Abstract
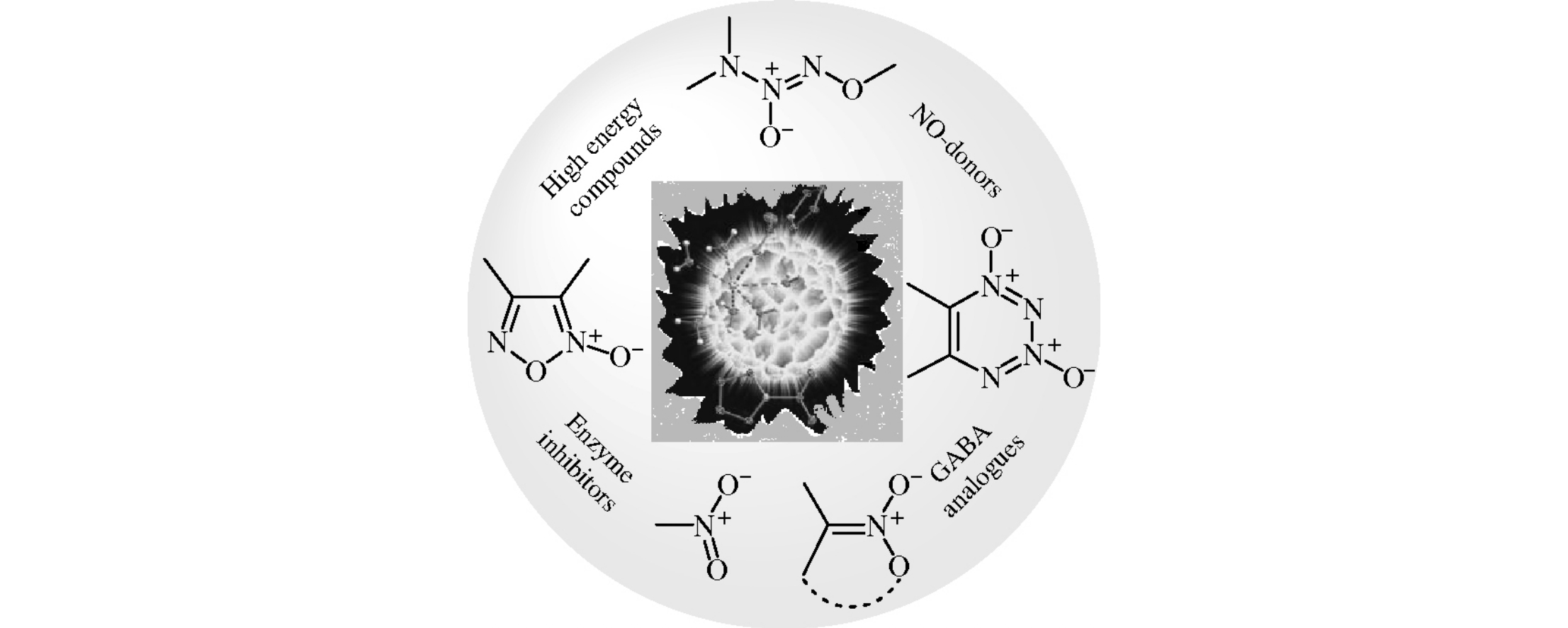
Organic compounds bearing semi-polar nitrogen–oxygen bonds are considered as perspective platform for designing socially important medications and high energy materials. Herein, recently developed original syntheses of monocyclic and fused 1,2,3,4-tetrazine 1,3-dioxides, various (methylene)bis(1-oxy-1-triazene 2-oxide) derivatives, furoxan-containing polyheterocyclic scaffolds and diastereoor enantiomerically enriched functionalized aliphatic nitro compounds are summarized. Prospects for their practical applications are outlined.
References
1.
10.1016/j.mencom.2015.11.001_bib0005
Agrawal
Organic Chemistry of Explosives,
2007
2.
(a) F. Furchgott, Angew. Chem. Int. Ed., 1999, 38, 1870; (b) F. Murad, Angew. Chem. Int. Ed., 1999, 38, 1856; (c) L. J. Ignarro, Angew. Chem. Int. Ed., 1999, 38, 1882.
3.
(a) M. Willmot, L. Gray, C. Gibson, S. Murphy and P. M. Bath, Nitric Oxide, 2005, 12, 141; (b) Nitric Oxide Donors: For Pharmaceutical and Biological Applications, eds. P. G. Wang, T.B. Cai and N. Taniguchi, Wiley-VCH, Weinheim, 2005; (c) R. Scatena, P. Bottoni, A. Pontoglio and B. Giardina, Curr. Med. Chem., 2010, 17, 61; (d) P. Krause, E. Waetzig, H. Acil, S. Koenig, K. Unthan-Fechner, D. Tsikas and I. Probst, Nitric Oxide, 2010, 23, 220; (e) R. A. M. Serafim, M.C. Primi, G.H. G. Trossini and E. I. Ferreira, Curr. Med. Chem., 2012, 19, 386.
4.
(a) Goodman and Gilman's the Pharmacological Basis of Therapeutics, eds. J. G. Hardman, L.E. Limbird and A. G. Gilman, McGraw-Hill, New York, 2001; (b) Basic Neurochemistry: Molecular, Cellular and Medical Aspects, eds. G. J. Siegel, R.W. Albers, S. Brady and D. Price, Academic Press, Elsevier, Boston, 2006.
5.

