Abstract
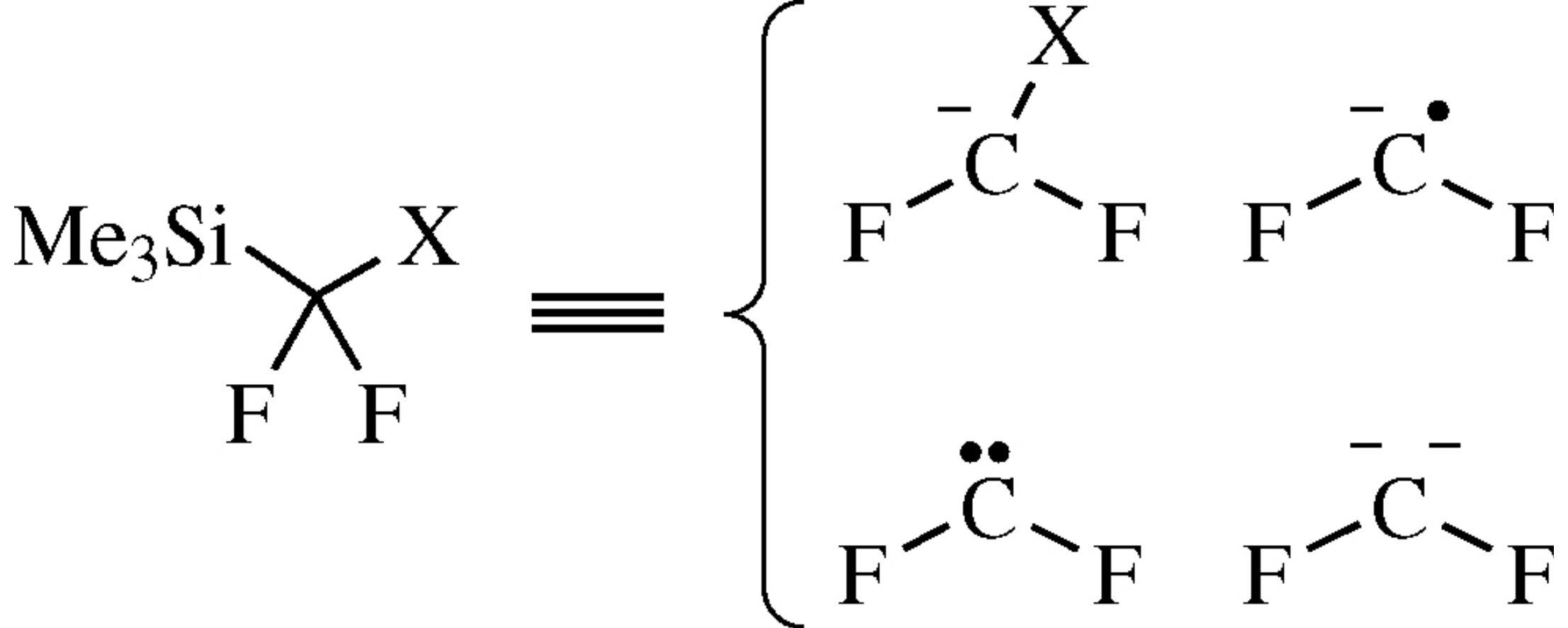
α-Fluorinated organosilanes Me3SiCF2X serve as versatile reagents for the synthesis of organofluorine compounds. Most reactions start from Lewis base activation of carbon–silicon bond through the intermediacy of five-coordinate siliconate species. Interaction of these silanes with suitable electrophiles leads to products of nucleophilic fluoroalkylation; an alternative pathway involves generation of difluorocarbene. These reagents can also be considered as equivalents of difluoromethylene radical anion and bis(carbanion) when carbon–heteroatom bond C–X is activated.
References
1.

Dolbier W.R.
Journal of Fluorine Chemistry,
2005
2.
10.1016/j.mencom.2015.07.001_sbref0005b
Kirsch
Modern Fluoroorganic Chemistry,
2004
3.
(a) K. Müller, C. Faeh and F. Diederich, Science, 2007, 317, 1881; (b) J. Wang, M. Sanchez-Roselló, J.L. Aceña, C. del Pozo, A.E. Sorochinsky, S. Fustero, V.A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432; (c) D. O’Hagan, J. Fluorine Chem., 2010, 131, 1071; (d) W. K. Hagmann, J. Med. Chem., 2008, 51, 4359; (e) Fluorine in Medicinal Chemistry and Chemical Biology, ed. I. Ojima, John Wiley & Sons, Chichester, 2009; (f) J.-P. Begue and D. Bonnet-Delpon, Bioorganic and Medicinal Chemistry of Fluorine, Wiley-VCH, Weinheim, 2008.
4.

