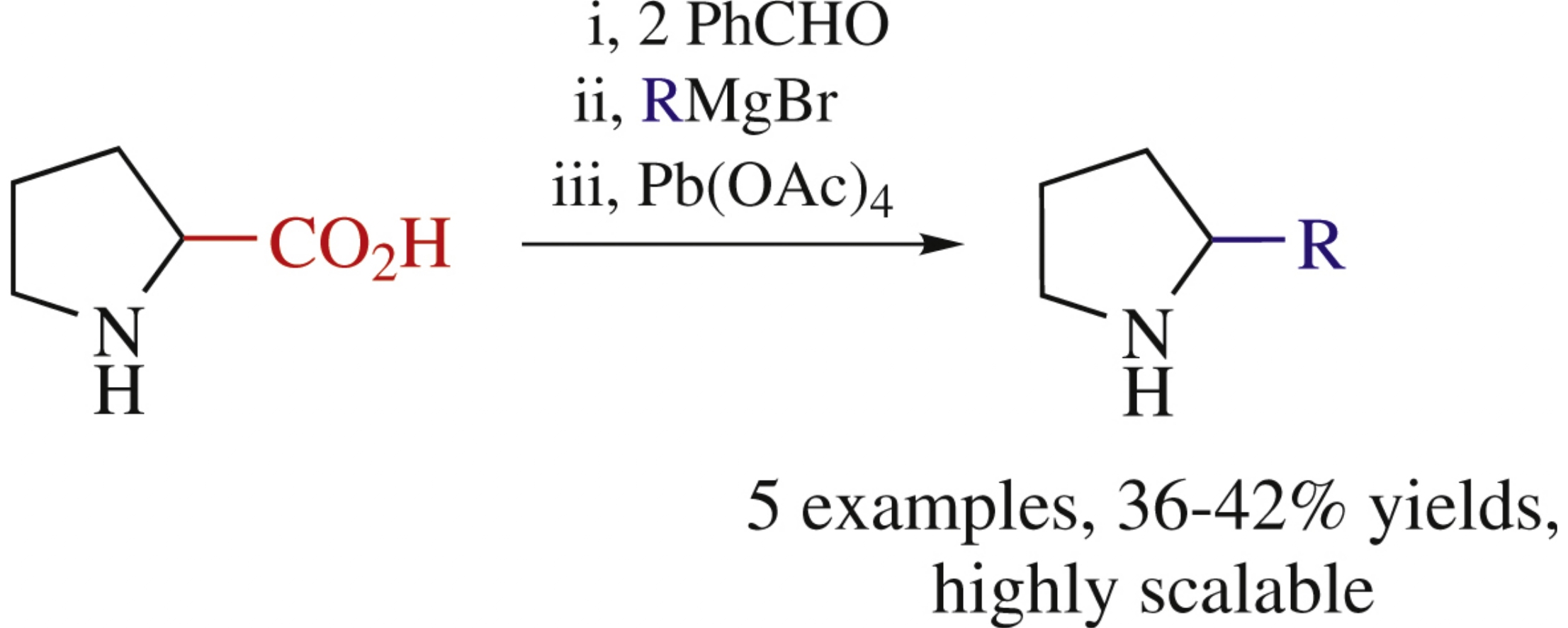
Abstract
Diphenyloxapyrrolizidines, products of the reaction between proline and benzaldehyde, are convenient building blocks for synthesizing 2-substituted pyrrolidines. The opening of their oxazolidine ring by treatment with Grignard reagents has been performed and conditions for subsequent removal of the N-hydroxyethyl moiety have been found. Though the yields are moderate (36–42%), the suggested synthesis of 2-alkyl(aryl)pyrrolidines is rather simple since it does not require purification of intermediate products and is easy scalable.
References
1.

Plunkett A.O.
Natural Product Reports,
1994
2.
![Construction of Enantiopure Pyrrolidine Ring System via Asymmetric [3+2]-Cycloaddition of Azomethine Ylides](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Pandey G., Banerjee P., Gadre S.R.
Chemical Reviews,
2006
3.

Mitchenson A., Nadin A.
Journal of the Chemical Society Perkin Transactions 1,
2000
4.
Tobacco Alkaloids and Related Compounds, ed. U. S. von Euler, Pergamon, New York, 1965.
5.

Shevchenko N.E., Vlasov K., Nenajdenko V.G., Röschenthaler G.
Tetrahedron,
2011
6.

Craig L.C., Bulbrook H., Hixon R.M.
Journal of the American Chemical Society,
1931
7.