Marqués-López E., Herrera R.P., Christmann M.
Natural Product Reports,
2010
6.

Churakov A.M., Tartakovsky V.A.
Chemical Reviews,
2004
7.
(a) Q. Wu, Y. Pan, X. Xia, Y. Shao, W. Zhu and H. Xiao, Struct. Chem., 2013, 24, 1579; (b) F. Xiang, Q. Wu, W. Zhu and H. Xiao, Can. J. Chem., 2013, 91, 1233; (c) Q. Wu, W. Zhu and H. Xiao, J. Chem. Eng. Data, 2013, 58, 2748.
8.

Tan B., Huang M., Huang H., Long X., Li J., Nie F., Huang J.
Propellants, Explosives, Pyrotechnics,
2013
9.

Christe K.O., Dixon D.A., Vasiliu M., Wagner R.I., Haiges R., Boatz J.A., Ammon T.L.
Propellants, Explosives, Pyrotechnics,
2015
10.
(a) M. S. Klenov, O.V. Anikin, A.M. Churakov, Yu. A. Strelenko, I.V. Fedyanin, I.V. Ananyev and V. A. Tartakovsky, Eur. J. Org. Chem., 2015, 6170; (b) A. Yu. Tyurin, A.M. Churakov, Yu. A. Strelenko, M.O. Ratnikov and V. A. Tartakovsky, Russ. Chem. Bull., Int. Ed., 2006, 55, 1648 (Izv. Akad. Nauk, Ser. Khim., 2006, 1589); (c) M. S. Klenov, A.M. Churakov, Yu. A. Strelenko, I.V. Ananyev, K.A. Lyssenko and V. A. Tartakovsky, Tetrahedron Lett., 2015, 56, 5437.
11.

Pyatakova N.V., Khropov Y.V., Churakov A.M., Tarasova N.I., Serezhenkov V.A., Vanin A.F., Tartakovsky V.A., Severina I.S.
Biochemistry (Moscow),
2002
12.
A. M. Churakov, S.L. Ioffe, V.A. Tartakovskii, O.G. Busygina, Yu. V. Khropov and I. S. Severina, RF Patent 2123526, 1998 (Chem. Abstr., 2000, 133, 55324).
13.
N. V. Pyatakova, A.M. Kozlov, A.M. Churakov, O. Yu. Smirnov, Yu. V. Khropov, N.S. Saprykina, N.G. Bogdanova, S.L. Ioffe, I.S. Severina and V. A. Tartakovsky, RF Patent 2192857, 2002 (Chem. Abstr., 2003, 138, 314555).
14.
N. V. Dolgova, A.M. Churakov, O. Yu. Smirnov, Yu. V. Khropov, N.G. Bogdanova, N.V. Mast, S.L. Ioffe, O.D. Lopina and V. A. Tartakovsky, RF Patent 2186108, 2002 (Chem. Abstr., 2003, 138, 133153).
15.

