Abstract
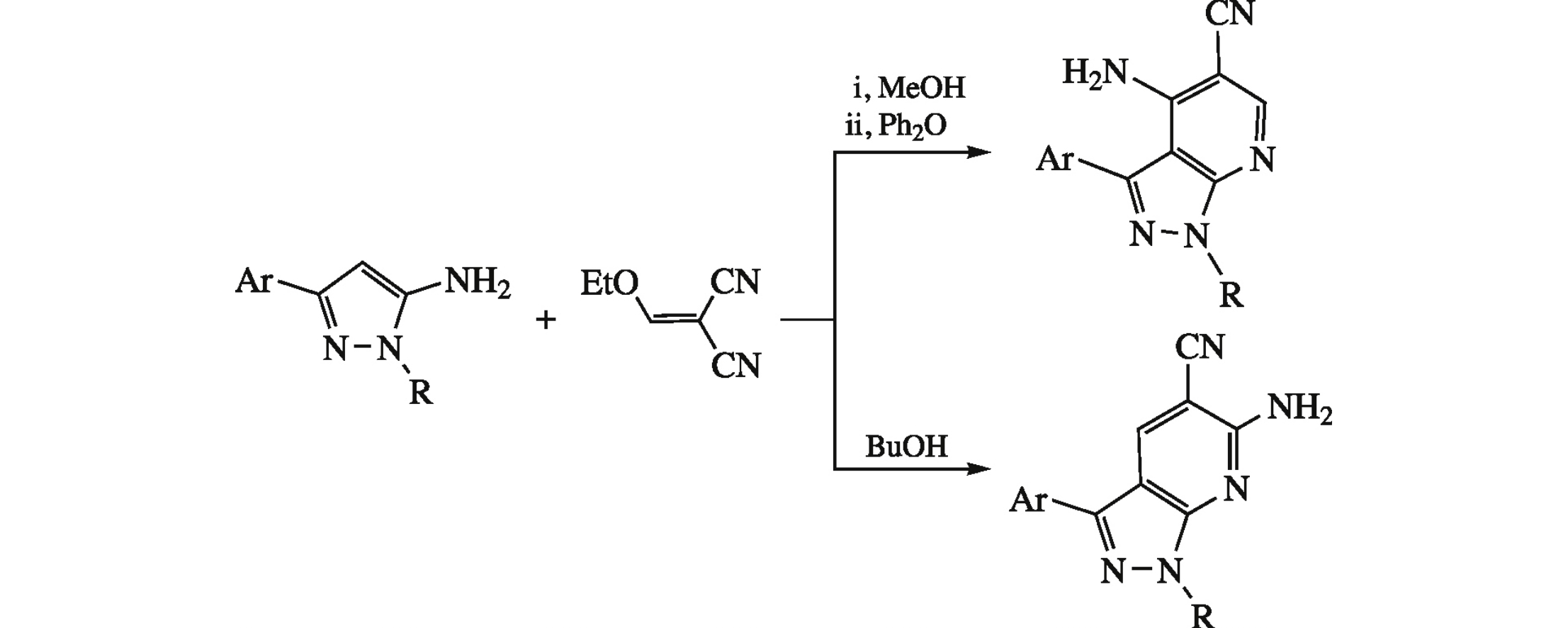
Boiling of 3-aryl-1-R-1H-pyrazol-5-amines with ethoxymethylidenemalononitrile in butanol affords 6-amino-3-aryl-1-R-1H-pyrazolo-[3,4-b]pyridine-5-carbonitriles, whereas low temperature condensation of the same reactants followed by heating to 230°C in diphenyl ether gives the isomeric 4-amino derivatives.
References
1.
10.1016/j.mencom.2015.09.023_bib0005
Elguero
Targets Heterocycl. Syst.,
2002
2.

