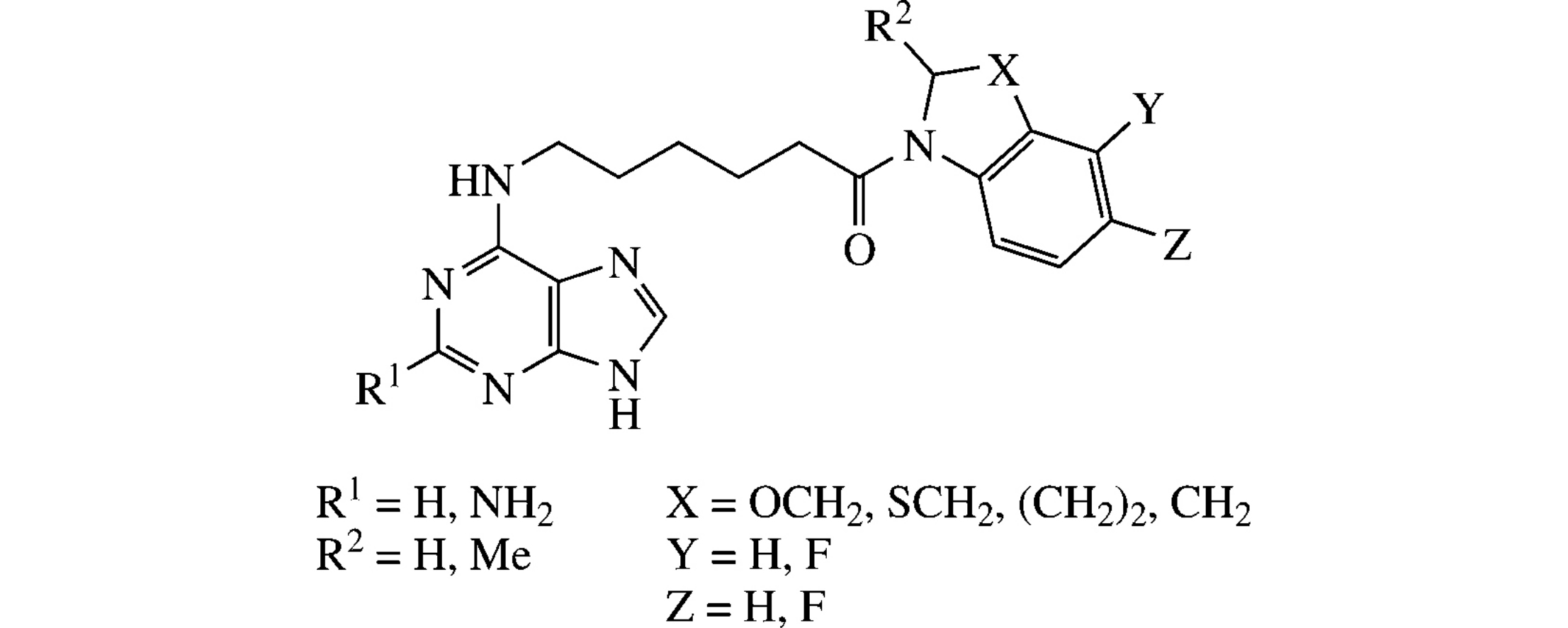
Abstract
Novel conjugates of purine and 2-aminopurine linked with heterocyclic amines, including chiral derivatives of 3,4-dihydro-2H-[1,4]benzoxazine, 3,4-dihydro-2H-[1,4]benzothiazine and 1,2,3,4-tetrahydroquinoline, by 6-aminohexanoyl fragment at the 6-position of purine moiety were obtained. For this purpose, replacement of the chlorine atom in 2-amino-6-chloropurine or 6-chloropurine by direct nucleophilic substitution reaction with 6-aminohexanamides or the coupling of 6-(purin-6-ylamino)-6-hexanoic acid with nitrogen heterocycles were used.
References
1.

Rosemeyer H.
Chemistry and Biodiversity,
2004
2.

The purines: Potent and versatile small molecule inhibitors and modulators of key biological targets
Legraverend M., Grierson D.S.
Bioorganic and Medicinal Chemistry,
2006
3.

De Clercq E.
Journal of Medicinal Chemistry,
2009
4.