Voronin A.A., Zelenov V.P., Churakov A.M., Strelenko Y.A., Fedyanin I.V., Tartakovsky V.A.
Tetrahedron,
2014
16.
![Synthesis of 1H-[1,2,3]triazolo[4,5-e][1,2,3,4]tetrazine 4,6-dioxide and its methyl derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Voronin A.A., Zelenov V.P., Churakov A.M., Strelenko Y.A., Tartakovsky V.A.
Russian Chemical Bulletin,
2015
17.
(a) V. G. Granik and N. B. Grigoriev, Oksid azota (NO). Novyi put’ k poisku lekarstv (Nitric oxide (NO). New Route to Drug Design), Vuzovskaya Kniga, Moscow, 2004 (in Russian); (b) J. A. Hrabie and L. K. Keefer, Chem. Rev., 2002, 102, 1135.
18.
(a) D. Biswas, R.J. Holland, J.R. Deschamps, Z. Cao, L.K. Keefer and J. E. Saavedra, J. Org. Chem., 2012, 77, 10804; (b) R. S. Drago and B. R. Karstetter, J. Am. Chem. Soc., 1961, 83, 1819; (c) D. Biswas, J.A. Hrabie, J.E. Saavedra, Z. Cao, L.K. Keeffer, J. Jvanic and R. J. Holland, J. Org. Chem., 2014, 79, 4612.
19.
(a) R. J. Holland, J.R. Klose, J.R. Deschampe, Z. Cao, L.K. Keefer and J. E. Saavedra, J. Org. Chem., 2014, 79, 9389; (b) R. S. Nandurdikar, A. E. Maciag, Z. Cao, L.K. Keefer and J. E. Saavedra, Bioorg. Med. Chem., 2012, 20, 2026.
20.

Smirnov G.A., Gordeev P.B., Nikitin S.V., Pokhvisneva G.V., Ternikova T.V., Luk´yanov O.A.
Russian Chemical Bulletin,
2015
21.

Jovene C., Chugunova E.A., Goumont R.
Mini-Reviews in Medicinal Chemistry,
2013
22.
10.1016/j.mencom.2015.11.001_bib0110
Korolev
Acta Naturale,
2013
23.
(a) L. I. Khmel’nitskii, S.S. Novikov and T. I. Godovikova, Khimiya furoksanov. Stroenie i sintez (Chemistry of Furoxans. Structure and Synthesis), 2nd edn., Nauka, Moscow, 1996 (in Russian); (b) L. I. Khmel’nitskii, S.S. Novikov and T. I. Godovikova, Khimiya furoksanov. Reaktsii i primenenie (Chemistry of Furoxans. Reactions and Application), 2nd edn., Nauka, Moscow, 1996.(in Russian).
24.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
25.
Cena C., Bertinaria M., Boschi D., Giorgis M., Gasco A.
Arkivoc,
2006
26.

Lazzarato L., Cena C., Rolando B., Marini E., Lolli M.L., Guglielmo S., Guaita E., Morini G., Coruzzi G., Fruttero R., Gasco A.
Bioorganic and Medicinal Chemistry,
2011
27.
(a) A. I. Stepanov, D.V. Dashko and A. A. Astrat’ev, Central Eur. J. Energ. Mater., 2012, 9, 329; (b) V. I. Pepekin, B.L. Korsunskii and Yu. N. Matyushin, Comb. Expl. Shock Waves, 2008, 44, 110 (Fiz. Goreniya Vzryva, 2008, 44, 123); (c) A. O. Chizhov, N.N. Makhova, I.V. Kuchurov, A.B. Sheremetev and S. G. Zlotin, Mendeleev Commun., 2014, 24, 165; (d) D. Fischer, T.M. Klapötke and J. Stierstorfer, Eur. J. Inorg. Chem., 2014, 5808.
28.
(a) N. N. Makhova, A.S. Kulikov, S.I. Molotov and E. L. Baryshnikova, Pure Appl. Chem., 2004, 76, 1691; (b) I. V. Ovchinnikov, A.O. Finogenov, M.A. Epishina, A.S. Kulikov, Yu. A. Strelenko and N. N. Makhova, Russ. Chem. Bull., Int. Ed., 2009, 58, 2137 (Izv. Akad. Nauk, Ser. Khim., 2009, 2072); (c) N. N. Makhova and A. C. Kulikov, Russ. Chem. Rev., 2013, 82, 1007; (d) L. L. Fershtat, I.V. Ovchinnikov and N. N. Makhova, Tetrahedron Lett., 2014, 55, 2398; (e) L. L. Fershtat, M.I. Struchkova, A.S. Goloveshkin, I.S. Bushmarinov and N. N. Makhova, Heteroat. Chem., 2014, 25, 226.
29.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
30.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
31.
Fershtat L.L., Ashirbaev S.S., Kulikov A.S., Kachala V.V., Makhova N.N.
Mendeleev Communications,
2015
32.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
33.

