Abstract
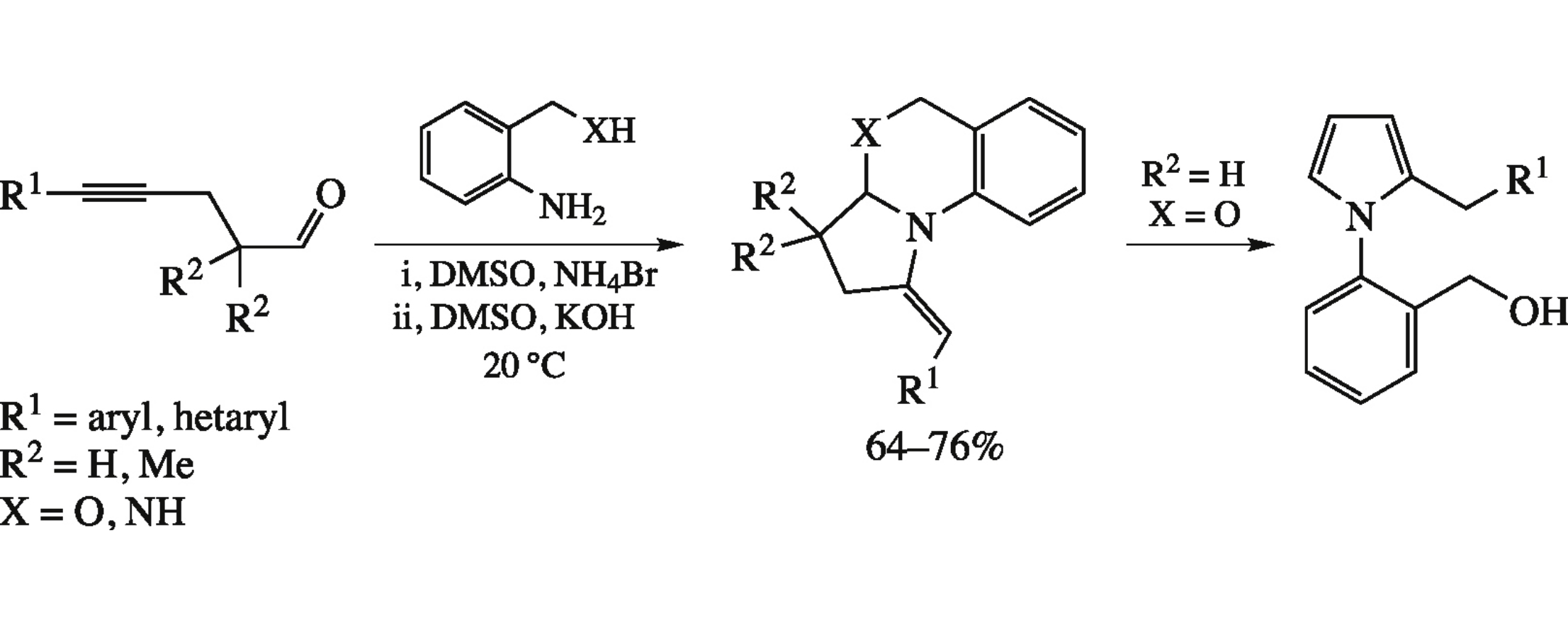
Cyclization of 5-arylalk-4-ynals with o-aminobenzylamine or o-aminobenzyl alcohol in DMSO under the sequential action of NH4Br and KOH affords (E)-1-arylmethylidene-1,2,3,3a,4,5-hexahydropyrrolo[1,2-a]quinazolines or (E)-1-arylmethylidene-1,2,3,3a-tetrahydro- 5H-pyrrolo-[1,2-a][3,1]benzoxazines; the latter without substituents in the 3-position easily isomerize into the corresponding 2-arylmethyl- 1-(2-hydroxymethylphenyl)pyrroles.
References
1.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
2.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2008
3.
K. N. Shavrin, V. D. Gvozdev and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2010, 59, 1451.(Izv. Akad. Nauk, Ser. Khim., 2010, 1418).
4.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2013
5.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2013
6.

Gvozdev V.D., Shavrin K.N., Nefedov O.M.
Russian Chemical Bulletin,
2013
7.

Gvozdev V.D., Shavrin K.N., Nefedov O.M.
Russian Chemical Bulletin,
2014
8.

