Abstract
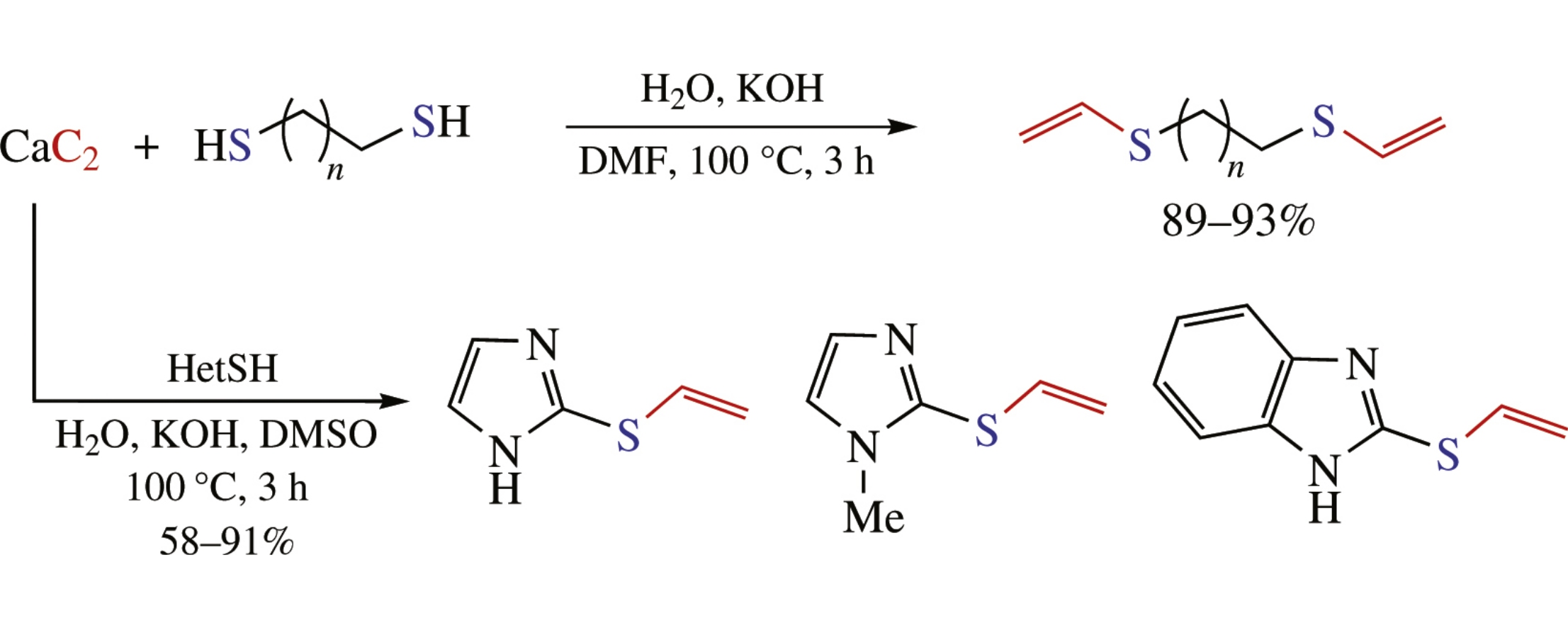
Calcium carbide was studied as a useful solid-state reagent to incorporate acetylene unit into synthetic procedures. Atom-economic thiol-yne click reaction was successfully performed with single and double additions. Heterocyclic thiols and aliphatic dithiols reacted with acetylene generated in situ from calcium carbide to afford corresponding vinyl sulfides and bis(thiovinyl)ethers in good to high yields.
References
1.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
2.

Ananikov V.P.
ACS Catalysis,
2015
3.
10.1016/j.mencom.2015.11.004_bib0010
Rodygin
Green Chem.,
2015
4.

Pearson W.H., Lee I.Y., Mi Y., Stoy P.
Journal of Organic Chemistry,
2004
5.

6.

Shaikh A.K., Cobb A.J., Varvounis G.
Organic Letters,
2012
7.
![Tandem photocyclization-intramolecular addition reactions of aryl vinyl sulfides. Observation of a novel [2 + 2] cycloaddition-allylic sulfide rearrangement](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Dittami J.P., Nie X.Y., Nie H., Ramanathan H., Buntel C., Rigatti S., Bordner J., Decosta D.L., Williard P.
Journal of Organic Chemistry,
1992
8.
![A novel entry into the bicyclo[5.4.0]undecane ring system](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Sage J.M., Raggon J.
Journal of Organic Chemistry,
1982
9.
![Short synthesis of bicyclo[3.2.2]nona-3,6,8-trien-2-one](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Sage J.M.
Journal of Organic Chemistry,
1983
10.
![[2 + 2] cycloadditions of 2,2-bis(trifluoromethyl)ethylene-1,1-dicarbonitrile with vinyl sulfides and ketene S,S-acetals](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Brückner R., Huisgen R.
Tetrahedron Letters,
1990
11.

Trost B.M., Tanigawa Y.
Journal of the American Chemical Society,
1979
12.

Kopping B., Chatgilialoglu C., Zehnder M., Giese B.
Journal of Organic Chemistry,
1992
13.

Moriarty R.M., Vaid R.K., Duncan M.P.
Synthetic Communications,
1987
14.

Davis F.A., Reddy R.T., Han W., Carroll P.J.
Journal of the American Chemical Society,
1992
15.

Glahsl G., Herrmann R.
Journal of the Chemical Society Perkin Transactions 1,
1988
16.

O'Mahony G.E., Ford A., Maguire A.R.
Journal of Sulfur Chemistry,
2012
17.

Liu J., Ueda M.
Journal of Materials Chemistry A,
2009
18.

Nakabayashi K., Abiko Y., Mori H.
Macromolecules,
2013
19.

Abiko Y., Matsumura A., Nakabayashi K., Mori H.
Polymer,
2014
20.

Kausar A., Zulfiqar S., Sarwar M.I.
Polymer Reviews,
2014
21.

Gusarova N.K., Chernysheva N.A., Yas’ko S.V., Trofimov B.A.
Russian Chemical Bulletin,
2013
22.
10.1016/j.mencom.2015.11.004_sbref0045b
Trofimov
Zh. Org. Khim.,
1973
23.

Fernandes A.C., Romão C.C.
Tetrahedron,
2006
24.

Yoo B.W., Song M.S., Park M.C.
Synthetic Communications,
2007
25.

Abele E., Dzenitis O., Rubina K., Lukevics E.
Chemistry of Heterocyclic Compounds,
2002
26.

Lo Conte M., Pacifico S., Chambery A., Marra A., Dondoni A.
Journal of Organic Chemistry,
2010
27.

Kondoh A., Takami K., Yorimitsu H., Oshima K.
Journal of Organic Chemistry,
2005
28.

Beletskaya I.P., Ananikov V.P.
Chemical Reviews,
2011
29.

Ananikov V.P., Orlov N.V., Beletskaya I.P.
Russian Chemical Bulletin,
2005
30.

Okimoto Y., Sakaguchi S., Ishii Y.
Journal of the American Chemical Society,
2002
31.

Petukhova N.P., Prilezhaeva E.N., Voropaev V.N.
Russian Chemical Bulletin,
1972