Fershtat L.L., Ananyev I.V., Makhova N.N.
RSC Advances,
2015
34.

Fershtat L.L., Epishina M.A., Ovchinnikov I.V., Kachala V.V., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
35.
Konstantinova L.S., Amelichev S.A., Zlotin S.G., Struchkova M.I., Godovikova T.I., Rakitin O.A.
Mendeleev Communications,
2015
36.
![3-R-4-Nitro-6,7-furoxanobenzo[d]isoxazoles – a new type of condensed nitroarenes capable of Diels–Alder reaction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2015
37.

Xuan B., Wang T., Chiou G.C., Dalinger I., Shkineva T.K., Shevelev S.A.
Journal of Ocular Pharmacology and Therapeutics,
2002
38.
Synthesis of nitric acid esters from alcohols in a dinitrogen pentoxide/carbon dioxide liquid system
Kuchurov I.V., Fomenkov I.V., Zlotin S.G., Tartakovsky V.A.
Mendeleev Communications,
2012
39.
(a) S. S. Novikov, G.A. Shvekhgeimer, V.V. Sevostyanova and V. A. Shlyapochnikov, Khimiya alifaticheskikh i tsikloalifaticheskikh nitrosoedinenii (Chemistry of Aliphatic and Cycloaliphatic Nitro Compounds), Khimiya, Moscow, 1974 (in Russian); (b) E. E. Gilbert and G. P. Sollott, J. Org. Chem., 1980, 45, 5405; (c) T. G. Archibald, K. Baum, M.C. Cohen and L. C. Garver, J. Org. Chem., 1989, 54, 2869; (d) R. P. Kashyap, A.P. Marchand, R. Sharma, W.H. Watson and U. R. Zope, J. Org. Chem., 1993, 58, 759; (e) P. E. Eaton, R. Gilardi, J. Hain, N. Kanomata, J. Li, K.A. Lukin and E. Punzalan, J. Am. Chem. Soc., 1997, 119, 9591.
40.
(a) L. S. Aitken, N.R. Arezki, A. Dell’Isola and A. J. A. Cobb, Synthesis, 2013, 45, 2627; (b) S. Z. Zard, Helv. Chim. Acta, 2012, 95, 1730; (c) R. Ballini and M. Petrini, ARKIVOC, 2009, 9, 195; (d) R. Ballini and M. Petrini, Tetrahedron, 2004, 60, 1017.
41.
10.1016/j.mencom.2015.11.001_bib0205
Ono
The Nitro Group in Organic Chemistry,
2001
42.

García J.M., Maestro M.A., Oiarbide M., Odriozola J.M., Razkin J., Palomo C.
Organic Letters,
2009
43.
(a) Y.-P. Li, Z.-J. Li and X.-B. Meng, Carbohydr. Res., 2011, 346, 1801; (b) R. G. Soengas and A. M. Estévez, Eur. J. Org. Chem., 2010, 5190; (c) L. Kh. Faizullina, M.G. Safarov, L.V. Spirikhin, V.S. Kolosnitsyn, Yu. A. Kondrova and F. A. Valeev, Russ. J. Org. Chem., 2011, 47, 914.(Zh. Org. Khim., 2011, 47, 897).
44.
45.
![Tandem [4+2]/[3+2] Cycloadditions of Nitroalkenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Denmark S.E., Thorarensen A.
Chemical Reviews,
1996
46.
(a) C.-Y. Zhu, X.-M. Deng, X.-L. Sun, J.-C. Zhenga and Y. Tang, Chem. Commun., 2008, 738; (b) T. Kano, A. Yamamoto, S. Song and K. Maruoka, Chem. Commun., 2011, 4358; (c) C. D. Schmidt, J. Kaschel, T.F. Schneider, D. Kratzert, D. Stalke and D. B. Werz, Org. Lett., 2013, 15, 6098.
47.

Risgaard T., Gothelf K.V., Jørgensen K.A.
Organic and Biomolecular Chemistry,
2003
48.

