Abstract
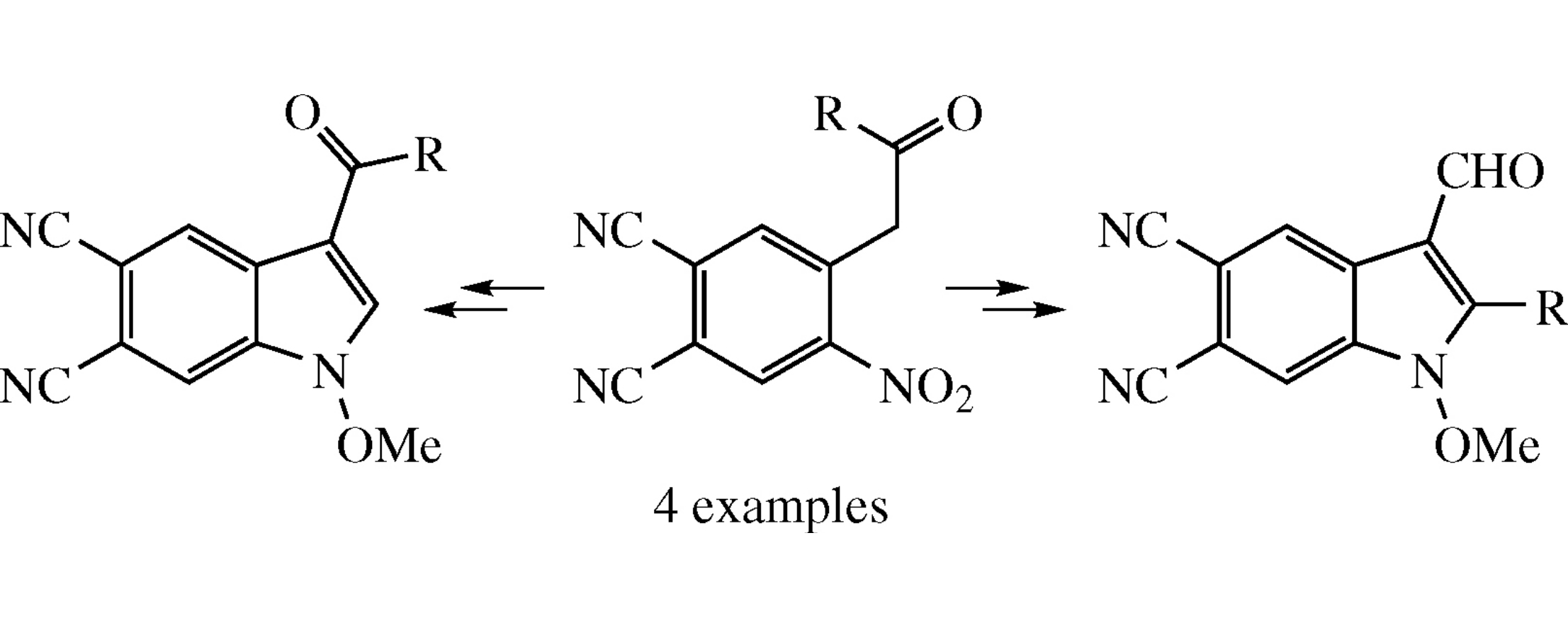
The Vilsmeier–Haack formylation of 4-(2-aryl-2-oxoethyl)-5-nitrophthalonitriles followed by reduction affords 3-acyl-1-hydroxy- 1H-indole-5,6-dicarbonitriles.
References
1.

Pedras M.S., Sarwar M.G., Suchy M., Adio A.M.
Phytochemistry,
2006
2.

Frost J.M., Dart M.J., Tietje K.R., Garrison T.R., Grayson G.K., Daza A.V., El-Kouhen O.F., Yao B.B., Hsieh G.C., Pai M., Zhu C.Z., Chandran P., Meyer M.D.
Journal of Medicinal Chemistry,
2009
3.

Davies I.W., Smitrovich J.H., Sidler R., Qu C., Gresham V., Bazaral C.
Tetrahedron,
2005
4.

Nicolaou K.C., Estrada A.A., Freestone G.C., Lee S.H., Alvarez-Mico X.
Tetrahedron,
2007
5.

LI W., LEET J.E., AX H.A., GUSTAVSON D.R., BROWN D.M., TURNER L., BROWN K., CLARK J., YANG H., FUNG-TOMC J., LAM K.S.
Journal of Antibiotics,
2003
6.

Granchi C., Roy S., Giacomelli C., Macchia M., Tuccinardi T., Martinelli A., Lanza M., Betti L., Giannaccini G., Lucacchini A., Funel N., León L.G., Giovannetti E., Peters G.J., Palchaudhuri R., et. al.
Journal of Medicinal Chemistry,
2011
7.
10.1016/j.mencom.2015.07.030_bib0035
Ottoni
Org. Lett.,
2001
8.

Guchhait S.K., Kashyap M., Kamble H.
Journal of Organic Chemistry,
2011
9.
10.1016/j.mencom.2015.07.030_bib0045
Wynne
Synthesis,
2004
10.

Anthony W.
Journal of Organic Chemistry,
1960
11.

Cacchi S., Fabrizi G.
Chemical Reviews,
2005
12.

Humphrey G.R., Kuethe J.T.
Chemical Reviews,
2006
13.

Okuro K., Gurnham J., Alper H.
Journal of Organic Chemistry,
2011
14.

Jiang T., Wang G.
Organic Letters,
2013
15.

Ma Y., You J., Song F.
Chemistry - A European Journal,
2012
16.

Wu W., Su W.
Journal of the American Chemical Society,
2011
17.

Alford J.S., Davies H.M.
Journal of the American Chemical Society,
2014
18.

Söderberg B., Banini S., Turner M., Minter A., Arrington A.
Synthesis,
2008
19.

Janreddy D., Kavala V., Bosco J.W., Kuo C., Yao C.
European Journal of Organic Chemistry,
2011
20.

Attar K., Camara H.D., Benchidmi M., Essassi Ε.M., Pierrot M.
Heterocyclic Communications,
2003
21.

Filimonov S.I., Chirkova Z.V., Abramov I.G., Firgang S.I., Stashina G.A., Strelenko Y.A., Khakimov D.V., Pivina T.S., Samet A.V., Suponitsky K.Y.
Tetrahedron,
2012
22.

Marson C.M.
Tetrahedron,
1992
23.

Prim D., Fuss A., Kirsch G., Silva A.M.
Journal of the Chemical Society Perkin Transactions 2,
1999
24.
S. I. Filimonov, Zh. V. Chirkova, V.S. Sharunov, I.G. Abramov, S.I. Firgang and G. A. Stashina, Chem. Heterocycl. Compd. (Engl. Transl.), 2012, 48, 427.(Khim. Geterotsikl. Soedin., 2012, 455).
25.
10.1016/j.mencom.2015.07.030_bib0125
Goerlitzer
Pharmazie,
1998
26.

Chirkova Z.V., Kabanova M.V., Filimonov S.I., Abramov I.G., Petzer A., Petzer J.P., Firgang S.I., Suponitsky K.Y.
Bioorganic and Medicinal Chemistry Letters,
2015
27.

NAGAYOSHI T., SAEKI S., HAMANA M.
Chemical and Pharmaceutical Bulletin,
2011