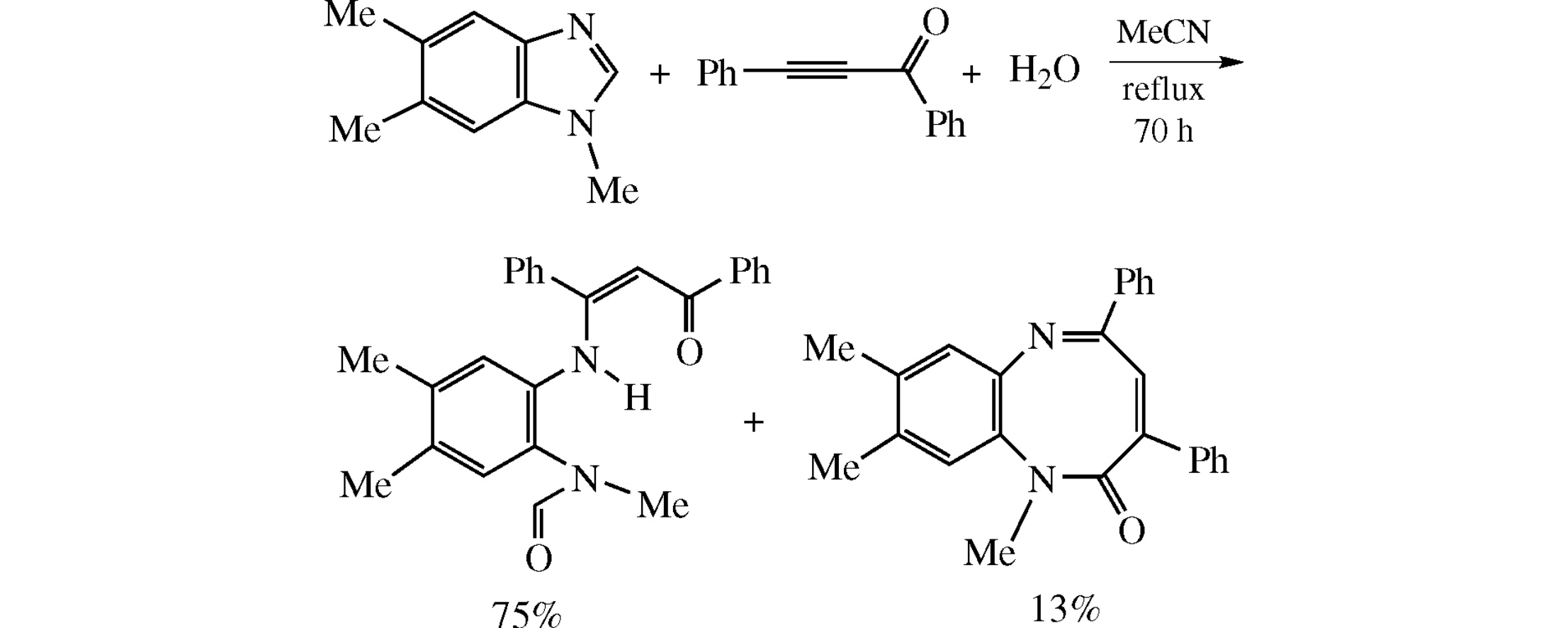
Abstract
Refluxing the mixture of 1,5,6-trimethylbenzimidazole, 1,3-diphenylprop-2-yn-1-one and water in MeCN for 70h affords (Z)-3-[2-(N-formyl-N-methylamino)-4,5-dimethylphenylamino]-1,3-diphenylprop-2-en-1-one (75%) and 1,8,9-trimethyl-3,5-diphenyl- 1,6-benzodiazocin-2(1H)-one (13%).
References
1.
(a) M. Adib, M. Mollahosseini, H. Yavari, M.H. Sayahi and H. R. Bijanzadeh, Synlett, 2004, 1086; (b) I. Yavari, M. Sabbaghan and Z. Hossaini, Synlett, 2006, 2501; (c) A. Shaabani, A.H. Rezayan, A. Sarvary, M. Heidary and S. W. Ng, Tetrahedron, 2009, 65, 6063; (d) Kh. Pourshamsian, N. Montazeri, S. Ali-Asgari, M.M. Zeydi and E. Biazar, Orient. J. Chem., 2011, 27, 1017; (e) Y. Shen, S. Cai, C. He, X. Lin, P. Lu and Y. Wang, Tetrahedron, 2011, 67, 8338; (f) K. V. Belyaeva, L.V. Andriyankova, L.P. Nikitina, A.G. Mal’kina, A.V. Afonin and B. A. Trofimov, Tetrahedron Lett., 2012, 53, 7040; (g) B. A. Trofimov, L.V. Andriyankova and K. V. Belyaeva, Chem. Heterocycl. Compd., 2012, 48, 147 (Khim. Geterotsikl. Soedin., 2012, 153) and references cited therein; (h) B. A. Trofimov, L.V. Andriyankova, L.P. Nikitina, K.V. Belyaeva, A.G. Mal’kina, A.V. Afonin and I. A. Ushakov, Synlett, 2012, 23, 2069; (i) K. V. Belyaeva, L.V. Andriyankova, L.P. Nikitina, G. Mal’kina, A.V. Afonin, I.A. Ushakov, I. Yu. Bagryanskaya and A. Trofimov, Tetrahedron, 2014, 70, 1091.
2.

Trofimov B.A., Andriyankova L.V., Nikitina L.P., Belyaeva K.V., Mal’kina A.G., Sobenina L.N., Afonin A.V., Ushakov I.A.
Organic Letters,
2013
3.

Acheson R.M., Wallis J.D., Woollard J.
Journal of the Chemical Society Perkin Transactions 1,
1979
4.
10.1016/j.mencom.2015.07.006_bib0020
Wallis
Acta Crystallogr.,
1992
5.

6.
(a) J. P. Vacca, J.S. Wai, L.S. Payne, R.C. A. Isaacs, W. Han, M. Egbertson and R. Pracitto, PCT Int. Appl., WO 2006121831 A2 20061116, 2006 (Chem. Abstr., 2006, 145, 505478); (b) S. Lohani, Z. Peng and A. E. Mckeown, U.S. Pat. Appl. Publ., US 20080280945 A1 20081113, 2008 (Chem. Abstr., 2008, 149, 556676); (c) R. C. A. Isaacs, J.S. Wai and T. E. Fisher, PCT Int. Appl., WO 2009154870 A1 20091223, 2009 (Chem. Abstr., 2009, 152, 97513).
7.

Reddy N.S., Reddy B.V.
Tetrahedron Letters,
2014
8.
(a) S. I. Miller and R. Tanaka, in Selective Organic Transformations, ed. B. S. Thyagarajan, Wiley-Interscience, New York, 1970, vol. 1, p 143; J. I. Dickstein and S. I. Miller, in The Chemistry of the Carbon– Carbon Triple Bond, ed. S. Patai, Wiley, New York, 1978, vol. 2, p. 814.
9.

Trofimov B.A., Andriyankova L.V., Belyaeva K.V., Mal’kina A.G., Nikitina L.P., Afonin A.V., Ushakov I.A.
Journal of Organic Chemistry,
2008
10.
Starikova O.V., Dolgushin G.V., Larina L.I., Komarova T.N., Lopyrev V.A.
Arkivoc,
2003
11.
10.1016/j.mencom.2015.07.006_bib0055
Zanina
Russ. Chem. Bull., 1995, 44, 689.,
1995