Gray N.S., Wodicka L., Thunnissen A.W., Norman T.C., Kwon S., Espinoza F.H., Morgan D.O., Barnes G., LeClerc S., Meijer L., Kim S., Lockhart D.J., Schultz P.G.
Science,
1998
5.

Meijer L., Raymond E.
Accounts of Chemical Research,
2003
6.
10.1016/j.mencom.2015.11.003_bib0030
Roecker
Clin. Med. Insights: Oncol.,
2010
7.

Demir Z., Guven E.B., Ozbey S., Kazak C., Atalay R.C., Tuncbilek M.
European Journal of Medicinal Chemistry,
2015
8.

Rodenko B., van der Burg A.M., Wanner M.J., Kaiser M., Brun R., Gould M., de Koning H.P., Koomen G.
Antimicrobial Agents and Chemotherapy,
2007
9.

Kato N., Sakata T., Breton G., Le Roch K.G., Nagle A., Andersen C., Bursulaya B., Henson K., Johnson J., Kumar K.A., Marr F., Mason D., McNamara C., Plouffe D., Ramachandran V., et. al.
Nature Chemical Biology,
2008
10.
F. B. Pallier and J. L. Haesslein, FR Patent, 2851248, C07D, 2004 (Chem. Abstr., 2004, 141, 206968).
11.
G. Chen, T.D. Cushing, B. Fisher, X. He, K. Li, Z. Li, L.R. McGee, V. Pattaropong, P. Faulder, J.L. Seganish and Y. Shin, PCT Patent, WO 2009158011, C07D, 2009.(Chem. Abstr., 2009, 152, 119631).
12.
C. Guibourdenche, S. Hintermann, K. Hurth, S. Jacquier, C. Kalis, H. Moebitz and N. Soldermann, PCT Patent, WO 2014128612, C07D, 2014.(Chem. Abstr., 2014, 161, 433034).
13.

Taldone T., Rodina A., DaGama Gomes E.M., Riolo M., Patel H.J., Alonso-Sabadell R., Zatorska D., Patel M.R., Kishinevsky S., Chiosis G.
Beilstein Journal of Organic Chemistry,
2013
14.

HUANG F., WANG G., COLEMAN T., LI N.
RNA,
2003
15.

Lim H., Archer C.T., Kodadek T.
Journal of the American Chemical Society,
2007
16.

Lim H., Archer C.T., Kim Y., Hutchens T., Kodadek T.
Chemical Communications,
2008
17.

Cancilla M.T., He M.M., Viswanathan N., Simmons R.L., Taylor M., Fung A.D., Cao K., Erlanson D.A.
Bioorganic and Medicinal Chemistry Letters,
2008
18.

Samsel M., Dzierzbicka K., Trzonkowski P.
Bioorganic and Medicinal Chemistry Letters,
2014
19.

Lin X., Robins M.J.
Organic Letters,
2000
20.

Véliz E.A., Beal P.A.
Journal of Organic Chemistry,
2001
21.

Thalassitis A., Hadjipavlou-Litina D.J., Litinas K.E., Miltiadou P.
Bioorganic and Medicinal Chemistry Letters,
2009
22.

Lakshman M.K., Frank J.
Organic and Biomolecular Chemistry,
2009
23.

Devine S.M., Gregg A., Figler H., McIntosh K., Urmaliya V., Linden J., Pouton C.W., White P.J., Bottle S.E., Scammells P.J.
Bioorganic and Medicinal Chemistry,
2010
24.

Imbach P., Capraro H., Furet P., Mett H., Meyer T., Zimmermann J.
Bioorganic and Medicinal Chemistry Letters,
1999
25.
Holý A., Votruba I., Tloušťová E., Masojídková M.
Collection of Czechoslovak Chemical Communications,
2002
26.

Huang L., Cherng Y., Cheng Y., Jang J., Chao Y., Cherng Y.
Tetrahedron,
2007
27.
10.1016/j.mencom.2015.11.003_bib0135
Hu
J. Mex. Chem. Soc.,
2010
28.
Vigorov A.Y., Krasnov V.P., Gruzdev D.A., Men'shikova A.A., Demin A.M., Levit G.L., Charushin V.N.
Mendeleev Communications,
2014
29.

Guénin E., Monteil M., Bouchemal N., Prangé T., Lecouvey M.
European Journal of Organic Chemistry,
2007
30.

Hausch F., Jäschke A.
Tetrahedron,
2001