Abstract
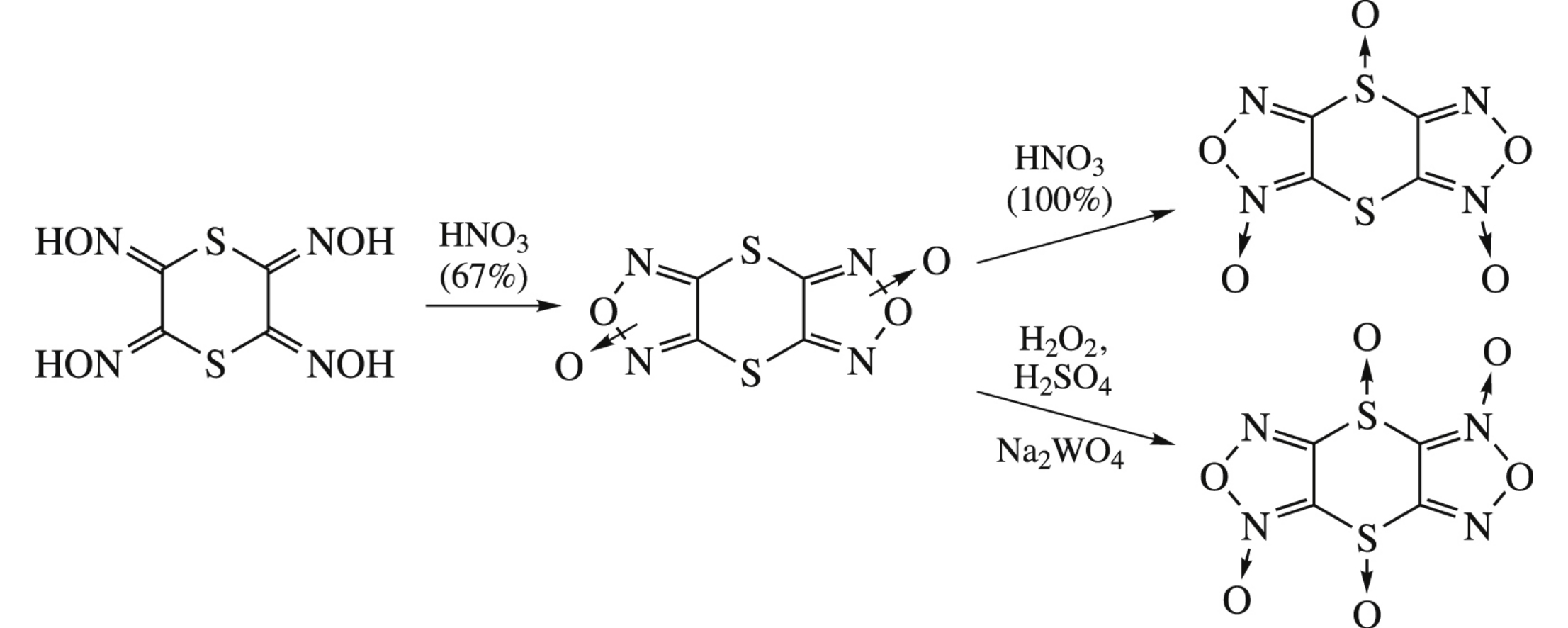
[1,4]Dithiino[2,3-c:5,6-c’]bis[1,2,5]oxadiazole di-N-oxide being an equilibrium mixture of two isomers was synthesized from 1,4-dithiane-2,3,5,6-tetraone tetraoxime and nitric acid; its oxidation to mono- and bis-S-oxides gave one isomer of each derivative.
References
1.
L. I. Khmel’nitskii, T.I. Godovikova and S. S. Novikov, Khimiya furoksanov. Struktura i sintez (Chemistry of Furoxans. Structure and Synthesis), Nauka, Moscow, 1996 (in Russian).
2.
L. I. Khmel’nitskii, T.I. Godovikova and S. S. Novikov, Khimiya furoksanov. Reaktsii i primenenie (Chemistry of Furoxans. Reactions and Application), Nauka, Moscow, 1996 (in Russian).
3.