Liang T., Neumann C.N., Ritter T.
Angewandte Chemie - International Edition,
2013
5.

Prakash G.K., Yudin A.K.
Chemical Reviews,
1997
6.

Liu X., Xu C., Wang M., Liu Q.
Chemical Reviews,
2014
7.
10.1016/j.mencom.2015.07.001_sbref0020c
Dilman
Eur. J. Org. Chem.,
2011
8.

Singh R.P., Shreeve J.M.
Tetrahedron,
2000
9.

Uneyama K.
Journal of Fluorine Chemistry,
2008
10.

Prakash G.K., Krishnamurti R., Olah G.A.
Journal of the American Chemical Society,
1989
11.

Ruppert I., Schlich K., Volbach W.
Tetrahedron Letters,
1984
12.
10.1016/j.mencom.2015.07.001_bib0035
Tyutyunov
Fluorine Notes,
2011
13.

Zhao Y., Huang W., Zheng J., Hu J.
Organic Letters,
2011
14.
10.1016/j.mencom.2015.07.001_sbref0040b
Tyutyunov
Fluorine Notes,
2011
15.

Fier P.S., Hartwig J.F.
Journal of the American Chemical Society,
2012
16.

Matheis C., Jouvin K., Goossen L.J.
Organic Letters,
2014
17.

Jiang X., Chen Z., Xu X., Qing F.
Organic Chemistry Frontiers,
2014
18.
10.1016/j.mencom.2015.07.001_sbref0040f
Gu
Nat. Commun.,
2014
19.

Kosobokov M.D., Dilman A.D., Levin V.V., Struchkova M.I.
Journal of Organic Chemistry,
2012
20.

Li L., Wang F., Ni C., Hu J.
Angewandte Chemie - International Edition,
2013
21.

Yudin A.K., Prakash G.K., Deffieux D., Bradley M., Bau R., Olah G.A.
Journal of the American Chemical Society,
1997
22.

Kosobokov M.D., Levin V.V., Struchkova M.I., Dilman A.D.
Organic Letters,
2014
23.

Kosobokov M.D., Levin V.V., Zemtsov A.A., Struchkova M.I., Korlyukov A.A., Arkhipov D.E., Dilman A.D.
Organic Letters,
2014
24.

Prakash G.K., Hu J., Olah G.A.
Journal of Organic Chemistry,
2003
25.

Pohmakotr M., Soorukram D., Kuhakarn C., Reutrakul V.
Synlett,
2014
26.

Ni C., Hu J.
Tetrahedron Letters,
2005
27.

Alexandrova A.V., Beier P.
Journal of Fluorine Chemistry,
2009
28.

Mizuta S., Shibata N., Akiti S., Fujimoto H., Nakamura S., Toru T.
Organic Letters,
2007
29.

Levin V.V., Dilman A.D., Belyakov P.A., Struchkova M.I., Tartakovsky V.A.
European Journal of Organic Chemistry,
2008
30.

Kosobokov M.D., Dilman A.D., Struchkova M.I., Belyakov P.A., Hu J.
Journal of Organic Chemistry,
2012
31.

Dilman A.D., Arkhipov D.E., Levin V.V., Belyakov P.A., Korlyukov A.A., Struchkova M.I., Tartakovsky V.A.
Journal of Organic Chemistry,
2007
32.
Dilman A.D., Levin V.V., Belyakov P.A., Korlyukov A.A., Struchkova M.I., Tartakovsky V.A.
Mendeleev Communications,
2009
33.

Dilman A.D., Arkhipov D.E., Levin V.V., Belyakov P.A., Korlyukov A.A., Struchkova M.I., Tartakovsky V.A.
Journal of Organic Chemistry,
2008
34.

Sani M., Volonterio A., Zanda M.
ChemMedChem,
2007
35.