BURCKHALTER J.H., SHORT J.H.
Journal of Organic Chemistry,
1958
8.

9.

Dunsmore C.J., Carr R., Fleming T., Turner N.J.
Journal of the American Chemical Society,
2006
10.

SEEMAN J.I.
Synthesis,
1977
11.

Hua D.H., Miao S.W., Bharathi S.N., Katsuhira T., Bravo A.A.
Journal of Organic Chemistry,
1990
12.

Willoughby C.A., Buchwald S.L.
Journal of Organic Chemistry,
1993
13.

Burgess L.E., Meyers A.I.
Journal of Organic Chemistry,
1992
14.

Zhang C., Seidel D.
Journal of the American Chemical Society,
2010
15.

Das D., Richers M.T., Ma L., Seidel D.
Organic Letters,
2011
16.

Ma L., Chen W., Seidel D.
Journal of the American Chemical Society,
2012
17.

Das D., Sun A.X., Seidel D.
Angewandte Chemie - International Edition,
2013
18.

Chen W., Wilde R.G., Seidel D.
Organic Letters,
2013
19.

Seidel D.
Accounts of Chemical Research,
2015
20.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
21.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2013
22.

Moshkin V.S., Sosnovskikh V.Y.
Tetrahedron Letters,
2014
23.
![3-Cyanochromones in [3+2] cycloadditions with an azomethine ylide derived from sarcosine and formaldehyde. A short synthesis of 1-benzopyrano[2,3-c:3,4-c′]dipyrrolidines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sosnovskikh V.Y., Kornev M.Y., Moshkin V.S.
Tetrahedron Letters,
2014
24.
![Substituted chromones in [3+2] cycloadditions with nonstabilized azomethine ylides: synthesis of 1-benzopyrano[2,3-c]pyrrolidines and 1-benzopyrano[2,3-c:3,4-c′]dipyrrolidines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sosnovskikh V.Y., Kornev M.Y., Moshkin V.S., Buev E.M.
Tetrahedron,
2014
25.

Rizzi G.P.
Journal of Organic Chemistry,
1970
26.
![Use of N-[(trimethylsilyl)methyl]amino ethers as capped azomethine ylide equivalents](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Padwa A., Dent W.
Journal of Organic Chemistry,
1987
27.

Orsini F., Pelizzoni F., Forte M., Destro R., Gariboldi P.
Tetrahedron,
1988
28.

Nyerges M., Fejes I., Virányi A., Groundwater P., Töke L.
Synthesis,
2004
29.

Ryan J.H., Spiccia N., Wong L.S., Holmes A.B.
Australian Journal of Chemistry,
2007
30.
![Functionalization of an sp3 C–H bond via a redox-neutral domino reaction: diastereoselective synthesis of hexahydropyrrolo[2,1-b]oxazoles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Rahman M., Bagdi A.K., Mishra S., Hajra A.
Chemical Communications,
2014
31.

Guerrier L., Royer J., Grierson D.S., Husson H.P.
Journal of the American Chemical Society,
1983
32.

Cisneros A., Fernández S., Hernández J.E.
Synthetic Communications,
1982
33.

Marazano C., G始isson Y., Mehmandoust M., C. Das B.
Heterocycles,
1994
34.

Agami C., Couty F., Evano G.
Tetrahedron Letters,
1999
35.

Fujioka H., Hirose H., Ohba Y., Murai K., Nakahara K., Kita Y.
Tetrahedron,
2007
36.
![Stereocontrolled preparation of cis- and rans-2,6-dialkylpiperidines via diastereoselective reaction of 1-aza-4-oxabicyclo[4.3.0]nonane derivatives with Grignard reagents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Poerwono H., Higashiyama K., Yamauchi T., Kubo H., Ohmiya S., Takahashi H.
Tetrahedron,
1998
37.

Kobayashi T., Hasegawa F., Hirose Y., Tanaka K., Mori H., Katsumura S.
Journal of Organic Chemistry,
2012
38.

TAKAHASHI H., HSIEH B.C., HIGASHIYAMA K.
Chemical and Pharmaceutical Bulletin,
2011
39.
![Synthesis and Pyrolysis of Some Cycloalkano[a]pyrroles1](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Patterson J.M., Brasch J., Drenchko P.
Journal of Organic Chemistry,
1962
40.
Malmberg M., Nyberg K., Thøgersen H., Krogsgaard-Larsen P., Örn U.
Acta Chemica Scandinavica,
2008
41.

Starr D.F., Bulbrook H., Hixon R.M.
Journal of the American Chemical Society,
1932