4.

Sheremetev A.B., Makhova N.N., Friedrichsen W.
Advances in Heterocyclic Chemistry,
2001
5.

Granik V.G., Grigoriev N.B.
Russian Chemical Reviews,
2011
6.

Cerecetto H., Porcal W.
Mini-Reviews in Medicinal Chemistry,
2005
7.

Granik V.G., Ryabova S.Y., Grigoriev N.B.
Russian Chemical Reviews,
1997
8.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
9.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
10.

Lazzarato L., Cena C., Rolando B., Marini E., Lolli M.L., Guglielmo S., Guaita E., Morini G., Coruzzi G., Fruttero R., Gasco A.
Bioorganic and Medicinal Chemistry,
2011
11.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
12.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
13.

Fershtat L.L., Struchkova M.I., Goloveshkin A.S., Bushmarinov I.S., Makhova N.N.
Heteroatom Chemistry,
2014
14.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
15.
![3-R-4-Nitro-6,7-furoxanobenzo[d]isoxazoles – a new type of condensed nitroarenes capable of Diels–Alder reaction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2015
16.
Bastrakov M.A., Starosotnikov A.M., Fedyanin I.V., Kachala V.V., Shevelev S.A.
Mendeleev Communications,
2014
17.
Leyva E., Leyva S., González-Balderas R.M., de Loera D., Jiménez-Cataño R.
Mendeleev Communications,
2013
18.
![Vasorelaxant and antiplatelet activity of 4,7-dimethyl-1,2,5-oxadiazolo[3,4-d]pyridazine 1,5,6-trioxide: Role of soluble guanylate cyclase, nitric oxide and thiols](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kots A.Y., Grafov M.A., Khropov Y.V., Betin V.L., Belushkina N.N., Busygina O.G., Yazykova M.Y., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Medvedeva N.A., Bulargina T.V., Severina I.S.
British Journal of Pharmacology,
2000
19.

Ghigo D., Calvino R., Heller R., Calvino R., Alessio P., Fruttero R., Gasco A., Bosia A., Pescarmona G.
Biochemical Pharmacology,
1992
20.

Ferioli R., Fazzini A., Folco G.C., Fruttero R., Calvino R., Gasco A., Bongrani S., Civelli M.
Pharmacological Research,
1993
21.

Sorba G., Medana C., Fruttero R., Cena C., Di Stilo A., Galli U., Gasco A.
Journal of Medicinal Chemistry,
1997
22.

Calvino R., Fruttero R., Ghigo D., Bosia A., Pescarmona G.P., Gasco A.
Journal of Medicinal Chemistry,
1992
23.

Grundmann C., Mini V., Dean J.M., Frommeld H.
European Journal of Organic Chemistry,
1965
24.

Nakayama J., Nakamura Y., Hoshino M.
Heterocycles,
1985
25.

Barabanova A.V., Fedotova O.V., Kharchenko V.G., Samokhvalov V.A.
Chemistry of Heterocyclic Compounds,
2001
26.

Rostami A., Akradi J.
Tetrahedron Letters,
2010
27.

Maślankiewicz M.J., Jaworska M., Lodowski P.
Journal of Heterocyclic Chemistry,
2007
28.

Zolfigol M.A., Amani K., Ghorbani-Choghamarani A., Hajjami M., Ayazi-Nasrabadi R., Jafari S.
Catalysis Communications,
2008
29.
L. A. Kazitsyna and N. B. Kupletskaya, Primenenie UF, IK, YaMR i mass-spektroskopii v organicheskoi khimii (Employment of UV, IR, NMR and Mass Spectroscopy in Organic Chemistry), MSU, Moscow, 1979 (in Russian).