Abstract
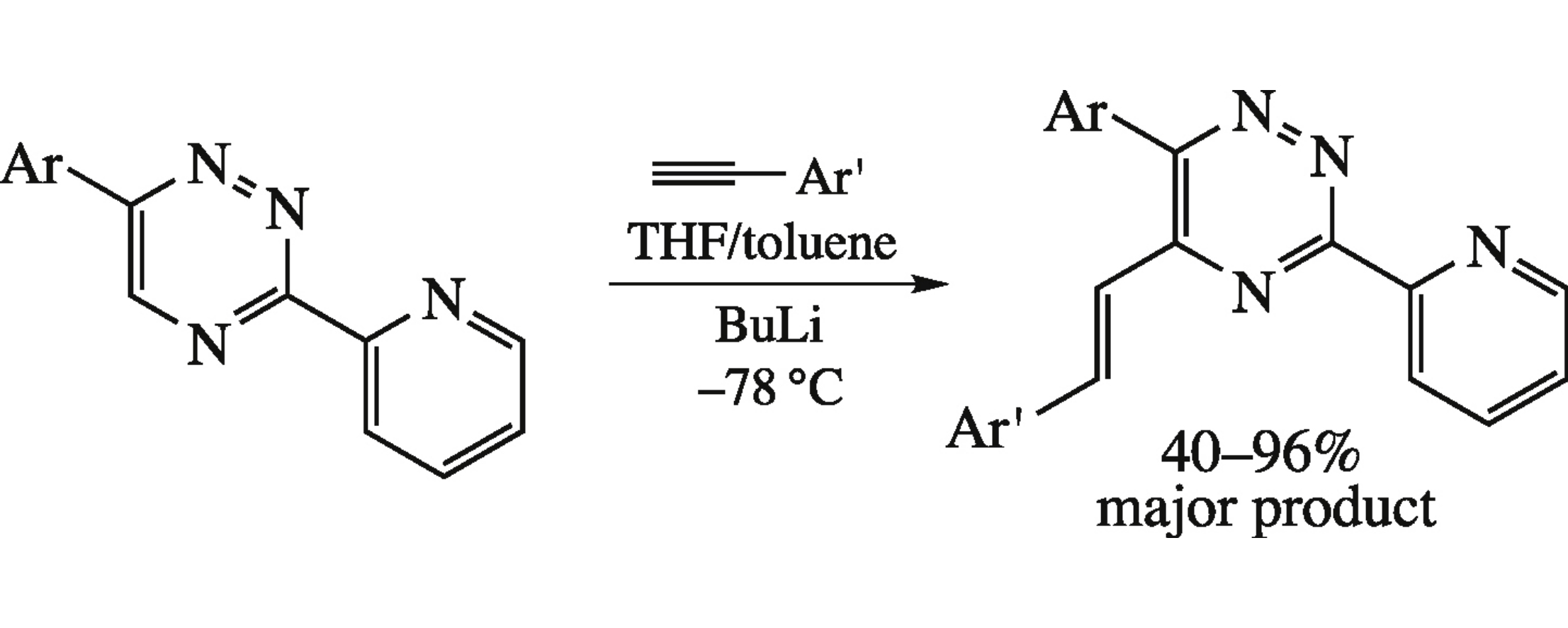
Reaction of 6-aryl-3-(2-pyridyl)-1,2,4-triazines with lithium 2-arylethynides affords (E)-6-aryl-5-arylvinyl-3-(2-pyridyl)-1,2,4-triazines.
References
1.

Heravi M.M., Sadjadi S.
Tetrahedron,
2009
2.
![Copper-Catalyzed Tandem Synthesis of Indolo-, Pyrrolo[2,1-a]isoquinolines, Naphthyridines and Bisindolo/Pyrrolo[2,1-a]isoquinolines via Hydroamination of ortho-Haloarylalkynes Followed by C-2 Arylation](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Verma A.K., Jha R.R., Chaudhary R., Tiwari R.K., Reddy K.S., Danodia A.
Journal of Organic Chemistry,
2012
3.

Taylor E.C., Macor J.E., Pont J.L.
Tetrahedron,
1987
4.

Ma Z., Ni F., Woo G.H., Lo S., Roveto P.M., Schaus S.E., Snyder J.K.
Beilstein Journal of Organic Chemistry,
2012
5.

Fadel S., Hajbi Y., Khouili M., Lazar S., Suzenet F., Guillaumet G.
Beilstein Journal of Organic Chemistry,
2014
6.

7.

Fukuzawa S., Hiroki H., Ogata K.
Synlett,
2013
8.

Novák Z., Kovács S., Zih-Perényi K., Révész Á.
Synthesis,
2012
9.