Trofimov B.A., Schmidt E.Y.
Russian Chemical Reviews,
2014
9.

Patil N.T., Mutyala A.K., Konala A., Tella R.B.
Chemical Communications,
2012
10.

David O., Calvet S., Chau F., Vanucci-Bacqué C., Fargeau-Bellassoued M., Lhommet G.
Journal of Organic Chemistry,
2004
11.

Jha R.R., Aggarwal T., Verma A.K.
Tetrahedron Letters,
2014
12.

Chen Z., Hu G., Li D., Chen J., Li Y., Zhou H., Xie Y.
Bioorganic and Medicinal Chemistry,
2009
13.

Dong G., Wang S., Miao Z., Yao J., Zhang Y., Guo Z., Zhang W., Sheng C.
Journal of Medicinal Chemistry,
2012
14.

Darras F.H., Kling B., Heilmann J., Decker M.
ACS Medicinal Chemistry Letters,
2012
15.

Johnson R.A., Gorman R.R., Wnuk R.J., Crittenden N.J., Aiken J.W.
Journal of Medicinal Chemistry,
1993
16.

Perola E., Xu K., Kollmeyer T.M., Kaufmann S.H., Prendergast F.G., Pang Y.
Journal of Medicinal Chemistry,
2000
17.

Sondhi S.M., Goyal R.N., Lahoti A.M., Singh N., Shukla R., Raghubir R.
Bioorganic and Medicinal Chemistry,
2005
18.

Marchand C., Beutler J.A., Wamiru A., Budihas S., Möllmann U., Heinisch L., Mellors J.W., Le Grice S.F., Pommier Y.
Antimicrobial Agents and Chemotherapy,
2008
19.
![SYNTHESIS, CHARACTERIZATION AND IN VITRO ANTIFUNGAL EVALUATION OF TETRAHYDROPYRAZOLO[1,5-c]QUINAZOLINES](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Insuasty B., Torres H., Quiroga J., Abonía R., Nogueras M., Sánchez A., Sortino M., Zacchino S., Low J.
Heterocyclic Communications,
2003
20.

Jakopović I.P., Kragol G., Forrest A.K., Frydrych C.S., Štimac V., Kapić S., Škugor M.M., Ilijaš M., Paljetak H.Č., Jelić D., Holmes D.J., Hickey D.M., Verbanac D., Eraković Haber V., Alihodžić S., et. al.
Bioorganic and Medicinal Chemistry,
2010
21.

Hitotsuyanagi Y., Naka Y., Yamagami K., Fujii A., Tahara T.
Journal of the Chemical Society Chemical Communications,
1995
22.
(a) J. B. Chylinska, M. Janowiec and T. B. Urbarnsky, J. Pharmacol., 1971, 43, 649; (b) G. P. Moloney, D. J. Craik and M. N. Iskander, J. Pharm. Sci., 1992, 81, 692; (c) N. A. Shakil, A. Dhawan, N. K. Sharma, V. Kumar, S. Kumar, M. Bose, H. G. Raj, C. E. Olsen, A. L. Cholli, L. A. Samuelson, J. Kumar, A. C. Watterson, V. S. Parmar and A. K. Prasad, Indian J. Chem., Sect. B, 2003, 42, 1958; (d) B. M. Gurupadayya, Y. N. Manohara and M. Gopal, Indian J. Heterocycl. Chem., 2005, 15, 113.
23.

Gemma S., Camodeca C., Brindisi M., Brogi S., Kukreja G., Kunjir S., Gabellieri E., Lucantoni L., Habluetzel A., Taramelli D., Basilico N., Gualdani R., Tadini-Buoninsegni F., Bartolommei G., Moncelli M.R., et. al.
Journal of Medicinal Chemistry,
2012
24.

Largeron M., Lockhart B., Pfeiffer B., Fleury M.
Journal of Medicinal Chemistry,
1999
25.

Koini E.N., Papazafiri P., Vassilopoulos A., Koufaki M., Horváth Z., Koncz I., Virág L., Papp G.J., Varró A., Calogeropoulou T.
Journal of Medicinal Chemistry,
2009
26.

Wang H., Ganesan A.
Journal of Organic Chemistry,
2000
27.
B. Masesane I., Muriithi E., H. Tabane T.
Bulletin of the Chemical Society of Ethiopia,
2014
28.

Raju B.C., Theja N.D., Kumar J.A.
Synthetic Communications,
2008