Gauthier J.Y., Chauret N., Cromlish W., Desmarais S., Duong L.T., Falgueyret J., Kimmel D.B., Lamontagne S., Léger S., LeRiche T., Li C.S., Massé F., McKay D.J., Nicoll-Griffith D.A., Oballa R.M., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
36.

Kosobokov M.D., Struchkova M.I., Arkhipov D.E., Korlyukov A.A., Dilman A.D.
Journal of Fluorine Chemistry,
2013
37.
10.1016/j.mencom.2015.07.001_bib0105
Kosobokov
Russ. Chem. Bull., Int. Ed., 2014, 63, 549.,
2014
38.

Li Y., Hu J.
Angewandte Chemie - International Edition,
2007
39.

Singh R.P., Kirchmeier R.L., Shreeve J.M.
Organic Letters,
1999
40.

Sosnovskikh V.Y., Sevenard D.V., Usachev B.I., Röschenthaler G.-.
Tetrahedron Letters,
2003
41.

Sosnovskikh V.Y., Usachev B.I., Sevenard D.V., Röschenthaler G.
Journal of Organic Chemistry,
2003
42.

Sosnovskikh V.Y., Usachev B.I., Sevenard D.V., Röschenthaler G.
Journal of Fluorine Chemistry,
2005
43.
10.1016/j.mencom.2015.07.001_sbref0120d
Sosnovskikh
Russ. Chem. Bull., Int. Ed., 2006, 55, 1687.,
2006
44.

Dilman A.D., Levin V.V., Belyakov P.A., Struchkova M.I., Tartakovsky V.A.
Tetrahedron Letters,
2008
45.

Zemtsov A.A., Levin V.V., Dilman A.D., Struchkova M.I., Tartakovsky V.A.
Journal of Fluorine Chemistry,
2011
46.

Zemtsov A.A., Levin V.V., Dilman A.D., Struchkova M.I., Belyakov P.A., Tartakovsky V.A.
Tetrahedron Letters,
2009
47.

Hu J., Ni C.
Synthesis,
2014
48.
Nefedov O.M., Volchkov N.V.
Mendeleev Communications,
2006
49.

Wang F., Luo T., Hu J., Wang Y., Krishnan H.S., Jog P.V., Ganesh S.K., Prakash G.K., Olah G.A.
Angewandte Chemie - International Edition,
2011
50.

Prakash G.K., Krishnamoorthy S., Ganesh S.K., Kulkarni A., Haiges R., Olah G.A.
Organic Letters,
2013
51.

Levin V.V., Dilman A.D., Belyakov P.A., Struchkova M.I., Tartakovsky V.A.
Journal of Fluorine Chemistry,
2009
52.

Levin V.V., Kozlov M.A., Song Y., Dilman A.D., Belyakov P.A., Struchkova M.I., Tartakovsky V.A.
Tetrahedron Letters,
2008
53.

Tsymbal A.V., Kosobokov M.D., Levin V.V., Struchkova M.I., Dilman A.D.
Journal of Organic Chemistry,
2014
54.

Wu S., Yu Q.
Acta Chimica Sinica,
1989
55.

Kosobokov M.D., Levin V.V., Struchkova M.I., Dilman A.D.
Organic Letters,
2015
56.

Fedorov O.V., Kosobokov M.D., Levin V.V., Struchkova M.I., Dilman A.D.
Journal of Organic Chemistry,
2015
57.
10.1016/j.mencom.2015.07.001_bib0180
Kageshima
Synlett,
2015
58.

Song X., Chang J., Zhu D., Li J., Xu C., Liu Q., Wang M.
Organic Letters,
2015
59.

Levin V.V., Zemtsov A.A., Struchkova M.I., Dilman A.D.
Organic Letters,
2013
60.

Levin V.V., Zemtsov A.A., Struchkova M.I., Dilman A.D.
Journal of Fluorine Chemistry,
2015
61.
10.1016/j.mencom.2015.07.001_bib0200
Smirnov
Russ. Chem. Bull., Int. Ed., 2014, 63, 2564.,
2014
62.

Zemtsov A.A., Kondratyev N.S., Levin V.V., Struchkova M.I., Dilman A.D.
Journal of Organic Chemistry,
2014
63.

Smirnov V.O., Struchkova M.I., Arkhipov D.E., Korlyukov A.A., Dilman A.D.
Journal of Organic Chemistry,
2014