Chebanov V.A., Gura K.A., Desenko S.M.
Topics in Heterocyclic Chemistry,
2010
3.
Emary T.I., Abdelmohsen S.A.
JOURNAL OF ADVANCES IN CHEMISTRY,
2014
4.
10.1016/j.mencom.2015.09.023_bib0020
Hasan
Int. J. Pharm. Sci.,
2013
5.
![Design and synthesis of 1,4,5,6-tetrahydropyrrolo[3,4-c]pyrazoles and pyrazolo[3,4-b]pyridines for Aurora-A kinase inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Shi J., Xu G., Zhu W., Ye H., Yang S., Luo Y., Han J., Yang J., Li R., Wei Y., Chen L.
Bioorganic and Medicinal Chemistry Letters,
2010
6.
10.1016/j.mencom.2015.09.023_bib0030
Mohamed
Der Pharma Chemica,
2010
7.

Geraldo R., Bello M., Dias L., Vera M., Nagashima T., Abreu P., Santos M., Albuquerque M., Cabral L., Freitas A., Kalil M., Rodrigues C., Castro H.
Journal of Atherosclerosis and Thrombosis,
2010
8.
![Enaminonitriles in Heterocyclic Synthesis: New Routes for the Synthesis of Some Novel Azolo[1,5-a]pyrimidine, Pyrimido[1,2-a]benzimidazole, Pyrido[1,2-a]benimdazole, Pyrazolo[3,4-b]pyridine, Pyrazole and Pyrimidine Derivatives](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Al-Zaydi K.M., Al-Shiekh M.A., Hafez E.A.
Journal of Chemical Research,
2000
9.
![Studies with Condensed Azines: New Routes toPyrazolo[3,4-b ]pyridines andPyrrolo[3,2-b ]pyridines](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Al-Mousawi S.M., Kaul K., Mohammad M.A., Elnagdi M.H.
Journal of Chemical Research,
1997
10.

Ochiai H., Ishida A., Ohtani T., Kusumi K., Kishikawa K., Yamamoto S., Takeda H., Obata T., Nakai H., Toda M.
Bioorganic and Medicinal Chemistry,
2004
11.

Panda N., Karmakar S., Jena A.K.
Chemistry of Heterocyclic Compounds,
2011
12.
![Design, Synthesis, and Pharmacological Profile of Novel Fused Pyrazolo[4,3-d]pyridine and Pyrazolo[3,4-b][1,8]naphthyridine Isosteres: A New Class of Potent and Selective Acetylcholinesterase Inhibitors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Barreiro E.J., Camara C.A., Verli H., Brazil-Más L., Castro N.G., Cintra W.M., Aracava Y., Rodrigues C.R., Fraga C.A.
Journal of Medicinal Chemistry,
2003
13.

Toche R.B., Bhavsar D.C., Kazi M.A., Bagul S.M., Jachak M.N.
Journal of Heterocyclic Chemistry,
2010
14.
![Studies on the acetylation of 3,6-diamino-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Chioua M., Soriano E., Samadi A., Marco-Contelles J.
Journal of Heterocyclic Chemistry,
2010
15.
![Synthesis of pyrazolo-annelated heterocyclic ring compounds such as pyrazolo[3,4-b]pyridines and pyrazolo[4′,3′:5,6]-pyrido[2,3-d]pyrimidines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Jachak M.N., Avhale A.B., Toche R.B., Sabnis R.W.
Journal of Heterocyclic Chemistry,
2007
16.
![Regioisomeric 4-amino- and 6-aminopyrazolo[3,4-b]pyridines: synthesis and structure determination by NMR spectroscopy and X-ray diffraction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Petrov A.A., Kasatochkin A.N., Emelina E.E., Haukka M.
Russian Chemical Bulletin,
2012
17.

HIGASHINO T., SUZUKI K., HAYASHI E.
Chemical and Pharmaceutical Bulletin,
2011
18.
![Reinvestigating the Reaction of 1H-Pyrazol-5-amines with 4,5-Dichloro-1,2,3-dithiazolium Chloride: A Route to Pyrazolo[3,4-c]isothiazoles and Pyrazolo[3,4-d]thiazoles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Koyioni M., Manoli M., Manolis M.J., Koutentis P.A.
Journal of Organic Chemistry,
2014