Abstract
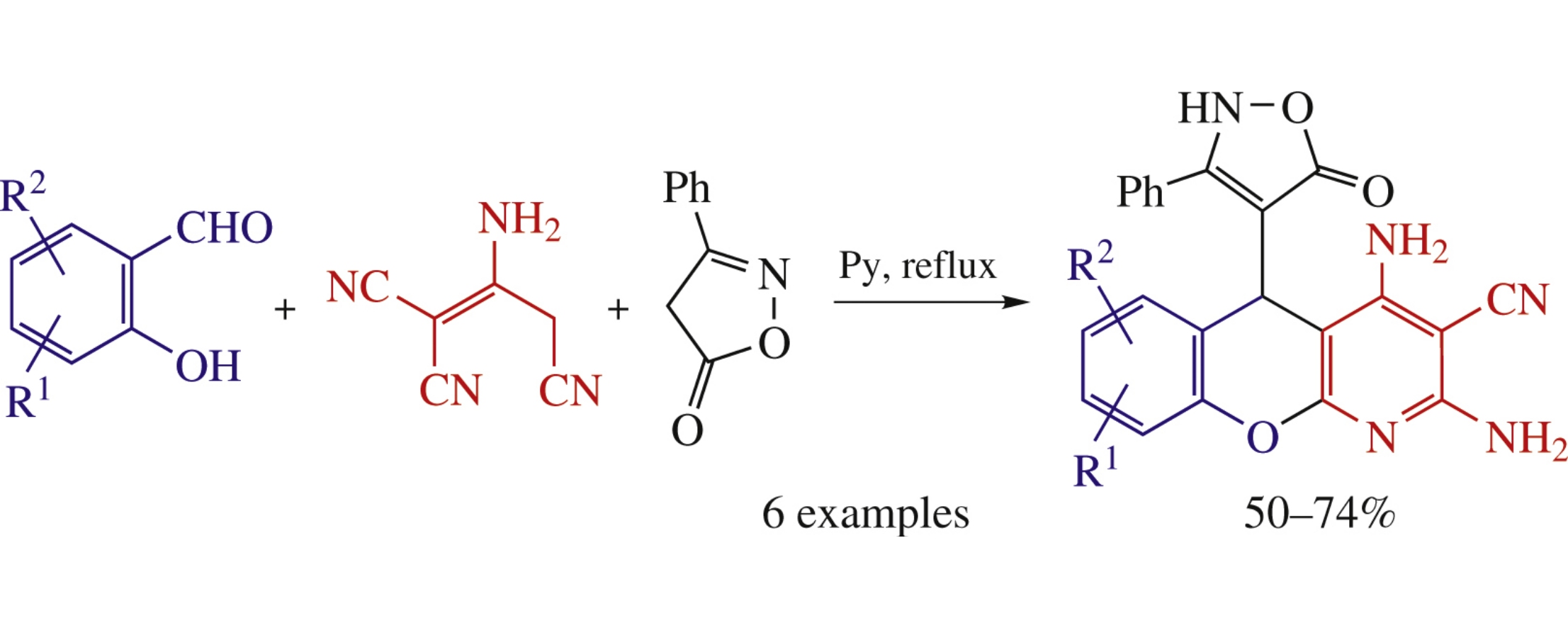
The new multicomponent reaction of salicylaldehydes, 2-aminoprop-1-ene-1,1,3-tricarbonitrile and 3-phenylisoxazol-5(4H)-one gives substituted 2,4-diamino-5-(5-oxo-3-aryl-2,5-dihydroisoxazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitriles (50–74% yields), which are promising for biomedical applications.
References
1.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
2.

DeSimone R., Currie K., Mitchell S., Darrow J., Pippin D.
Combinatorial Chemistry and High Throughput Screening,
2004
3.

Evans B.E., Rittle K.E., Bock M.G., DiPardo R.M., Freidinger R.M., Whitter W.L., Lundell G.F., Veber D.F., Anderson P.S., Chang R.S., Lotti V.J., Cerino D.J., Chen T.B., Kling P.J., Kunkel K.A., et. al.
Journal of Medicinal Chemistry,
1988
4.
10.1016/j.mencom.2015.11.008_bib0020
Johansen
PCT Int. Appl. WO 2008/33466,
2008
5.
10.1016/j.mencom.2015.11.008_bib0025
Zhao
PCT Int. Appl. WO 02/067764,
2002
6.
10.1016/j.mencom.2015.11.008_bib0030
Collard
PCT Int. Appl. WO2013/36403,
2013
7.
10.1016/j.mencom.2015.11.008_bib0035
Tomiyama
Pat. Appl. EP1767534,
2007
8.
10.1016/j.mencom.2015.11.008_bib0040
Cai
PCT Int. Appl. WO 2008/5572,
2008
9.
10.1016/j.mencom.2015.11.008_bib0045
Hunter
Pat. Appl. US 2007/208134,
2007
10.

Skommer J., Wlodkowic D., Mättö M., Eray M., Pelkonen J.
Leukemia Research,
2006
11.

Kasibhatla S., Gourdeau H., Meerovitch K., Drewe J., Reddy S., Qiu L., Zhang H., Bergeron F., Bouffard D., Yang Q., Herich J., Lamothe S., Cai S.X., Tseng B.
Molecular Cancer Therapeutics,
2004
12.

Gourdeau H., Leblond L., Hamelin B., Desputeau C., Dong K., Kianicka I., Custeau D., Boudreau C., Geerts L., Cai S., Drewe J., Labrecque D., Kasibhatla S., Tseng B.
Molecular Cancer Therapeutics,
2004
13.

Anderson D.R., Hegde S., Reinhard E., Gomez L., Vernier W.F., Lee L., Liu S., Sambandam A., Snider P.A., Masih L.
Bioorganic and Medicinal Chemistry Letters,
2005
14.
![Synthetic reactions of 2-(2-amino-3-cyano-4H-[1]benzopyran-4-yl)propane-1,3-dinitrile with reactive methylene compounds](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
O'Callaghan C.N., McMurry T.B., O'Brien J.E.
Journal of the Chemical Society Perkin Transactions 1,
1995
15.
![Chemical and electrocatalytic cascade cyclization of salicylaldehyde with three molecules of malononitrile: ‘one-pot’ simple and efficient way to the chromeno[2,3-b]pyridine scaffold](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Gorbunov S.V., Vereshchagin A.N., Nasybullin R.F., Goloveshkin A.S., Bushmarinov I.S., Egorov M.P.
Tetrahedron,
2014
16.
10.1016/j.mencom.2015.11.008_bib0080
Multicomponent Reactions
Wiley-VCH,
2005
18.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
19.

Evdokimov N.M., Kireev A.S., Yakovenko A.A., Antipin M.Y., Magedov I.V., Kornienko A.
Journal of Organic Chemistry,
2007
20.

Evdokimov N.M., Magedov I.V., Kireev A.S., Kornienko A.
Organic Letters,
2006
21.
![A new one-pot three-component synthesis of 2,4-diamino-5H-chromeno[2,3-b]pyridine-3-carbonitrile derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shaabani A., Hajishaabanha F., Mofakham H., Maleki A.
Molecular Diversity,
2009
22.

Bushmarinov I.S., Dmitrienko A.O., Korlyukov A.A., Antipin M.Y.
Journal of Applied Crystallography,
2012