Abstract
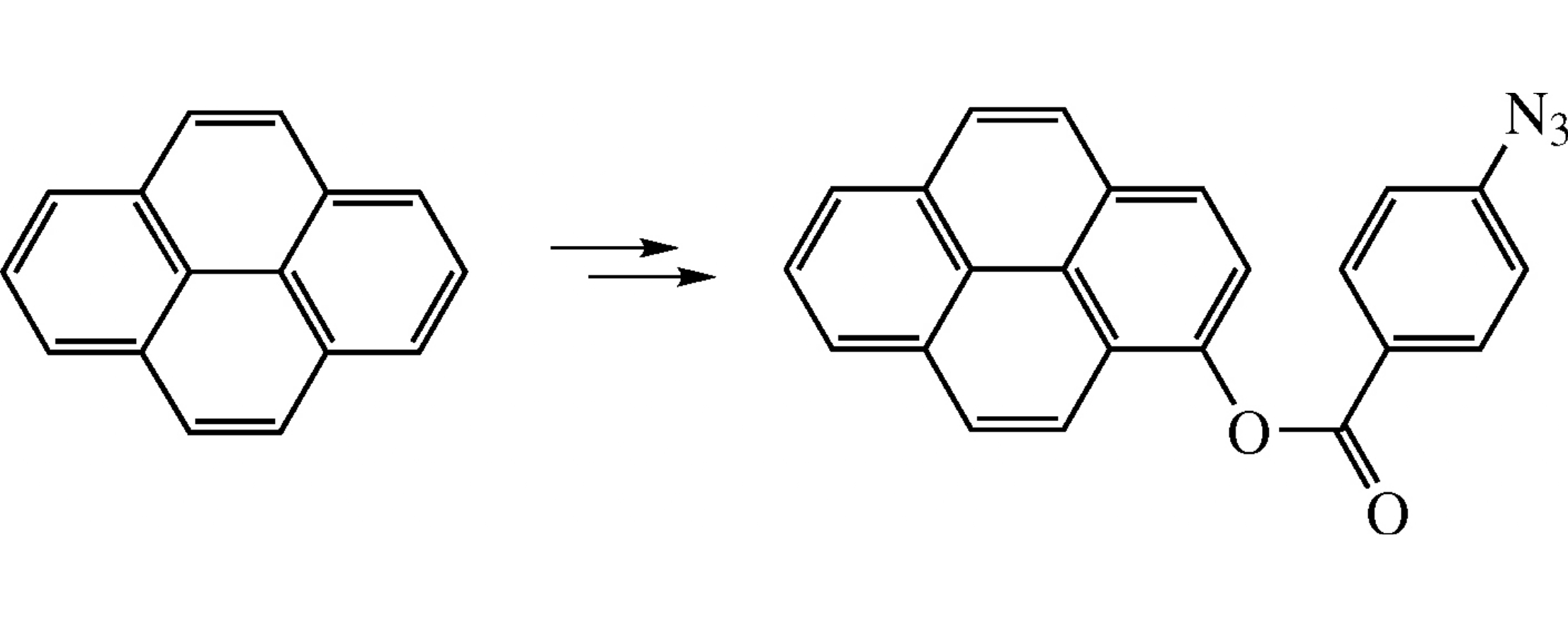
Lead tetraacetate oxidation of pyrene followed by hydrolysis affords 1-hydroxypyrene whose esterification with 4-azidobenzoic acid gives a new bifunctional luminophore characterized by UV and luminescence spectroscopy.
References
1.
Barabanov I.I., Pritchina E.A., Takaya T., Gritsan N.P.
Mendeleev Communications,
2008
2.

Shields C.J., Falvey D.E., Schuster G.B., Buchardt O., Nielsen P.E.
Journal of Organic Chemistry,
1988
3.
M. I. Dobrikov, S.A. Gaidamakov, A.A. Koshkin and V. V. Vlasov, Dokl. Akad. Nauk, 1996, 351, 687.(in Russian).
4.

Bichenkova E.V., Marks D., Dobrikov M.I., Vlassov V.V., Morris G.A., Douglas K.T.
Journal of Biomolecular Structure and Dynamics,
1999
5.

Dobrikov M.I.
Russian Chemical Reviews,
1999
6.
Gritsan N., Platz M.
2009
7.

Gritsan N.P., Pritchina E.A., Barabanov I.I., Burdzinski G.T., Platz M.S.
Journal of Physical Chemistry C,
2009
8.

MILLON R., OLOMUCKI M., GALL J., GOLINSKA B., EBEL J., EHRESMANN B.
FEBS Journal,
1980
9.
W. H. Gumprecht, Org. Synth., 1973, coll. vol. 5, 632;
10.

11.
W. Kern, U.S. Patent, 2018792, 1935.
12.
10.1016/j.mencom.2015.07.008_sbref0045d
Mataka
Rep. Inst. Adv. Mat. Study,
1996
13.

Babu P., Sangeetha N.M., Vijaykumar P., Maitra U., Rissanen K., Raju A.R.
Chemistry - A European Journal,
2003
14.

Harvey R.G., Hahn J.T., Bukowska M., Jackson H.
Journal of Organic Chemistry,
1990
15.

Riley J.M., Alkan S., Chen A., Shapiro M., Khan W.A., Murphy, W.R., Hanson J.E.
Macromolecules,
2001
16.

Fieser L.F., Clapp R.C., Daudt W.H.
Journal of the American Chemical Society,
1942
17.
![Improved synthesis of benzo[a]pyren-1-ol and isolation of a covalent benzo[a]pyrene–lead compound](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Harvey R.G., Cho H.
Journal of the Chemical Society Chemical Communications,
1975
18.
10.1016/j.mencom.2015.07.008_bib0060
El-Bayoumy
Cancer Res,
1983
19.

Scheepers P.T., Fijneman P.H., Beenakkers M.F., de Lepper A.J., Thuis H.J., Stevens D., Van Rooij J.G., Noordhoek J., Bos R.P.
Fresenius Journal of Analytical Chemistry,
1995
20.

Holloway M.P., Biaglow M.C., McCoy E.C., Anders M., Rosenkranz H.S., Howard P.C.
Mutation Research/Genetic Toxicology,
1987
21.

Mukaiyama T., Usui M., Shimada E., Saigo K.
Chemistry Letters,
1975
22.
10.1016/j.mencom.2015.07.008_bib0080
Birks
Photophysics of Aromatic Molecules,
1969
23.

Geiger M.W., Turro N.J.
Photochemistry and Photobiology,
1975