Abstract
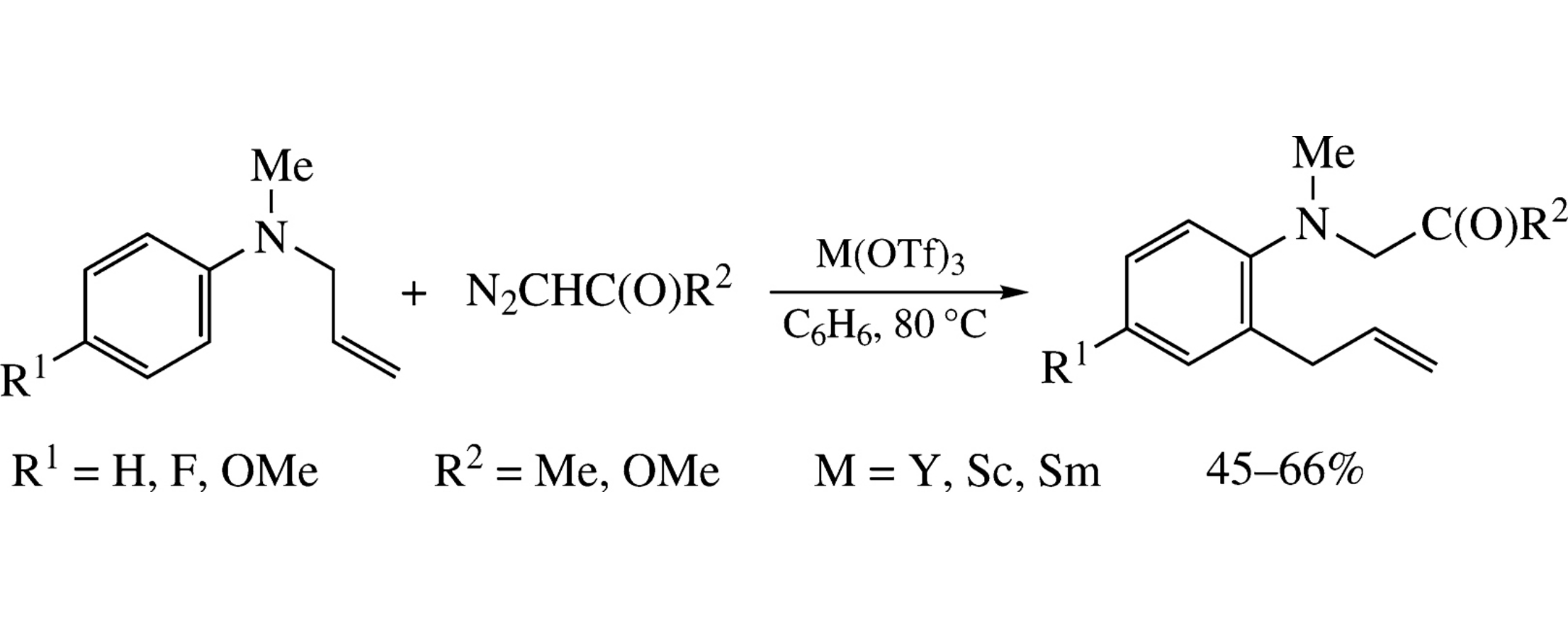
N-Allyl-N-methylanilines react with methyl diazoacetate or diazoacetone in the presence of Y, Sc or Sm triflates to give the corresponding methyl N-(2-allylaryl)-N-methylglycinates or 2-allyl-N-methyl-N-(2-oxopropyl)aniline. The formation of these compounds involves the aza-Claisen rearrangement.
References
1.

Maas G.
Topics in Current Chemistry,
1987
2.

Tomilov Y.V., Dokitchev V.A., Dzhemilev U.M., Nefedov O.M.
Russian Chemical Reviews,
1993
3.
O. M. Nefedov, E.A. Shapiro and A. B. Dyatkin, in Supplement B: The Chemistry of Acid Derivatives, ed. S. Patai, Wiley, New York, 1992, ch. 25
4.
10.1016/j.mencom.2015.11.013_bib0020
Zollinger
Diazo Chemistry II: Aliphatic, Inorganic and Organometallic Compounds,
1995
5.
10.1016/j.mencom.2015.11.013_bib0025
Doyle
Modern Catalytic Methods for Organic Synthesis with Diazo Compounds,
1998
6.
10.1016/j.mencom.2015.11.013_bib0030
Zhou
Synlett,
2010
7.

Aviv I., Gross Z.
Chemistry - A European Journal,
2008
8.
![Ruthenium Porphyrin Catalyzed Tandem Sulfonium/Ammonium Ylide Formation and [2,3]-Sigmatropic Rearrangement. A Concise Synthesis of (±)-Platynecine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhou C., Yu W., Chan P.W., Che C.
Journal of Organic Chemistry,
2004
9.

Novikov R.A., Platonov D.N., Dokichev V.A., Tomilov Y.V., Nefedov O.M.
Russian Chemical Bulletin,
2010
10.
10.1016/j.mencom.2015.11.013_sbref0050a
Nubbemeyer
2007
11.

Nubbemeyer U.
Topics in Current Chemistry,
2005
12.
10.1016/j.mencom.2015.11.013_sbref0050c
Sharma
J. Curr. Chem. Pharm. Sci.,
2013
13.
![A Simple and Practical Approach to the Dibenzo[c,f]thiazolo[3,2-a]azepines: A Novel Fused Tetracyclic Azepine System](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Palma A., Galeano N., Bahsas A.
Synthesis,
2010
14.

González I., Bellas I., Souto A., Rodríguez R., Cruces J.
Tetrahedron Letters,
2008
15.

Honda M., Morita H., Nagakura I.
Journal of Organic Chemistry,
1997
16.

O'Sullivan S., Doni E., Tuttle T., Murphy J.A.
Angewandte Chemie - International Edition,
2013
17.

Shakhbazyan L.G., Babakhanyan A.V., Grigoryan D.V., Kocharyan S.T.
Russian Journal of General Chemistry,
2003
18.

Zhang J., Jiang J., Li Y., Zhao Y., Wan X.
Organic Letters,
2013