Ooi T., Doda K., Maruoka K.
Journal of the American Chemical Society,
2003
49.
![Synthesis of PDE IVb Inhibitors. 3. Synthesis of (+)-, (−)-, and (±)-7-[3-(Cyclopentyloxy)-4-methoxyphenyl]hexahydro-3H-pyrrolizin-3-one via Reductive Domino Transformations of 3-β-Carbomethoxyethyl-Substituted Six-Membered Cyclic Nitronates](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sukhorukov A.Y., Boyko Y.D., Nelyubina Y.V., Gerard S., Ioffe S.L., Tartakovsky V.A.
Journal of Organic Chemistry,
2012
50.
![Novel Formal [3+3] Cycloaddition of Silyl Nitronates with Activated Cyclopropanes and Its Application in the Synthesis of Pyrroline-N-oxides](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Ioffe S., Mikhaylov A., Novikov R., Khomutova Y., Arkhipov D., Korlyukov A., Tabolin A., Tomilov Y.
Synlett,
2014
51.

Wilson J.E., Casarez A.D., MacMillan D.W.
Journal of the American Chemical Society,
2009
52.

Sukhorukov A.Y., Kapatsyna M.A., Yi T.L., Park H.R., Naumovich Y.A., Zhmurov P.A., Khomutova Y.A., Ioffe S.L., Tartakovsky V.A.
European Journal of Organic Chemistry,
2014
53.

Sukhorukov A.Y., Ioffe S.L.
Chemical Reviews,
2011
54.

Halimehjani A.Z., Namboothiri I.N., Hooshmand S.E.
RSC Advances,
2014
55.
(a) Science of Synthesis: Asymmetric Organocatalysis, eds. B. List and K. Maruoka, Thieme, Stuttgart, 2012, vols. 1, 2; (b) Comprehensive Enantioselective Organocatalysis: Catalysts, Reactions, and Applications, ed. P. I. Dalco, Wiley-VCH, Weinheim, 2013, vols. 1-3.
56.

Dambrova M., Zvejniece L., Liepinsh E., Cirule H., Zharkova O., Veinberg G., Kalvinsh I.
European Journal of Pharmacology,
2008
57.

Smith D.F.
Journal of Neural Transmission,
1984
58.

Gotoh H., Ishikawa H., Hayashi Y.
Organic Letters,
2007
59.

Palomo C., Landa A., Mielgo A., Oiarbide M., Puente Á., Vera S.
Angewandte Chemie - International Edition,
2007
60.

Ishikawa H., Suzuki T., Hayashi Y.
Angewandte Chemie - International Edition,
2009
61.

Huo L., Ma A., Zhang Y., Ma D.
Advanced Synthesis and Catalysis,
2012
62.

Davis T.A., Vilgelm A.E., Richmond A., Johnston J.N.
Journal of Organic Chemistry,
2013
63.

Dobish M.C., Villalta F., Waterman M.R., Lepesheva G.I., Johnston J.N.
Organic Letters,
2012
64.

Rana N.K., Huang H., Zhao J.C.
Angewandte Chemie - International Edition,
2014
65.

Rodrigo E., García Ruano J.L., Cid M.B.
Journal of Organic Chemistry,
2013
66.

Xie H., Zhang Y., Zhang S., Chen X., Wang W.
Angewandte Chemie - International Edition,
2011
67.

Kowalczyk R., Nowak A.E., Skarżewski J.
Tetrahedron Asymmetry,
2013
68.

Chavan S.P., Garai S., Pawar K.P.
Tetrahedron Letters,
2013
69.

Liu L., Zhang S., Xue F., Lou G., Zhang H., Ma S., Duan W., Wang W.
Chemistry - A European Journal,
2011
70.

Alemán J., Cabrera S.
Chemical Society Reviews,
2013
71.
10.1016/j.mencom.2015.11.001_bib0355
Kristensen
2013
72.

Siyutkin D.E., Kucherenko A.S., Zlotin S.G.
2013
73.

Maltsev O.V., Kucherenko A.S., Beletskaya I.P., Tartakovsky V.A., Zlotin S.G.
European Journal of Organic Chemistry,
2010
74.
Maltsev O.V., Kucherenko A.S., Zlotin S.G.
Mendeleev Communications,
2011
75.

Maltsev O.V., Chizhov A.O., Zlotin S.G.
Chemistry - A European Journal,
2011
76.

Kucherenko A.S., Lisnyak V.G., Chizhov A.O., Zlotin S.G.
European Journal of Organic Chemistry,
2014
77.

Noyori R.
Chemical Communications,
2005
78.

Nigmatov A.G., Kuchurov I.V., Siyutkin D.E., Zlotin S.G.
Tetrahedron Letters,
2012
79.

Kuchurov I.V., Nigmatov A.G., Kryuchkova E.V., Kostenko A.A., Kucherenko A.S., Zlotin S.G.
Green Chemistry,
2014