Benniston A.C., Harriman A., Lawrie D.J., Mayeux A., Rafferty K., Russell O.D.
Dalton Transactions,
2003
10.

Joshi H.S., Jamshidi R., Tor Y.
Angewandte Chemie - International Edition,
1999
11.
10.1016/j.mencom.2015.09.003_sbref0025a
Yamaguchi
J. Am. Chem. Soc.,
2001
12.
10.1016/j.mencom.2015.09.003_sbref0025b
Yang
J. Am. Chem. Soc.,
1998
13.

Andrew T.L., Swager T.M.
Journal of Polymer Science, Part B: Polymer Physics,
2011
14.

Chinchilla R., Nájera C.
Chemical Reviews,
2007
15.
10.1016/j.mencom.2015.09.003_sbref0030b
Karak
RSC Adv.,
2014
16.

Bakherad M.
Applied Organometallic Chemistry,
2013
17.

Chinchilla R., Nájera C.
Chemical Society Reviews,
2011
18.

Anabuki S., Tokuji S., Aratani N., Osuka A.
Organic Letters,
2012
19.
(a) D. N. Kozhevnikov, V.N. Kozhevnikov, A.M. Prokhorov, M.M. Ustinova, V.L. Rusinov, O.N. Chupakhin, G.G. Aleksandrov and B. Köenig, Tetrahedron Lett., 2006, 47, 869; (b) A. M. Prokhorov, P.A. Slepukhin and D. N. Kozhevnikov, J. Organomet. Chem., 2008, 693, 1886.
20.

Prokhorov A.M., Mąkosza M., Chupakhin O.N.
Tetrahedron Letters,
2009
21.
10.1016/j.mencom.2015.09.003_bib0050
Chupakhin
Nucleophilic Aromatic Substitution of Hydrogen,
1994
22.

23.

Kozhevnikov D.N., Rusinov V.L., Chupakhin O.N.
Advances in Heterocyclic Chemistry,
2002
24.

Pabst G.R., Pfüller O.C., Sauer J.
Tetrahedron,
1999
25.
(a) V. N. Kozhevnikov, D. N. Kozhevnikov, T. V. Nikitina, V. L. Rusinov, O. N. Chupakhin, M. Zabel and B. König, J. Org. Chem., 2003, 68, 2882; (b) A. M. Prokhorov, D. N. Kozhevnikov, V. L. Rusinov, O. N. Chupakhin, I. V. Glukhov, M. Yu. Antipin, O. N. Kazheva, A. N. Chekhlov and O. A. Dyachenko, Organometallics, 2006, 25, 2972; (c) I. S. Kovalev, D. S. Kopchuk, A. F. Khasanov, G. V. Zyryanov, V. L. Rusinov and O. N. Chupakhin, Mendeleev Commun., 2014, 24, 117.
26.
![Synthesis and Pharmacological Evaluation of Phenylethynyl[1,2,4]methyltriazines as Analogues of 3-Methyl-6-(phenylethynyl)pyridine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Carroll F.I., Kotturi S.V., Navarro H.A., Mascarella S.W., Gilmour B.P., Smith F.L., Gabra B.H., Dewey W.L.
Journal of Medicinal Chemistry,
2007
27.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
28.

Kozhevnikov D.N., Kataeva N.N., Rusinov V.L., Chupakhin O.N.
Russian Chemical Bulletin,
2004
29.
10.1016/j.mencom.2015.09.003_bib0090
Beveridge
Synthesis,
2010
30.

Yamaguchi M., Akamatsu K., Amemiya R.
Heterocycles,
2004
31.

Guram A.S., Jordan R.F.
Journal of Organic Chemistry,
1992
32.
![[Ru(η5-C5H5)(η6-C10H8)]PF6as a catalyst precursor for the one-pot direct C–H alkenylation of nitrogen heterocycles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Lynam J.M., Milner L.M., Mistry N.S., Slattery J.M., Warrington S.R., Whitwood A.C.
Dalton Transactions,
2014
33.

Sopbué Fondjo E., Döpp D., Henkel G.
Tetrahedron,
2006
34.
![Studies with condensed amino-thiophenes: Further investigation of reactivity of amino-thieno-coumarines and amino-thieno-benzo[h]coumarines toward electron-poor olefins and acetylenes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Abdelkhalik M.M., Negm A.M., Elkhouly A.I., Elnagdi M.H.
Heteroatom Chemistry,
2004
35.

Reinhoudt D.N., Geevers J., Trompenaars W.P., Harkema S., Van Hummel G.J.
Journal of Organic Chemistry,
1981
36.

Zhao D., Shen Q., Zhou Y., Li J.
Organic and Biomolecular Chemistry,
2013